Using Self-Organizing Map Algorithm to Reveal Stabilities of Parameter Sensitivity Rankings in Microbial Kinetic Models: A Case for Microalgae
Abstract
:1. Introduction
2. Methodology
2.1. Algae Model Parameter Sensitivity Analysis
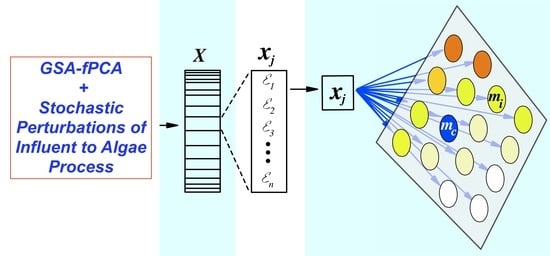
2.2. SOM Training on Parameter Sensitivity Index and Ranking
3. Results
3.1. Effectiveness of GSA-fPCA in Calculating the Sensitivity Indices for Model Parameters Ranking
3.2. SOM Component Plane Projection of the Morris Sensitivity Index and Parameter Ranks
4. Discussion
5. Conclusions
6. Recommendations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
- (i)
- Algae model solution form:
- (ii)
- Projection of model variableontobasis functions:
- (iii)
- Calculation of Morris sensitivity indexusing basis function scores’s:
References
- Zhao, B.; Su, Y. Macro assessment of microalgae-based CO2 sequestration: Environmental and energy effects. Algal Res. 2020, 51, 102066. [Google Scholar] [CrossRef]
- Gaskill, M. NASA—Building Better Life Support Systems for Future Space Travel. 2016. Available online: https://www.nasa.gov/mission_pages/station/research/news/photobioreactor-better-life-support (accessed on 21 May 2022).
- A Scott, S.; Davey, M.P.; Dennis, J.S.; Horst, I.; Howe, C.J.; Lea-Smith, D.J.; Smith, A.G. Biodiesel from algae: Challenges and prospects. Curr. Opin. Biotechnol. 2010, 21, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tibbetts, S.M.; McGinn, P.J. Microalgae as Sources of High-Quality Protein for Human Food and Protein Supplements. Foods 2021, 10, 3002. [Google Scholar] [CrossRef] [PubMed]
- Solimeno, A.; Samsó, R.; Uggetti, E.; Sialve, B.; Steyer, J.-P.; Gabarró, A.; García, J. New mechanistic model to simulate microalgae growth. Algal Res. 2015, 12, 350–358. [Google Scholar] [CrossRef] [Green Version]
- Lee, E.; Jalalizadeh, M.; Zhang, Q. Growth kinetic models for microalgae cultivation: A review. Algal Res. 2015, 12, 497–512. [Google Scholar] [CrossRef]
- Marsullo, M.; Mian, A.; Ensinas, A.; Manente, G.; Lazzaretto, A.; Marechal, F. Dynamic Modeling of the Microalgae Cultivation Phase for Energy Production in Open Raceway Ponds and Flat Panel Photobioreactors. Front. Energy Res. 2015, 3, 41. [Google Scholar] [CrossRef] [Green Version]
- Darvehei, P.; Bahri, P.A.; Moheimani, N.R. Model development for the growth of microalgae: A review. Renew. Sustain. Energy Rev. 2018, 97, 233–258. [Google Scholar] [CrossRef]
- Fogler, H.S. Elements of Chemical Reaction Engineering, 5th ed.; Prentice Hall: Hoboken, NJ, USA, 2016. [Google Scholar]
- Hassam, S.; Ficara, E.; Leva, A.; Harmand, J. A generic and systematic procedure to derive a simplified model from the anaerobic digestion model No. 1 (ADM1). Biochem. Eng. J. 2015, 99, 193–203. [Google Scholar] [CrossRef]
- Bernard, O.; Hadj-Sadok, Z.; Dochain, D.; Genovesi, A.; Steyer, J.-P. Dynamical model development and parameter identification for an anaerobic wastewater treatment process. Biotechnol. Bioeng. 2001, 75, 424–438. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.D. Factorial Sampling Plans for Preliminary Computational Experiments. Technometrics 1991, 33, 161–174. [Google Scholar] [CrossRef]
- Fortela, D.L.B.; Farmer, K.; Zappi, A.; Sharp, W.W.; Revellame, E.; Gang, D.; Zappi, M. A Methodology for Global Sensitivity Analysis of Activated Sludge Models: Case Study with Activated Sludge Model No. 3 (ASM 3). Water Environ. Res. 2019, 91, 865–876. [Google Scholar] [CrossRef] [PubMed]
- Fortela, D.L.B.; Sharp, W.W.; Revellame, E.D.; Hernandez, R.; Gang, D.; Zappi, M.E. Computational evaluation for effects of feedstock variations on the sensitivities of biochemical mechanism parameters in anaerobic digestion kinetic models. Biochem. Eng. J. 2019, 143, 212–223. [Google Scholar] [CrossRef]
- Sumner, T.; Shephard, E.; Bogle, I.D.L. A methodology for global-sensitivity analysis of time-dependent outputs in systems biology modelling. J. R. Soc. Interface 2012, 9, 2156–2166. [Google Scholar] [CrossRef] [PubMed]
- Kohonen, T. Essentials of the self-organizing map. Neural Netw. 2013, 37, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Kohonen, T. Self-Organizing Maps, 3rd ed.; Infromation Sciences; Kohonen, T., Ed.; Springer: New York, NY, USA, 2001. [Google Scholar]
- Johnsson, M. (Ed.) Applications of Self-Organizing Maps; IntechOpen: London, UK, 2012. [Google Scholar]
- Ogwueleka, T.C.; Samson, B. The effect of hydraulic retention time on microalgae-based activated sludge process for Wupa sewage treatment plant, Nigeria. Environ. Monit. Assess. 2020, 192, 271. [Google Scholar] [CrossRef] [PubMed]
- Fortela, D.L.B.; Crawford, M.; Delattre, A.; Kowalski, S.; Lissard, M.; Fremin, A.; Sharp, W.; Revellame, E.; Hernandez, R.; Zappi, M. Using Self-Organizing Maps to Elucidate Patterns among Variables in Simulated Syngas Combustion. Clean Technol. 2020, 2, 156–169. [Google Scholar] [CrossRef]
- Vesanto, J.; Himberg, J.; Alhoniemi, E.; Parhankangas, J. SOM Toolbox for MATLAB 5; Helsinki University of Technology: Espoo, Finland, 2000. [Google Scholar]
- Kohonen, T. MATLAB Implementations and Applications of the Self-Organizing Map; Unigrafia Bookstore Helsinki: Helsinki, Finland, 2014. [Google Scholar]
- Bernard, O.; Rémond, B. Validation of a simple model accounting for light and temperature effect on microalgal growth. Bioresour. Technol. 2012, 123, 520–527. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.; Guo, J.; Ni, B.-J.; Wang, S.; Yang, Q.; Peng, Y. A Novel Protocol for Model Calibration in Biological Wastewater Treatment. Sci. Rep. 2015, 5, 8493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Béchet, Q.; Shilton, A.; Guieysse, B. Modeling the effects of light and temperature on algae growth: State of the art and critical assessment for productivity prediction during outdoor cultivation. Biotechnol. Adv. 2013, 31, 1648–1663. [Google Scholar] [CrossRef] [PubMed]




| Variable Definition | Symbol | Units | Lower Bound | Upper Bound |
|---|---|---|---|---|
| Ammonium nitrogen | g-NH4+-N/m3 | 15 | ||
| Ammonia nitrogen | g-NH3-N/m3 | 6 | ||
| Nitrate nitrogen | g-NO3−-N/m3 | 90 | ||
| Dissolved oxygen | g-O2/m3 | 10 | ||
| Dissolved carbon dioxide | g-CO2-C/m3 | 8 | ||
| Bicarbonate | g-HCO3−-C/m3 | 200 | ||
| Carbonate | g-CO32−-C/m3 | 12 | ||
| Hydrogen ions | g-H/m3 | |||
| Hydroxide ions | g-OH−-H/m3 | |||
| Microalgae biomass | g-COD/m3 | 200 |
| Parameter Definition | Symbol | Units | Nominal − 30% | Nominal | Nominal + 30% |
|---|---|---|---|---|---|
| Microalgae Processes | |||||
| Maximum growth rate of microalgae | d−1 | 1.36 | 1.84 | ||
| Endogenous respiration constant | d−1 | 0.085 | 0.115 | ||
| Inactivation constant | d−1 | 0.085 | 0.115 | ||
| Affinity constant of microalgae on carbon species | gC m−3 | 0.003672 | 0.004968 | ||
| CO2 inhibition constant of microalgae | gC m−3 | 102 | 138 | ||
| Affinity constant of microalgae on nitrogen species | gN m−3 | 0.085 | 0.115 | ||
| Affinity constant of microalgae on dissolved oxygen | gO2 m−3 | 0.17 | 0.23 | ||
| Photosynthetic Thermal Factor | |||||
| Optimum temperature for microalgae growth | 21.25 | 28.75 | |||
| Actual temperature for microalgae growth | 20 | varies | 40 | ||
| Normalized parameter | --- | 11.05 | 14.95 | ||
| Light Factor | |||||
| Parameter activation | (µE m−2)−1 | 0.00164475 | 0.00222525 | ||
| Parameter inhibition | (µE m−2)−1 | ||||
| Parameter production | s−1 | 0.1241 | 0.1460 | 0.1679 | |
| Parameter recovery | s−1 | 0.00040766 | 0.00055154 | ||
| Light Intensity | (µE m−2)−1 | 170 | 230 | ||
| Transfer of Gases to the Atmosphere | |||||
| Mass transfer coefficient for oxygen | d−1 | 3.4 | 4.6 | ||
| Mass transfer coefficient for dioxide carbon | d−1 | 0.595 | 0.805 | ||
| Mass transfer coefficient for ammonia | d−1 | 0.595 | 0.805 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fortela, D.L.B.; DeLattre, A.M.; Sharp, W.W.; Revellame, E.D.; Zappi, M.E. Using Self-Organizing Map Algorithm to Reveal Stabilities of Parameter Sensitivity Rankings in Microbial Kinetic Models: A Case for Microalgae. Clean Technol. 2023, 5, 38-50. https://doi.org/10.3390/cleantechnol5010003
Fortela DLB, DeLattre AM, Sharp WW, Revellame ED, Zappi ME. Using Self-Organizing Map Algorithm to Reveal Stabilities of Parameter Sensitivity Rankings in Microbial Kinetic Models: A Case for Microalgae. Clean Technologies. 2023; 5(1):38-50. https://doi.org/10.3390/cleantechnol5010003
Chicago/Turabian StyleFortela, Dhan Lord B., Alyssa M. DeLattre, Wayne W. Sharp, Emmanuel D. Revellame, and Mark E. Zappi. 2023. "Using Self-Organizing Map Algorithm to Reveal Stabilities of Parameter Sensitivity Rankings in Microbial Kinetic Models: A Case for Microalgae" Clean Technologies 5, no. 1: 38-50. https://doi.org/10.3390/cleantechnol5010003