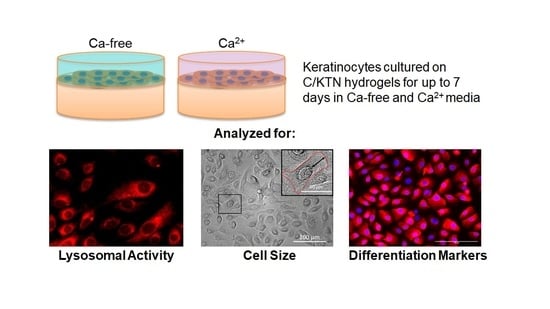
Keratin Promotes Differentiation of Keratinocytes Seeded on Collagen/Keratin Hydrogels
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collagen Extraction
2.2. Kerateine Extraction
2.3. Cell Culture
2.4. Collagen/Kerateine Hydrogel Fabrication and Cell Seeding
2.5. Cell Viability Assay
2.6. Glucocerebrosidase (GBA) Assay
2.7. Lysotracker Staining
2.8. Differentiation Marker Staining
2.9. Flow Cytometry
2.10. Statistical Analysis
3. Results
3.1. Lysosomal Activity Influenced by KTN and Increased Calcium Concentration
3.2. Keratinocyte Size Influenced by KTN and Increased Calcium Concentration
3.3. Involucrin and CK14 Expression
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eckert, R.L.; Efimova, T.; Dashti, S.R.; Balasubramanian, S.; Deucher, A.; Crish, J.F.; Sturniolo, M.; Bone, F. Keratinocyte survival, differentiation, and death: Many roads lead to mitogen-activated protein kinase. In Journal of Investigative Dermatology Symposium Proceedings; Blackwell Publishing Inc.: Oxford, UK, 2002; Volume 7, pp. 36–40. [Google Scholar]
- Eckert, R.L.; Crish, J.F.; Robinson, N.A. The Epidermal Keratinocyte as A Model for the Study of Gene Regulation and Cell Differentiation; Elsevier: Amsterdam, The Netherlands, 1997; Volume 77. [Google Scholar]
- Eckhart, L.; Lippens, S.; Tschachler, E.; Declercq, W. Cell death by cornification. Biochim. Biophys. Acta—Mol. Cell Res. 2013, 1833, 3471–3480. [Google Scholar] [CrossRef]
- Bikle, D.D.; Xie, Z.; Tu, C.L. Calcium regulation of keratinocyte differentiation. Expert Rev. Endocrinol. Metab. 2012, 7, 461–472. [Google Scholar] [CrossRef] [Green Version]
- Seo, E.Y.; Namkung, J.H.; Lee, K.M.; Lee, W.H.; Im, M.; Kee, S.H.; Geon, T.P.; Yang, J.M.; Seo, Y.J.; Park, J.K.; et al. Analysis of calcium-inducible genes in keratinocytes using suppression subtractive hybridization and cDNA microarray. Genomics 2005, 86, 528–538. [Google Scholar] [CrossRef] [PubMed]
- Ng, D.C.; Su, M.J.; Kim, R.; Bikle, D.D. Regulation of involucrin gene expression by calcium in normal human keratinocytes. Front. Biosci. 1996, 1, 16–24. [Google Scholar] [CrossRef]
- Rothnagel, J.A.; Greenhalgh, D.A.; Gagne, T.A.; Longley, M.A.; Roop, D.R. Identification of a Calcium-Inducible, Epidermal-Specific Regulatory Element in the 3’-Flanking Region of the Human Keratin 1 Gene. J. Investig. Dermatol. 1993, 101, 506–513. [Google Scholar] [CrossRef] [Green Version]
- Huff, C.A.; Yuspa, S.H.; Rosenthal, D. Identification of control elements 3’ to the human keratin 1 gene that regulate cell type and differentiation-specific expression—PubMed. J. Biol Chem. 1993, 377–384. Available online: https://pubmed.ncbi.nlm.nih.gov/7677999/ (accessed on 29 September 2022). [CrossRef]
- Elsholz, F.; Harteneck, C.; Muller, W.; Friedland, K. Calcium—A central regulator of keratinocyte differentiation in health and disease. Eur. J. Dermatol. 2014, 24, 650–661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warhol, M.J.; Roth, J.; Lucocq, J.M.; Pinkus, G.S.; Rice, R.H. Immuno-ultrastructural localization of involucrin in squamous epithelium and cultured keratinocytes. J. Histochem. Cytochem. 1985, 33, 141–149. [Google Scholar] [CrossRef]
- Hennings, H.; Michael, D.; Cheng, C.; Steinert, P.; Holbrook, K.; Yuspa, S.H. Calcium regulation of growth and differentiation of mouse epidermal cells in culture. Cell 1980, 19, 245–254. [Google Scholar] [CrossRef]
- Zamansky, G.B.; Nguyen, U.; Chou, I.N. An immunofluorescence study of the calcium-induced coordinated reorganization of microfilaments, keratin intermediate filaments, and microtubules in cultured human epidermal keratinocytes. J. Investig. Dermatol. 1991, 97, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Monteleon, C.L.; Agnihotri, T.; Dahal, A.; Liu, M.; Rebecca, V.W.; Beatty, G.L.; Amaravadi, R.K.; Ridky, T.W. Lysosomes Support the Degradation, Signaling, and Mitochondrial Metabolism Necessary for Human Epidermal Differentiation. J. Investig. Dermatol. 2018, 138, 1945–1954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borowiec, A.S.; Delcourt, P.; Dewailly, E.; Bidaux, G. Optimal Differentiation of In Vitro Keratinocytes Requires Multifactorial External Control. PLoS ONE 2013, 8, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Bernstam, L.I.; Vaughan, F.L.; Bernstein, I.A. Keratinocytes grown at the air-liquid interface. Vitr. Cell. Dev. Biol. 1986, 22. [Google Scholar] [CrossRef]
- Prunieras, M.; Regnier, M.; Woodley, D. Methods for cultivation of keratinocytes with an air-liquid interface. J. Investig. Dermatol. 1983, 81, S28–S33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGrath, J.A.; Eady, R.A.J.; Pope, F.M. Anatomy and organization of human skin. Rook’s Textb. Dermatol. 2004, 10, 9781444317633. [Google Scholar]
- Ehrlich, F.; Fischer, H.; Langbein, L.; Praetzel-Wunder, S.; Ebner, B.; Figlak, K.; Weissenbacher, A.; Sipos, W.; Tschachler, E.; Eckhart, L. Differential evolution of the epidermal keratin cytoskeleton in terrestrial and aquatic mammals. Mol. Biol. Evol. 2019, 36, 328–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolarsick, P.A.J.; Kolarsick, M.A.; Goodwin, C. Anatomy and Physiology of the Skin. J. Dermatol. Nurses. Assoc. 2011, 3, 203–213. [Google Scholar] [CrossRef] [Green Version]
- Reijnders, C.M.A.; Van Lier, A.; Roffel, S.; Kramer, D.; Scheper, R.J.; Gibbs, S. Development of a Full-Thickness Human Skin Equivalent in Vitro Model Derived from TERT-Immortalized Keratinocytes and Fibroblasts. Tissue Eng.—Part A 2015, 21, 2448–2459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dickson, M.A.; Hahn, W.C.; Ino, Y.; Ronfard, V.; Wu, J.Y.; Weinberg, R.A.; Louis, D.N.; Li, F.P.; Rheinwald, J.G. Human Keratinocytes That Express hTERT and Also Bypass a p16INK4a-Enforced Mechanism That Limits Life Span Become Immortal yet Retain Normal Growth and Differentiation Characteristics. Mol. Cell. Biol. 2000, 20, 1436–1447. [Google Scholar] [CrossRef] [Green Version]
- Rheinwald, J.G.; Hahn, W.C.; Ramsey, M.R.; Wu, J.Y.; Guo, Z.; Tsao, H.; De Luca, M.; Catricalà, C.; O’Toole, K.M. A Two-Stage, p16INK4A- and p53-Dependent Keratinocyte Senescence Mechanism That Limits Replicative Potential Independent of Telomere Status. Mol. Cell. Biol. 2002, 22, 5157–5172. [Google Scholar] [CrossRef] [Green Version]
- Briley, B.; Shapiro, B. hTERT-Immortalized and Primary Keratinocytes Differentiate into Epidermal Structures in 3D Organotypic Culture. ATCC.org. Available online: https://www.atcc.org/resources/application-notes/htert-immortalized-and-primary-keratinocytes-differentiate (accessed on 6 April 2020).
- Van Drongelen, V.; Danso, M.O.; Mulder, A.; Mieremet, A.; Van Smeden, J.; Bouwstra, J.A.; El Ghalbzouri, A. Barrier properties of an N/TERT-based human skin equivalent. Tissue Eng.—Part A 2014, 20, 3041–3049. [Google Scholar] [CrossRef] [PubMed]
- van Drongelen, V.; Haisma, E.M.; Out-Luiting, J.J.; Nibbering, P.H.; El Ghalbzouri, A. Reduced filaggrin expression is accompanied by increased Staphylococcus aureus colonization of epidermal skin models. Clin. Exp. Allergy 2014, 44, 1515–1524. [Google Scholar] [CrossRef] [PubMed]
- Alloul-Ramdhani, M.; Tensen, C.P.; El Ghalbzouri, A. Performance of the N/TERT epidermal model for skin sensitizer identification via Nrf2-Keap1-ARE pathway activation. Toxicol. Vitr. 2014, 28, 982–989. [Google Scholar] [CrossRef] [PubMed]
- Rouse, J.G.; Van Dyke, M.E. A Review of Keratin-Based Biomaterials for Biomedical Applications. Materials 2010, 3, 999–1014. [Google Scholar] [CrossRef] [Green Version]
- Moll, R.; Divo, M.; Langbein, L. The human keratins: Biology and pathology. Histochem. Cell Biol. 2008, 129, 705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKittrick, J.; Chen, P.Y.; Bodde, S.G.; Yang, W.; Novitskaya, E.E.; Meyers, M.A. The Structure, Functions, and Mechanical Properties of Keratin. JOM 2012, 64, 449–468. [Google Scholar] [CrossRef]
- Buchanan, C.F.; Voigt, E.E.; Szot, C.S.; Freeman, J.W.; Vlachos, P.P.; Rylander, M.N. Three-dimensional microfluidic collagen hydrogels for investigating flow-mediated tumor-endothelial signaling and vascular organization. Tissue Eng. Part C Methods 2013, 20, 64–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.; Taraballi, F.; Tan, L.P.; Ng, K.W. Human keratin hydrogels support fibroblast attachment and proliferation in vitro. Cell Tissue Res. 2012, 347, 795–802. [Google Scholar] [CrossRef]
- Arnette, C.; Koetsier, J.L.; Hoover, P.; Getsios, S.; Green, K.J. In vitro model of the epidermis: Connecting protein function to 3D structure. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 2016; Volume 569, pp. 287–308. ISBN 0076-6879. [Google Scholar]
- Ikuta, S.; Sekino, N.; Hara, T.; Saito, Y.; Chida, K. Mouse epidermal keratinocytes in three-dimensional organotypic coculture with dermal fibroblasts form a stratified sheet resembling skin. Biosci. Biotechnol. Biochem. 2006, 70, 610050121. [Google Scholar] [CrossRef] [Green Version]
- Antoine, E.E.; Vlachos, P.P.; Rylander, M.N. Tunable collagen I hydrogels for engineered physiological tissue micro-environments. PLoS ONE 2015, 10, e0122500. [Google Scholar] [CrossRef] [Green Version]
- Antoine, E.E.; Vlachos, P.P.; Rylander, M.N. Review of collagen I hydrogels for bioengineered tissue microenvironments: Characterization of mechanics, structure, and transport. Tissue Eng. Part B Rev. 2014, 20, 683–696. [Google Scholar] [CrossRef] [PubMed]
- Zuniga, K.; Gadde, M.; Scheftel, J.; Senecal, K.; Cressman, E.; Van Dyke, M.; Rylander, M.N. Collagen/kerateine multi-protein hydrogels as a thermally stable extracellular matrix for 3D in vitro models. Int. J. Hyperth. 2021, 38, 830–845. [Google Scholar] [CrossRef]
- Mahanty, S.; Dakappa, S.S.; Shariff, R.; Patel, S.; Swamy, M.M.; Majumdar, A.; Gangi Setty, S.R. Keratinocyte differentiation promotes ER stress-dependent lysosome biogenesis. Cell Death Dis. 2019, 10, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lavker, R.M.; Gedeon Matoltsy, A. Formation of horny cells: The fate of cell organelles and differentiation products in ruminal epithelium. J. Cell Biol. 1970, 44, 501–512. [Google Scholar] [CrossRef]
- Boer, D.E.C.; van Smeden, J.; Bouwstra, J.A.; Aerts, J.M.F. Glucocerebrosidase: Functions in and Beyond the Lysosome. J. Clin. Med. 2020, 9, 736. [Google Scholar] [CrossRef] [Green Version]
- Holleran, W.M.; Takagi, Y.; Menon, G.K.; Jackson, S.M.; Lee, J.M.; Feingold, K.R.; Elias, P.M.; Takagi, Y.; Menon, G.K.; Jackson, S.M.; et al. Permeability Barrier Requirements Regulate Epidermal P-Glucocerebrosidase. Available online: https://n.d.www.jlr.org (accessed on 29 July 2020).
- Holleran, W.M.; Ginns, E.I.; Menon, G.K.; Grundmann, J.U.; Fartasch, M.; McKinney, C.E.; Elias, P.M.; Sidransky, E. Consequences of β-glucocerebrosidase deficiency in epidermis. Ultrastructure and permeability barrier alterations in Gaucher disease. J. Clin. Investig. 1994, 93, 1756–1764. [Google Scholar] [CrossRef] [Green Version]
- Holleran, W.M.; Takagi, Y.; Menon, G.K.; Legler, G.; Feingold, K.R.; Elias, P.M. Processing of epidermal glucosylceramides is required for optimal mammalian cutaneous permeability barrier function. J. Clin. Investig. 1993, 91, 1656–1664. [Google Scholar] [CrossRef] [Green Version]
- Dielschneider, R.F.; Henson, E.S.; Gibson, S.B. Lysosomes as Oxidative Targets for Cancer Therapy. Oxid. Med. Cell. Longev. 2017, 2017, 3749157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bocheńska, K.; Moskot, M.; Malinowska, M.; Jakóbkiewicz-Banecka, J.; Szczerkowska-Dobosz, A.; Purzycka-Bohdan, D.; Pleńkowska, J.; Slomiński, B.; Gabig-Cimińska, M. Lysosome alterations in the human epithelial cell line hacat and skin specimens: Relevance to psoriasis. Int. J. Mol. Sci. 2019, 20, 2255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takahashi, H.; Aoki, N.; Nakamura, S.; Asano, K.; Ishida-Yamamoto, A.; Iizuka, H. Cornified cell envelope formation is distinct from apoptosis in epidermal keratinocytes. J. Dermatol. Sci. 2000, 23, 161–169. [Google Scholar] [CrossRef]
- Harr, M.W.; Distelhorst, C.W. Apoptosis and Autophagy: Decoding Calcium Signals that Mediate Life or Death. Cold Spring Harb. Perspect. Biol. 2010, 2, a005579. [Google Scholar] [CrossRef]
- Fennelly, C.; Amaravadi, R.K. Lysosomal Biology in Cancer. Methods Mol. Biol. 2017, 1594, 293. [Google Scholar] [CrossRef] [Green Version]
- Barrandon, Y.; Green, H. Cell size as a determinant of the clone-forming ability of human keratinocytes. Proc. Natl. Acad. Sci. USA 1985, 82, 5390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watt, F.M.; Green, H. Involucrin synthesis is correlated with cell size in human epidermal cultures. J. Cell Biol. 1981, 90, 738–742. [Google Scholar] [CrossRef]
- Furuta, K.; Ikeda, M.; Nakayama, Y.; Nakamura, K.; Tanaka, M.; Hamasaki, N.; Himeno, M.; Hamilton, S.R.; August, J.T. Expression of lysosome-associated membrane proteins in human colorectal neoplasms and inflammatory diseases. Am. J. Pathol. 2001, 159, 449–455. [Google Scholar] [CrossRef] [Green Version]
- Vanhoutteghem, A.; Djian, P.; Green, H. Ancient origin of the gene encoding involucrin, a precursor of the cross-linked envelope of epidermis and related epithelia. Proc. Natl. Acad. Sci. USA 2008, 105, 15481–15486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watt, F.M.; Green, H. Stratification and terminal differentiation of cultured epidermal cells. Nature 1982, 295, 434–436. [Google Scholar] [CrossRef]
- Sanz-Gómez, N.; Freije, A.; Gandarillas, A. Keratinocyte Differentiation by Flow Cytometry. Methods Mol. Biol. 2019, 2109, 83–92. [Google Scholar] [CrossRef]
- Jakobsen, N.D.; Kaiser, K.; Ebbesen, M.F.; Lauritsen, L.; Gjerstorff, M.F.; Kuntsche, J.; Brewer, J.R. The ROC skin model: A robust skin equivalent for permeation and live cell imaging studies. Eur. J. Pharm. Sci. 2022, 178, 106282. [Google Scholar] [CrossRef]
- Rikken, G.; Niehues, H.; van den Bogaard, E.H. Organotypic 3D Skin Models: Human Epidermal Equivalent Cultures from Primary Keratinocytes and Immortalized Keratinocyte Cell Lines. Methods Mol. Biol. 2020, 2154, 45–61. [Google Scholar] [CrossRef]
- Tito, A.; Minale, M.; Riccio, S.; Grieco, F.; Colucci, M.G.; Apone, F. A Triticum vulgare Extract Exhibits Regenerating Activity During the Wound Healing Process. Clin. Cosmet. Investig. Dermatol. 2020, 13, 21. [Google Scholar] [CrossRef] [PubMed]
- Roy, D.C.; Tomblyn, S.; Isaac, K.M.; Kowalczewski, C.J.; Burmeister, D.M.; Burnett, L.R.; Christy, R.J. Ciprofloxacin-loaded keratin hydrogels reduce infection and support healing in a porcine partial-thickness thermal burn. Wound Repair Regen. 2016, 24, 657–668. [Google Scholar] [CrossRef]
- Ye, W.; Qin, M.; Qiu, R.; Li, J. Keratin-based wound dressings: From waste to wealth. Int. J. Biol. Macromol. 2022, 211, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Konop, M.; Sulejczak, D.; Czuwara, J.; Kosson, P.; Misicka, A.; Lipkowski, A.W.; Rudnicka, L. The role of allogenic keratin-derived dressing in wound healing in a mouse model. Wound Repair Regen. 2017, 25, 62–74. [Google Scholar] [CrossRef]
- Tang, A.; Li, Y.; Yao, Y.; Yang, X.; Cao, Z.; Nie, H.; Yang, G. Injectable keratin hydrogels as hemostatic and wound dressing materials. Biomater. Sci. 2021, 9, 4169–4177. [Google Scholar] [CrossRef] [PubMed]
- Roy, D.C.; Tomblyn, S.; Burmeister, D.M.; Wrice, N.L.; Becerra, S.C.; Burnett, L.R.; Saul, J.M.; Christy, R.J. Ciprofloxacin-Loaded Keratin Hydrogels Prevent Pseudomonas aeruginosa Infection and Support Healing in a Porcine Full-Thickness Excisional Wound. Adv. Wound Care 2015, 4, 457–468. [Google Scholar] [CrossRef]





| Hydrogel | Collagen Concentration | KTN Concentration | wt%/vol% |
|---|---|---|---|
| 100% Collagen | 4.0 mg/mL | 0.0 mg/ml | 4.0 |
| 50/50 C/KTN | 4.0 mg/mL | 4.0 mg/ml | 8.0 |
| Sample Group | Medium | p-Value |
|---|---|---|
| Plate, Ker-CT | Ca-free vs. Ca+ | Day 2 (p < 0.0001)—**** Day 4 (p > 0.9999)—ns Day 7 (p = 0.0392)—* |
| Ca-free vs. Ca+/FBS | Day 2 (p = 0.0216)—* Day 4 (p < 0.0001)—**** Day 7 (p = 0.0317)—* | |
| Ca+ vs. Ca+/FBS | Day 2 (p < 0.0001)—**** Day 4 (p < 0.0001)—**** Day 7 (p > 0.9999)—ns | |
| 100% Collagen, Ker-CT | Ca-free vs. Ca+ | Day 2 (p = 0.9925)—ns Day 4 (p = 0.2850)—ns Day 7 (p = 0.0329)—* |
| Ca-free vs. Ca+/FBS | Day 2 (p > 0.9999)—ns Day 4 (p < 0.0001)—*** Day 7 (p = 0.2709)—ns | |
| Ca+ vs. Ca+/FBS | Day 2 (p = 0.9859)—ns Day 4 (p < 0.0001)—**** Day 7 (p = 0.0002)—*** | |
| 50/50 C/KTN, Ker-CT | Ca-free vs. Ca+ | Day 2 (p = 0.0017)—* Day 4 (p = 0.0005)—*** Day 7 (p = 0.2412)—ns |
| Ca-free vs. Ca+/FBS | Day 2 (p = 0.9961)—ns Day 4 (p = 0.3243)—ns Day 7 (p = 0.0724)—ns | |
| Ca+ vs. Ca+/FBS | Day 2 (p = 0.0161)—* Day 4 (p < 0.0001)—**** Day 7 (p = 0.9984)—ns | |
| Plate, NHEK | Ca-free vs. Ca+ | Day 2 (p < 0.0001)—**** Day 4 (p < 0.0001)—**** Day 7 (p < 0.0001)—**** |
| Ca-free vs. Ca+/FBS | Day 2 (p < 0.0001)—**** Day 4 (p < 0.0001)—**** Day 7 (p < 0.0001)—**** | |
| Ca+ vs. Ca+/FBS | Day 2 (p = 0.4885)—ns Day 4 (p = 0.0526)—ns Day 7 (p = 0.9928)—ns | |
| 100% Collagen, NHEK | Ca-free vs. Ca+ | Day 2 (p < 0.0001)—**** Day 4 (p < 0.0001)—**** Day 7 (p = 0.0007)—*** |
| Ca-free vs. Ca+/FBS | Day 2 (p = 0.0020)—** Day 4 (p < 0.0001)—**** Day 7 (p < 0.0001)—**** | |
| Ca+ vs. Ca+/FBS | Day 2 (p < 0.0001)—**** Day 4 (p = 0.0006)—*** Day 7 (p = 0.0138)—* | |
| 50/50 C/KTN, NHEK | Ca-free vs. Ca+ | Day 2 (p < 0.0001)—**** Day 4 (p < 0.0001)—**** Day 7 (p < 0.0001)—**** |
| Ca-free vs. Ca+/FBS | Day 2 (p < 0.0001)—**** Day 4 (p < 0.0001)—**** Day 7 (p < 0.0001)—**** | |
| Ca+ vs. Ca+/FBS | Day 2 (p = 0.1154)—ns Day 4 (p < 0.0001)—**** Day 7 (p = 0.0173)—* |
| Sample Group | Medium | DL (µm) |
|---|---|---|
| Plate | Ca-free | Day 2—5.96 ± 0.45 Day 4—6.26 ± 0.40 |
| Ca+ | Day 2—7.74 ± 0.59 Day 4—8.46 ± 0.53 | |
| 100% Collagen | Ca-free | Day 2—4.97 ± 0.39 Day 4—5.36 ± 0.28 |
| Ca+ | Day 2—7.17 ± 0.53 Day 4—11.5 ± 0.60 | |
| 50/50 C/KTN | Ca-free | Day 2—4.84 ± 0.28 Day 4—8.18 ± 0.73 |
| Ca+ | Day 2—7.76 ± 0.48 Day 4—12.23 ± 0.76 |
| Sample Group | Media | Day 2 vs. Day 4 |
|---|---|---|
| Plate | Ca-free—CTCF | CK14 (p = 0.0013)—** Involucrin (p = 0.2088)—ns |
| Ca+—CTCF | CK14 (p < 0.0001)—**** Involucrin (p = 0.4185)—ns | |
| Ca-free—total fluorescence | CK14 (p = 0.0235)—* Involucrin (p = 0.0349)—* | |
| Ca+—total fluorescence | CK14 (p = 0.0235)—ns Involucrin (p = 0.2073)—ns | |
| 100% Collagen | Ca-free—CTCF | CK14 (p = 0.0025)—** Involucrin (p = 0.2609)—ns |
| Ca+—CTCF | CK14 (p < 0.0001)—**** Involucrin (p = 0.9924)—ns | |
| Ca-free—total fluorescence | CK14 (p = 0.0079)—** Involucrin (p = 0.0012)—** | |
| Ca+—total fluorescence | CK14 (p = 0.0433)—* Involucrin (p = 0.8726)—ns | |
| 50/50 C/KTN | Ca-free—CTCF | CK14 (p < 0.0001)—**** Involucrin (p = 0.0559)—ns |
| Ca+—CTCF | CK14 (p < 0.0001)—**** Involucrin (p = 0.4680)—ns | |
| Ca-free—total fluorescence | CK14 (p > 0.9999)—ns Involucrin (p = 0.0272)—* | |
| Ca+—total fluorescence | CK14 (p = 0.6690)—ns Involucrin (p = 0.4151)—ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuniga, K.; Ghousifam, N.; Sansalone, J.; Senecal, K.; Van Dyke, M.; Rylander, M.N. Keratin Promotes Differentiation of Keratinocytes Seeded on Collagen/Keratin Hydrogels. Bioengineering 2022, 9, 559. https://doi.org/10.3390/bioengineering9100559
Zuniga K, Ghousifam N, Sansalone J, Senecal K, Van Dyke M, Rylander MN. Keratin Promotes Differentiation of Keratinocytes Seeded on Collagen/Keratin Hydrogels. Bioengineering. 2022; 9(10):559. https://doi.org/10.3390/bioengineering9100559
Chicago/Turabian StyleZuniga, Kameel, Neda Ghousifam, John Sansalone, Kris Senecal, Mark Van Dyke, and Marissa Nichole Rylander. 2022. "Keratin Promotes Differentiation of Keratinocytes Seeded on Collagen/Keratin Hydrogels" Bioengineering 9, no. 10: 559. https://doi.org/10.3390/bioengineering9100559