Polymeric DNA Hydrogels and Their Applications in Drug Delivery for Cancer Therapy
Abstract
:1. Introduction
2. The Preparation and Drug Delivery Applications of DNA Hydrogels
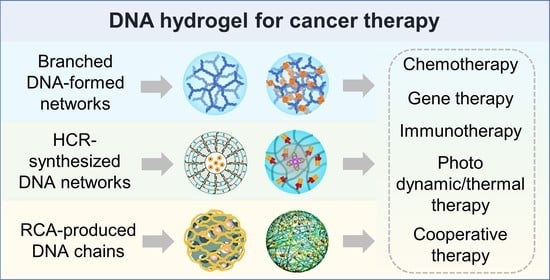
2.1. Branched DNA Formed Hydrogel
2.1.1. Branched DNA
2.1.2. Application in Cancer Therapy
2.2. HCR-Synthesized DNA Networks Formed Hydrogel
2.2.1. HCR-Synthesized DNA Networks
2.2.2. Application in Cancer Therapy
2.3. RCA-Produced DNA Chain-Based Hydrogel
2.3.1. RCA-Produced DNA Chain
2.3.2. Application in Cancer Therapy
3. Conclusions and Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hamidi, M.; Azadi, A.; Rafiei, P. Hydrogel nanoparticles in drug delivery. Adv. Drug Delivery. Rev. 2008, 60, 1638–1649. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Hydrogels for tissue engineering. Chem. Rev. 2001, 101, 1869–1879. [Google Scholar] [CrossRef]
- Peppas, N.A.; Hilt, J.Z.; Khademhosseini, A.; Langer, R. Hydrogels in biology and medicine: From molecular principles to bionanotechnology. Adv. Mater. 2006, 18, 1345–1360. [Google Scholar] [CrossRef]
- Jen, A.C.; Wake, M.C.; Mikos, A.G. Review: Hydrogels for cell immobilization. Biotechnol. Bioeng. 1996, 50, 357–364. [Google Scholar] [CrossRef]
- Langer, R.; Peppas, N.A. Advances in biomaterials, drug delivery, and bionanotechnology. AIChE J. 2003, 49, 2990–3006. [Google Scholar] [CrossRef]
- Del Valle, L.J.; Diaz, A.; Puiggali, J. Hydrogels for Biomedical Applications: Cellulose, Chitosan, and Protein/Peptide Derivatives. Gels 2017, 3, 27. [Google Scholar] [CrossRef] [Green Version]
- Seliktar, D.; Black, R.A.; Vito, R.P.; Nerem, R.M. Dynamic mechanical conditioning of collagen-gel blood vessel constructs induces remodeling in vitro. Ann. Biomed. Eng. 2000, 28, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Auger, F.A.; Rouabhia, M.; Goulet, F.; Berthod, F.; Moulin, V.; Germain, L. Tissue-engineered human skin substitutes developed from collagen-populated hydrated gels: Clinical and fundamental applications. Med. Biol. Eng. Comput. 1998, 36, 801–812. [Google Scholar] [CrossRef]
- Bao, Z.; Xian, C.; Yuan, Q.; Liu, G.; Wu, J. Natural Polymer-Based Hydrogels with Enhanced Mechanical Performances: Preparation, Structure, and Property. Adv. Healthc. Mater 2019, 8, e1900670. [Google Scholar] [CrossRef]
- Sanmartín-Masiá, E.; Poveda-Reyes, S.; Gallego Ferrer, G. Extracellular matrix–inspired gelatin/hyaluronic acid injectable hydrogels. Int. J. Polym. Mater. Po. 2016, 66, 280–288. [Google Scholar] [CrossRef]
- Ibrahim, A.G.; Sayed, A.Z.; Abd El-Wahab, H.; Sayah, M.M. Synthesis of a hydrogel by grafting of acrylamide-co-sodium methacrylate onto chitosan for effective adsorption of Fuchsin basic dye. Int. J. Biol. Macromol. 2020, 159, 422–432. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.G.; Elkony, A.M.; El-Bahy, S.M. Methylene blue uptake by gum arabic/acrylic amide/3-allyloxy-2-hydroxy-1-propanesulfonic acid sodium salt semi-IPN hydrogel. Int. J. Biol. Macromol. 2021, 186, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Yi, H. Facile Micromolding-Based Fabrication of Biopolymeric-Synthetic Hydrogel Microspheres with Controlled Structures for Improved Protein Conjugation. Chem. Mater. 2015, 27, 3988–3998. [Google Scholar] [CrossRef] [Green Version]
- Ibrahim, A.G.; Abdel Hai, F.; Abd El-Wahab, H.; Aboelanin, H. Methylene blue removal using a novel hydrogel containing 3-Allyloxy-2-hydroxy-1-propanesulfonic acid sodium salt. Adv. Polym. Technol. 2018, 37, 3561–3573. [Google Scholar] [CrossRef]
- Wichterle, O.; Lim, D. Hydrophilic Gels for Biological Use. Nature 1960, 185, 117–118. [Google Scholar] [CrossRef]
- Ende, M.T.A.; Peppas, N.A. Analysis of drug distribution in hydrogels using fourier transform infrared microscopy. Pharm. Res. 1995, 12, 2030–2035. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Otte, A.; Park, K. Evolution of drug delivery systems: From 1950 to 2020 and beyond. J. Control. Release 2022, 342, 53–65. [Google Scholar] [CrossRef]
- Stile, R.A.; Burghardt, W.R.; Healy, K.E. Synthesis and Characterization of Injectable Poly(N-isopropylacrylamide)-Based Hydrogels That Support Tissue Formation in Vitro. Macromolecules 1999, 32, 7370–7379. [Google Scholar] [CrossRef]
- Heskins, M.; Guillet, J.E. Solution Properties of Poly(N-isopropylacrylamide). J. Macromol. Sci., Pure Appl. Chem. 1968, 2, 1441–1455. [Google Scholar] [CrossRef]
- Li, X.; Gao, Y.; Serpe, M.J. Stimuli-Responsive Assemblies for Sensing Applications. Gels 2016, 2, 8. [Google Scholar] [CrossRef]
- Li, F.; Zhang, X.; Hu, S.X.; Lv, Z.Y.; Lv, J.G.; Yu, W.T.; Xu, X.H.; Yang, D.Y. Bioinspired Mechanically Responsive Hydrogel upon Redox Mediated by Dynamic Coordination between Telluroether and Platinum Ions. Chem. Mater. 2020, 32, 2156–2165. [Google Scholar] [CrossRef]
- Du, X.; Zhou, J.; Shi, J.; Xu, B. Supramolecular Hydrogelators and Hydrogels: From Soft Matter to Molecular Biomaterials. Chem. Rev. 2015, 115, 13165–13307. [Google Scholar] [CrossRef]
- Tang, J.D.; Mura, C.; Lampe, K.J. Stimuli-Responsive, Pentapeptide, Nanofiber Hydrogel for Tissue Engineering. J. Am. Chem. Soc. 2019, 141, 4886–4899. [Google Scholar] [CrossRef] [PubMed]
- Lungu, R.; Paun, M.A.; Peptanariu, D.; Ailincai, D.; Marin, L.; Nichita, M.V.; Paun, V.A.; Paun, V.P. Biocompatible Chitosan-Based Hydrogels for Bioabsorbable Wound Dressings. Gels 2022, 8, 107. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.; Shao, Y.; Zhou, X.; Liu, Q.; Zhu, Z.; Zhou, B.; Dong, Y.; Stephanopoulos, N.; Gui, S.; Yan, H.; et al. Highly Permeable DNA Supramolecular Hydrogel Promotes Neurogenesis and Functional Recovery after Completely Transected Spinal Cord Injury. Adv. Mater. 2021, 33, e2102428. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.M.; Zhou, W.; Chen, Z.Z.; Hu, Y.; Yang, Y.; Zhang, G.Z.; Yang, Z.H. A Supramolecular Hydrogel Enabled by the Synergy of Hydrophobic Interaction and Quadruple Hydrogen Bonding. Gels 2022, 8, 244. [Google Scholar] [CrossRef]
- Park, N.; Um, S.H.; Funabashi, H.; Xu, J.; Luo, D. A cell-free protein-producing gel. Nat. Mater. 2009, 8, 432–437. [Google Scholar] [CrossRef]
- Mo, F.; Jiang, K.; Zhao, D.; Wang, Y.; Song, J.; Tan, W. DNA hydrogel-based gene editing and drug delivery systems. Adv. Drug Delivery. Rev. 2021, 168, 79–98. [Google Scholar] [CrossRef]
- Nagahara, S.; Matsuda, T. Hydrogel formation via hybridization of oligonucleotides derivatized in water-soluble vinyl polymers. Polym. Gels Netw. 1996, 4, 111–127. [Google Scholar] [CrossRef]
- Seeman, N.C.; Sleiman, H.F. DNA nanotechnology. Nat. Rev. Mater. 2018, 3, 17068. [Google Scholar] [CrossRef]
- Seeman, N.C. DNA in a material world. Nature 2003, 421, 427–431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, Y.; Yao, C.; Zhu, Y.; Yang, L.; Luo, D.; Yang, D. DNA Functional Materials Assembled from Branched DNA: Design, Synthesis, and Applications. Chem. Rev. 2020, 120, 9420–9481. [Google Scholar] [CrossRef]
- Prausnitz, M.R.; Mitragotri, S.; Langer, R. Current status and future potential of transdermal drug delivery. Nat. Rev. Drug Discov. 2004, 3, 115–124. [Google Scholar] [CrossRef]
- Ullyot, G.E.; Ullyot, B.H.; Slater, L.B. The metamorphosis of smith-kline & french laboratories to smith kline beecham: 1925–1998. Bull Hist Chem 2000, 25, 16–21. [Google Scholar]
- Park, K.; Skidmore, S.; Hadar, J.; Garner, J.; Park, H.; Otte, A.; Soh, B.K.; Yoon, G.; Yu, D.J.; Yun, Y.; et al. Injectable, long-acting PLGA formulations: Analyzing PLGA and understanding microparticle formation. J. Control. Release 2019, 304, 125–134. [Google Scholar] [CrossRef]
- Davis, F.F. Commentary—The origin of pegnology. Adv. Drug Deliv. Rev. 2002, 54, 457–458. [Google Scholar] [CrossRef]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal Formulations in Clinical Use: An Updated Review. Pharmaceutics 2017, 9, 33. [Google Scholar] [CrossRef]
- Neerooa, B.; Ooi, L.T.; Shameli, K.; Dahlan, N.A.; Islam, J.M.M.; Pushpamalar, J.; Teow, S.Y. Development of Polymer-Assisted Nanoparticles and Nanogels for Cancer Therapy: An Update. Gels 2021, 7, 60. [Google Scholar] [CrossRef]
- Shahrousvand, M.; Hajikhani, M.; Nazari, L.; Aghelinejad, A.; Shahrousvand, M.; Irani, M.; Rostami, A. Preparation of colloidal nanoparticles PVA-PHEMA from hydrolysis of copolymers of PVAc-PHEMA as anticancer drug carriers. Nanotechnology 2022, 33, 14. [Google Scholar] [CrossRef]
- Madej, M.; Kurowska, N.; Strzalka-Mrozik, B. Polymeric Nanoparticles—Tools in a Drug Delivery System in Selected Cancer Therapies. Appl. Sci. 2022, 12, 9479. [Google Scholar] [CrossRef]
- Korzhikov-Vlakh, V.; Tennikova, T. Nanogels Capable of Triggered Release. In Tunable Hydrogels: Smart Materials for Biomedical Applications; Lavrentieva, A., Pepelanova, I., Seliktar, D., Eds.; Advances in Biochemical Engineering-Biotechnology; Springer International Publishing Ag: Cham, Switzerland, 2021; Volume 178, pp. 99–146. [Google Scholar]
- Soni, K.S.; Desale, S.S.; Bronich, T.K. Nanogels: An overview of properties, biomedical applications and obstacles to clinical translation. J. Control. Release 2016, 240, 109–126. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Qu, Y.; Zhang, Z.; Huang, H.; Xu, Y.; Shen, F.; Wang, L.; Sun, L. Injectable DNA Hydrogel-Based Local Drug Delivery and Immunotherapy. Gels 2022, 8, 400. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Jiao, X.; Liu, S.; Hao, M.; Cheng, S.; Zhang, P.; Wen, Y. Functional DNA-based hydrogel intelligent materials for biomedical applications. J. Mater. Chem. B 2020, 8, 1991–2009. [Google Scholar] [CrossRef]
- Li, J.; Zheng, C.; Cansiz, S.; Wu, C.C.; Xu, J.H.; Cui, C.; Liu, Y.; Hou, W.J.; Wang, Y.Y.; Zhang, L.Q.; et al. Self-assembly of DNA Nanohydrogels with Controllable Size and Stimuli-Responsive Property for Targeted Gene Regulation Therapy. J. Am. Chem. Soc. 2015, 137, 1412–1415. [Google Scholar] [CrossRef]
- Shao, Y.; Sun, Z.Y.; Wang, Y.; Zhang, B.D.; Liu, D.; Li, Y.M. Designable Immune Therapeutical Vaccine System Based on DNA Supramolecular Hydrogels. ACS Appl. Mater. Interfaces 2018, 10, 9310–9314. [Google Scholar] [CrossRef]
- Song, J.; Lee, M.; Kim, T.; Na, J.; Jung, Y.; Jung, G.Y.; Kim, S.; Park, N. A RNA producing DNA hydrogel as a platform for a high performance RNA interference system. Nat. Commun. 2018, 9, 4331. [Google Scholar] [CrossRef] [Green Version]
- Song, J.; Im, K.; Hwang, S.; Hur, J.; Nam, J.; Ahn, G.O.; Hwang, S.; Kim, S.; Park, N. DNA hydrogel delivery vehicle for light-triggered and synergistic cancer therapy. Nanoscale 2015, 7, 9433–9437. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Zhang, Y.; Sun, J.; Ouyang, J.; Na, N. SiRNA-templated 3D framework nucleic acids for chemotactic recognition, and programmable and visualized precise delivery for synergistic cancer therapy. Chem. Sci. 2021, 12, 15353–15361. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.; Lai, W.; Wang, F.; Li, L.; Fan, C.; Pei, H. Programming Drug Delivery Kinetics for Active Burst Release with DNA Toehold Switches. J. Am. Chem. Soc. 2019, 141, 20354–20364. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Lv, Z.; Zhang, X.; Dong, Y.; Ding, X.; Li, Z.; Li, S.; Yao, C.; Yang, D. Supramolecular Self-Assembled DNA Nanosystem for Synergistic Chemical and Gene Regulations on Cancer Cells. Angew. Chem. Int. Ed. 2021, 60, 25557–25566. [Google Scholar] [CrossRef]
- Li, F.; Yu, W.; Zhang, J.; Dong, Y.; Ding, X.; Ruan, X.; Gu, Z.; Yang, D. Spatiotemporally programmable cascade hybridization of hairpin DNA in polymeric nanoframework for precise siRNA delivery. Nat. Commun. 2021, 12, 1138. [Google Scholar] [CrossRef]
- Liao, W.C.; Lilienthal, S.; Kahn, J.S.; Riutin, M.; Sohn, Y.S.; Nechushtai, R.; Willner, I. pH- and ligand-induced release of loads from DNA-acrylamide hydrogel microcapsules. Chem. Sci. 2017, 8, 3362–3373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, W.; Jiang, T.; Lu, Y.; Reiff, M.; Mo, R.; Gu, Z. Cocoon-like self-degradable DNA nanoclew for anticancer drug delivery. J. Am. Chem. Soc. 2014, 136, 14722–14725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, F.; Song, N.; Dong, Y.; Li, S.; Li, L.; Liu, Y.; Li, Z.; Yang, D. A Proton-Activatable DNA-Based Nanosystem Enables Co-Delivery of CRISPR/Cas9 and DNAzyme for Combined Gene Therapy. Angew. Chem. Int. Ed. 2022, 61, e202116569. [Google Scholar] [CrossRef]
- Yao, C.; Zhu, C.; Tang, J.; Ou, J.; Zhang, R.; Yang, D. T Lymphocyte-Captured DNA Network for Localized Immunotherapy. J. Am. Chem. Soc. 2021, 143, 19330–19340. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Li, L.; Li, F.; Liu, C.; Huang, M.; Li, J.; Gao, F.; Ruan, X.; Yang, D. An Energy-Storing DNA-Based Nanocomplex for Laser-Free Photodynamic Therapy. Adv. Mater. 2022, 34, e2109920. [Google Scholar] [CrossRef]
- Zhao, J.; Guo, Y.; Tong, Z.; Zhang, R.; Yao, C.; Yang, D. Spatio-Temporal Controlled Gene-Chemo Drug Delivery in a DNA Nanocomplex to Overcome Multidrug Resistance of Cancer Cells. ACS Appl. Bio. Mater. 2022, 5, 3795–3805. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Dong, Y.; Yao, C.; Yang, D. Construction of Branched DNA-based Nanostructures for Diagnosis, Therapeutics and Protein Engineering. Chem. Asian J. 2022, 17, e202200310. [Google Scholar] [CrossRef]
- Campolongo, M.J.; Tan, S.J.; Xu, J.; Luo, D. DNA nanomedicine: Engineering DNA as a polymer for therapeutic and diagnostic applications. Adv. Drug. Deliv. Rev. 2010, 62, 606–616. [Google Scholar] [CrossRef]
- Um, S.H.; Lee, J.B.; Park, N.; Kwon, S.Y.; Umbach, C.C.; Luo, D. Enzyme-catalysed assembly of DNA hydrogel. Nat. Mater. 2006, 5, 797–801. [Google Scholar] [CrossRef]
- Hartman, M.R.; Yang, D.; Tran, T.N.; Lee, K.; Kahn, J.S.; Kiatwuthinon, P.; Yancey, K.G.; Trotsenko, O.; Minko, S.; Luo, D. Thermostable branched DNA nanostructures as modular primers for polymerase chain reaction. Angew. Chem. Int. Ed. 2013, 52, 8699–8702. [Google Scholar] [CrossRef] [PubMed]
- Cheng, E.; Xing, Y.; Chen, P.; Yang, Y.; Sun, Y.; Zhou, D.; Xu, L.; Fan, Q.; Liu, D. A pH-Triggered, Fast-Responding DNA Hydrogel. Angew. Chem. Int. Ed. 2009, 48, 7660–7663. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Cheng, E.; Yang, Y.; Chen, P.; Zhang, T.; Sun, Y.; Yang, Z.; Liu, D. Self-assembled DNA hydrogels with designable thermal and enzymatic responsiveness. Adv. Mater. 2011, 23, 1117–1121. [Google Scholar] [CrossRef] [PubMed]
- Nishida, Y.; Ohtsuki, S.; Araie, Y.; Umeki, Y.; Endo, M.; Emura, T.; Hidaka, K.; Sugiyama, H.; Takahashi, Y.; Takakura, Y.; et al. Self-assembling DNA hydrogel-based delivery of immunoinhibitory nucleic acids to immune cells. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 123–130. [Google Scholar] [CrossRef]
- Zhang, J.; Guo, Y.; Pan, G.; Wang, P.; Li, Y.; Zhu, X.; Zhang, C. Injectable Drug-Conjugated DNA Hydrogel for Local Chemotherapy to Prevent Tumor Recurrence. ACS Appl. Mater. Interfaces 2020, 12, 21441–21449. [Google Scholar] [CrossRef]
- Song, W.; Song, P.; Sun, Y.; Zhang, Z.; Zhou, H.; Zhang, X.; He, P. Self-Assembly of Multifunctional DNA Nanohydrogels with Tumor Microenvironment-Responsive Cascade Reactions for Cooperative Cancer Therapy. ACS Biomater. Sci. Eng. 2021, 7, 5165–5174. [Google Scholar] [CrossRef]
- Wei, H.; Zhao, Z.; Wang, Y.; Zou, J.; Lin, Q.; Duan, Y. One-Step Self-Assembly of Multifunctional DNA Nanohydrogels: An Enhanced and Harmless Strategy for Guiding Combined Antitumor Therapy. ACS Appl. Mater. Interfaces 2019, 11, 46479–46489. [Google Scholar] [CrossRef]
- Tang, W.; Han, L.; Duan, S.; Lu, X.; Wang, Y.; Wu, X.; Liu, J.; Ding, B. An Aptamer-Modified DNA Tetrahedron-Based Nanogel for Combined Chemo/Gene Therapy of Multidrug-Resistant Tumors. ACS Appl. Bio. Mater. 2021, 4, 7701–7707. [Google Scholar] [CrossRef]
- Dirks, R.M.; Pierce, N.A. Triggered amplification by hybridization chain reaction. Proc. Natl. Acad. Sci. USA 2004, 101, 15275–15278. [Google Scholar] [CrossRef] [Green Version]
- Lu, Z.; Shi, Y.; Ma, Y.; Jia, B.; Li, X.; Guan, X.; Li, Z. Fast and specific enrichment and quantification of cancer-related exosomes by DNA-nanoweight-assisted centrifugation. Anal. Chem. 2022, 94, 9466–9471. [Google Scholar] [CrossRef]
- Wan, S.; Zhang, L.; Wang, S.; Liu, Y.; Wu, C.; Cui, C.; Sun, H.; Shi, M.; Jiang, Y.; Li, L.; et al. Molecular Recognition-Based DNA Nanoassemblies on the Surfaces of Nanosized Exosomes. J. Am. Chem. Soc. 2017, 139, 5289–5292. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Wu, M.; Hu, Q.; Bai, H.; Zhang, S.; Shen, Y.; Tang, G.; Ping, Y. Redox-Activated Light-Up Nanomicelle for Precise Imaging-Guided Cancer Therapy and Real-Time Pharmacokinetic Monitoring. ACS Nano 2016, 10, 11385–11396. [Google Scholar] [CrossRef]
- Lee, J.B.; Peng, S.; Yang, D.; Roh, Y.H.; Funabashi, H.; Park, N.; Rice, E.J.; Chen, L.; Long, R.; Wu, M.; et al. A mechanical metamaterial made from a DNA hydrogel. Nat. Nanotechnol. 2012, 7, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Abdullah, R.; Hu, X.; Bai, H.; Fan, H.; He, L.; Liang, H.; Zou, J.; Liu, Y.; Sun, Y.; et al. Engineering of Bioinspired, Size-Controllable, Self-Degradable Cancer-Targeting DNA Nanoflowers via the Incorporation of an Artificial Sandwich Base. J. Am. Chem. Soc. 2019, 141, 4282–4290. [Google Scholar] [CrossRef] [PubMed]
- Zu, H.; Gao, D. Non-viral Vectors in Gene Therapy: Recent Development, Challenges, and Prospects. AAPS J. 2021, 23, 78. [Google Scholar] [CrossRef]
- Han, S.; Park, Y.; Kim, H.; Nam, H.; Ko, O.; Lee, J.B. Double Controlled Release of Therapeutic RNA Modules through Injectable DNA-RNA Hybrid Hydrogel. ACS Appl. Mater. Interfaces 2020, 12, 55554–55563. [Google Scholar] [CrossRef] [PubMed]
- Emens, L.A.; Ascierto, P.A.; Darcy, P.K.; Demaria, S.; Eggermont, A.M.M.; Redmond, W.L.; Seliger, B.; Marincola, F.M. Cancer immunotherapy: Opportunities and challenges in the rapidly evolving clinical landscape. Eur. J. Cancer 2017, 81, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Sun, W.; Wright, G.; Wang, A.Z.; Gu, Z. Inflammation-Triggered Cancer Immunotherapy by Programmed Delivery of CpG and Anti-PD1 Antibody. Adv. Mater. 2016, 28, 8912–8920. [Google Scholar] [CrossRef] [Green Version]
- Fan, W.; Huang, P.; Chen, X. Overcoming the Achilles’ heel of photodynamic therapy. Chem. Soc. Rev. 2016, 45, 6488–6519. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, Z.; Zuo, D.; Li, L.; Li, F.; Yang, D. A Synergistic DNA-polydopamine-MnO2 Nanocomplex for Near-Infrared-Light-Powered DNAzyme-Mediated Gene Therapy. Nano Lett. 2021, 21, 5377–5385. [Google Scholar] [CrossRef]








| Type of DNA Hydrogels | Formulation | Strategy Advantages | Strategy Limitations | Delivered Drugs | Application in Cancer Therapy | Ref. |
|---|---|---|---|---|---|---|
| Non-enzyme-mediated branched hydrogel | stimuli responsiveness (pH, temperature) | controllable symmetry; multivalency; enzyme-free | high concentration for preparing hydrogel | ASOs; CPG | immunotherapy | [45,46] |
| Enzyme-mediated branched hydrogel | strand extension (ligase) | controllable symmetry; multivalency; short reaction time | high concentration for preparing hydrogel; high cost | Camptothecin; DOX; transcribed siRNA | chemotherapy; chemo-photo thermal synergistic therapy gene therapy | [47,48] |
| Pure hydrogel formed by HCR-synthesized networks | linear/clamped amplification | isothermal amplification; enzyme-free; convenient operation | high requirements for sequence design; require reaction carrier (AuNP, cell membrane, …) for HCR | DOX; siRNA | chemo-gene synergistic therapy; chemotherapy | [49,50] |
| Hybrid hydrogel formed by HCR-synthesized networks | hybrid with other organic/inorganic material before HCR amplification | isothermal amplification; enzyme-free; high stability | high requirements for sequence design; lower initiation efficiency influenced by complex conformation | siRNA; DOX | gene therapy; chemotherapy | [51,52,53] |
| Pure hydrogel formed by RCA-produced long DNA chain | physical crosslinking after RCA reaction | isothermal amplification; convenient operation | high requirements for sequence (template and primer) design; high cost low stability | DOX; DNAzyme and CRISPR/Cas9 system; siRNA; CPG | chemotherapy; gene therapy; immunotherapy | [54,55,56] |
| Hybrid hydrogel formed by RCA-produced long DNA chain | hybrid with other organic/inorganic material after RCA amplification | isothermal amplification high stability | high requirements for sequence (template and primer) design high cost; complex operation for hybridization | SiPcCl2; DOX; ASOs | gene-photo thermal synergistic therapy; chemo-gene synergistic therapy | [57,58] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Song, W.; Li, F. Polymeric DNA Hydrogels and Their Applications in Drug Delivery for Cancer Therapy. Gels 2023, 9, 239. https://doi.org/10.3390/gels9030239
Li J, Song W, Li F. Polymeric DNA Hydrogels and Their Applications in Drug Delivery for Cancer Therapy. Gels. 2023; 9(3):239. https://doi.org/10.3390/gels9030239
Chicago/Turabian StyleLi, Jing, Wenzhe Song, and Feng Li. 2023. "Polymeric DNA Hydrogels and Their Applications in Drug Delivery for Cancer Therapy" Gels 9, no. 3: 239. https://doi.org/10.3390/gels9030239