Preparation of Porous Composite Phase Na Super Ionic Conductor Adsorbent by In Situ Process for Ultrafast and Efficient Strontium Adsorption from Wastewater
Abstract
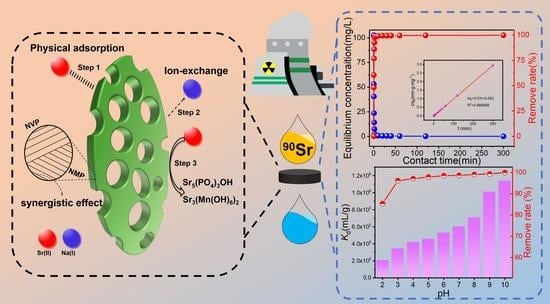
:1. Introduction
2. Materials and Methods
2.1. Material and Reagents
2.2. Synthesis of Na3V2PO4@NaMnPO4 (NVP@NMP)
2.3. Characterization
2.4. Batch Adsorption Experiments
3. Results and Discussion
3.1. Characterization of NVP@NMP
3.2. Effects of Adsorbent Dosage and pH on Sr Adsorption
3.3. Kinetics of Sr2+ Adsorption
3.4. Effects of Concentrations and Temperature on Sr2+ Adsorption
3.5. Effects of Competitive Ions on Sr2+ Adsorption
3.6. Sr2+ Adsorption with Actual Water
3.7. Adsorption Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, X.; Liu, Y. Nanomaterials for Radioactive Wastewater Decontamination. Environ. Sci. Nano 2020, 7, 1008–1040. [Google Scholar] [CrossRef]
- Wang, K.; Wang, F.; Chen, F.; Cui, X.; Wei, Y.; Shao, L.; Yu, M. One-Pot Preparation of NaA Zeolite Microspheres for Highly Selective and Continuous Removal of Sr(II) from Aqueous Solution. ACS Sustain. Chem. Eng. 2019, 7, 2459–2470. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Tsukimoto, S.; Nakayama, K.S.; Asao, N. Ultrafine Sodium Titanate Nanowires with Extraordinary Sr Ion-Exchange Properties. Nano Lett. 2015, 15, 2980–2984. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.Y.; Ding, D.; Sun, M.; Cheng, L.; Wang, C. Effective and Rapid Adsorption of Sr2+ Ions by a Hydrated Pentasodium Cluster Templated Zinc Thiostannate. Inorg. Chem. 2019, 58, 10184–10193. [Google Scholar] [CrossRef]
- Xiang, S.; Mao, H.; Geng, W.; Xu, Y.; Zhou, H. Selective Removal of Sr(II) from Saliferous Radioactive Wastewater by Capacitive Deionization. J. Hazard. Mater. 2022, 431, 128591. [Google Scholar] [CrossRef]
- Almasri, D.A.; Kayvani Fard, A.; McKay, G.; Kochkodan, V.; Atieh, M.A. A Novel Adsorptive Ceramic Membrane for Efficient Strontium Removal. J. Water Process Eng. 2020, 37, 101538. [Google Scholar] [CrossRef]
- Sihn, Y.; Yang, H.M.; Park, C.W.; Yoon, I.H.; Kim, I. Post-Substitution of Magnesium at CaI of Nano-Hydroxyapatite Surface for Highly Efficient and Selective Removal of Radioactive 90Sr from Groundwater. Chemosphere 2022, 295, 133874. [Google Scholar] [CrossRef]
- Lu, T.; Zhu, Y.; Wang, W.; Qi, Y.; Wang, A. Interconnected Superporous Adsorbent Prepared via Yeast-Based Pickering HIPEs for High-Efficiency Adsorption of Rb+, Cs+ and Sr2+. Chem. Eng. J. 2019, 361, 1411–1422. [Google Scholar] [CrossRef]
- Alby, D.; Charnay, C.; Heran, M.; Prelot, B.; Zajac, J. Recent Developments in Nanostructured Inorganic Materials for Sorption of Cesium and Strontium: Synthesis and Shaping, Sorption Capacity, Mechanisms, and Selectivity—A Review. J. Hazard. Mater. 2018, 344, 511–530. [Google Scholar] [CrossRef]
- Ding, B.; Zheng, P.; Ma, P.; Lin, J. Manganese Oxide Nanomaterials: Synthesis, Properties, and Theranostic Applications. Adv. Mater. 2020, 32, 1905823. [Google Scholar] [CrossRef]
- Huo, J.; Yu, G.; Wang, J. Efficient Removal of Co(II) and Sr(II) from Aqueous Solution Using Polyvinyl Alcohol/Graphene Oxide/MnO2 Composite as a Novel Adsorbent. J. Hazard. Mater. 2021, 411, 125117. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Gu, P.; Yan, S.; Dong, L.; Zhang, G. Na/Zn/Sn/S (NaZTS): Quaternary Metal Sulfide Nanosheets for Efficient Adsorption of Radioactive Strontium Ions. Chem. Eng. J. 2020, 379, 122227. [Google Scholar] [CrossRef]
- Burns, P.C.; Ewing, R.C.; Navrotsky, A. Nuclear Fuel in a Reactor Accident. Science 2012, 335, 1184–1188. [Google Scholar] [CrossRef]
- Li, W.A.; Peng, Y.C.; Ma, W.; Huang, X.Y.; Feng, M.L. Rapid and Selective Removal of Cs+ and Sr2+ Ions by Two Zeolite-Type Sulfides via Ion Exchange Method. Chem. Eng. J. 2022, 442, 136377. [Google Scholar] [CrossRef]
- Mautner, A.; Kwaw, Y.; Weiland, K.; Mvubu, M.; Botha, A.; John, M.J.; Mtibe, A.; Siqueira, G.; Bismarck, A. Natural Fibre-Nanocellulose Composite Filters for the Removal of Heavy Metal Ions from Water. Ind. Crop. Prod. 2019, 133, 325–332. [Google Scholar] [CrossRef]
- Vishwakarma, R.K.; Narayanam, P.K.; Umamaheswari, R.; Sundararajan, K. Surface Modified and Functionalized Graphene Oxide Membranes for Separation of Strontium from Aqueous Solutions. J. Environ. Manag. 2021, 298, 113443. [Google Scholar] [CrossRef]
- James, A.M.; Harding, S.; Robshaw, T.; Bramall, N.; Ogden, M.D.; Dawson, R. Selective Environmental Remediation of Strontium and Cesium Using Sulfonated Hyper-Cross-Linked Polymers (SHCPs). ACS Appl. Mater. Interfaces 2019, 11, 22464–22473. [Google Scholar] [CrossRef]
- Zhao, Y.M.; Cheng, L.; Wang, K.Y.; Hao, X.; Wang, J.; Zhu, J.Y.; Sun, M.; Wang, C. PH-Controlled Switch over Coadsorption and Separation for Mixed Cs+ and Sr2+ by an Acid-Resistant Potassium Thioantimonate. Adv. Funct. Mater. 2022, 32, 2112717. [Google Scholar] [CrossRef]
- Ullah, I.; Gloria, A.; Zhang, W.; Ullah, M.W.; Wu, B.; Li, W.; Domingos, M.; Zhang, X. Synthesis and Characterization of Sintered Sr/Fe-Modified Hydroxyapatite Bioceramics for Bone Tissue Engineering Applications. ACS Biomater. Sci. Eng. 2020, 6, 375–388. [Google Scholar] [CrossRef]
- Xu, S.; Ning, S.; Wang, Y.; Wang, X.; Dong, H.; Chen, L.; Yin, X.; Fujita, T.; Wei, Y. Precise Separation and Efficient Enrichment of Palladium from Wastewater by Amino-Functionalized Silica Adsorbent. J. Clean. Prod. 2023, 396, 136479. [Google Scholar] [CrossRef]
- Chen, Y.; Ning, S.; Zhong, Y.; Li, Z.; Wang, J.; Chen, L.; Yin, X.; Fujita, T.; Wei, Y. Study on Highly Efficient Separation of Zirconium from Scandium with TODGA-Modified Macroporous Silica-Polymer Based Resin. Sep. Purif. Technol. 2023, 305, 122499. [Google Scholar] [CrossRef]
- Liang, J.; Li, J.; Li, X.; Liu, K.; Wu, L.; Shan, G. The Sorption Behavior of CHA-Type Zeolite for Removing Radioactive Strontium from Aqueous Solutions. Sep. Purif. Technol. 2020, 230, 115874. [Google Scholar] [CrossRef]
- Singha Deb, A.K.; Manju, M.; Sengupta, A.; Ali, S.M. Efficient Separation of Strontium Ions from Aqueous Solution by Dibenzo-18-Crown-6 Functionalized Resin: Static and Dynamic Adsorption Studies with Computational DFT Insights. Chem. Eng. J. Adv. 2022, 11, 100308. [Google Scholar] [CrossRef]
- Huang, T.; Zhang, S.W.; Zhou, L.; Liu, L.F. Electrokinetics Couples with the Adsorption of Activated Carbon-Supported Hydroxycarbonate Green Rust That Enhances the Removal of Sr Cations from the Stock Solution in Batch and Column. Sep. Purif. Technol. 2021, 265, 118531. [Google Scholar] [CrossRef]
- Milcent, T.; Hertz, A.; Barré, Y.; Grandjean, A. Influence of the Nb Content and Microstructure of Sitinakite-Type Crystalline Silicotitanates (CSTs) on Their Sr2+ and Cs+ Sorption Properties. Chem. Eng. J. 2021, 426, 15–19. [Google Scholar] [CrossRef]
- Li, W.; Yao, Z.; Zhou, C.; Wang, X.; Xia, X.; Gu, C.; Tu, J. Boosting High-Rate Sodium Storage Performance of N-Doped Carbon-Encapsulated Na3V2(PO4)3 Nanoparticles Anchoring on Carbon Cloth. Small 2019, 15, 1902432. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Xue, L.; Lü, X.; Gao, H.; Li, Y.; Xin, S.; Fu, G.; Cui, Z.; Zhu, Y.; Goodenough, J.B. NaxMV(PO4)3 (M = Mn, Fe, Ni) Structure and Properties for Sodium Extraction. Nano Lett. 2016, 16, 7836–7841. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, R.; Zhang, Z.; Tang, Y.; Jia, C.; Ji, X.; Wang, H. Understanding Crystal Structures, Ion Diffusion Mechanisms and Sodium Storage Behaviors of NASICON Materials. Energy Storage Mater. 2021, 34, 171–193. [Google Scholar] [CrossRef]
- Chen, M.; Hua, W.; Xiao, J.; Cortie, D.; Guo, X.; Wang, E.; Gu, Q.; Hu, Z.; Indris, S.; Wang, X.L.; et al. Development and Investigation of a NASICON-Type High-Voltage Cathode Material for High-Power Sodium-Ion Batteries. Angew. Chem.-Int. Ed. 2020, 59, 2449–2456. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, W.; Sun, K.; Guo, J.; Yuan, K.; Fu, J.; Zhang, T.; Zhang, X.; Long, H.; Zhang, Z.; et al. Manganese-Based Materials for Rechargeable Batteries beyond Lithium-Ion. Adv. Energy Mater. 2021, 11, 2100867. [Google Scholar] [CrossRef]
- Chowdhury, A.; Biswas, S.; Dhar, A.; Burada, P.S.; Chandra, A. Stable Na-Ion Supercapacitor under Non-Ambient Conditions Using Maricite-NaMnPO4 Nanoparticles. J. Power Sources 2021, 516, 230679. [Google Scholar] [CrossRef]
- Zhang, W.; Wu, Y.; Xu, Z.; Li, H.; Xu, M.; Li, J.; Dai, Y.; Zong, W.; Chen, R.; He, L.; et al. Rationally Designed Sodium Chromium Vanadium Phosphate Cathodes with Multi-Electron Reaction for Fast-Charging Sodium-Ion Batteries. Adv. Energy Mater. 2022, 12, 2201065. [Google Scholar] [CrossRef]
- Chowdhury, A.; Biswas, S.; Mandal, D.; Chandra, A. Facile Strategy of Using Conductive Additive Supported NaMnPO4 Nanoparticles for Delivering High Performance Na-Ion Supercapacitors. J. Alloys Compd. 2022, 902, 163733. [Google Scholar] [CrossRef]
- Liang, L.; Sun, X.; Zhang, J.; Hou, L.; Sun, J.; Liu, Y.; Wang, S.; Yuan, C. In Situ Synthesis of Hierarchical Core Double-Shell Ti-Doped LiMnPO4@NaTi2(PO4)3@C/3D Graphene Cathode with High-Rate Capability and Long Cycle Life for Lithium-Ion Batteries. Adv. Energy Mater. 2019, 9, 1802847. [Google Scholar] [CrossRef]
- Liang, L.; Sun, X.; Wu, C.; Hou, L.; Sun, J.; Zhang, X.; Yuan, C. Nasicon-Type Surface Functional Modification in Core-Shell LiNi0.5Mn0.3Co0.2O2@NaTi2(PO4)3 Cathode Enhances Its High-Voltage Cycling Stability and Rate Capacity toward Li-Ion Batteries. ACS Appl. Mater. Interfaces 2018, 10, 5498–5510. [Google Scholar] [CrossRef]
- Zhang, Z.; Gu, P.; Zhang, M.; Yan, S.; Dong, L.; Zhang, G. Synthesis of a Robust Layered Metal Sulfide for Rapid and Effective Removal of Sr2+ from Aqueous Solutions. Chem. Eng. J. 2019, 372, 1205–1215. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, J.; Xiong, H.; Yuan, Z.; Zhu, Y.; Hu, B. Constructing New Fe3O4@MnOx with 3D Hollow Structure for Efficient Recovery of Uranium from Simulated Seawater. Chemosphere 2021, 283, 131241. [Google Scholar] [CrossRef]
- Biomass, A.; Lucaci, A.R.; Bulgariu, D.; Ahmad, I. Equilibrium and Kinetics Studies of Metal Ions Biosorption on Alginate Extracted from Marine Red Algae Biomass (Callithamnion corymbosum sp.). Polymers 2020, 19, 1888. [Google Scholar]
- Liu, X.; Pang, H.; Liu, X.; Li, Q.; Zhang, N.; Mao, L.; Qiu, M.; Hu, B.; Yang, H.; Wang, X. Orderly Porous Covalent Organic Frameworks-Based Materials: Superior Adsorbents for Pollutants Removal from Aqueous Solutions. Innovation 2021, 2, 100076. [Google Scholar] [CrossRef]
- Lim, A.; Chew, J.J.; Ismadji, S.; Khaerudini, D.S.; Darsono, N.; Sunarso, J. Kinetic and Equilibrium Adsorption Study of Anionic Dyes Using Oil Palm Trunk-Derived Activated Carbon. Mater. Today Proc. 2022, 64, 1627–1638. [Google Scholar] [CrossRef]
- Yu, Y.; Zhou, L.; Tang, J.; Wu, P.; Feng, L.; Ge, B.; Chen, H.; Hu, J.; Song, S.; Zeng, T. Effective Removal of Co(II) and Sr(II) from Radiocative Wastes Using Covalent Triazine Frameworks: Kinetics and Isotherm Studies. Sep. Purif. Technol. 2021, 277, 119633. [Google Scholar] [CrossRef]
- Lei, H.; Muhammad, Y.; Wang, K.; Yi, M.; He, C.; Wei, Y.; Fujita, T. Facile Fabrication of Metakaolin/Slag-Based Zeolite Microspheres (M/SZMs) Geopolymer for the Efficient Remediation of Cs+ and Sr2+ from Aqueous Media. J. Hazard. Mater. 2021, 406, 124292. [Google Scholar] [CrossRef]
- Yin, J.; Yang, S.; He, W.; Zhao, T.; Li, C.; Hua, D. Biogene-Derived Aerogels for Simultaneously Selective Adsorption of Uranium(VI) and Strontium(II) by Co-Imprinting Method. Sep. Purif. Technol. 2021, 271, 118849. [Google Scholar] [CrossRef]
- Ahmadi, S.J.; Akbari, N.; Shiri-Yekta, Z.; Mashhadizadeh, M.H.; Hosseinpour, M. Removal of Strontium Ions from Nuclear Waste Using Synthesized MnO2-ZrO2 Nano-Composite by Hydrothermal Method in Supercritical Condition. Korean J. Chem. Eng. 2015, 32, 478–485. [Google Scholar] [CrossRef]
- Zhang, P.; Wang, Y.; Zhang, D. Removal of Nd(III), Sr(II), and Rb(I) Ions from Aqueous Solution by Thiacalixarene-Functionalized Graphene Oxide Composite as an Adsorbent. J. Chem. Eng. Data 2016, 61, 3679–3691. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y. Ultrafast Removal of Radioactive Strontium Ions from Contaminated Water by Nanostructured Layered Sodium Vanadosilicate with High Adsorption Capacity and Selectivity. J. Hazard. Mater. 2020, 398, 122907. [Google Scholar] [CrossRef]
- Shen, Z.; Yan, G.; Chen, G.; Cao, L.; Tang, X.; Sun, Y.; Liu, J.; Yang, S.; Lin, L.; Zeng, X. Preparation and Strontium Adsorption Behaviors of a New Sodium Manganese Silicate Material. Sep. Purif. Technol. 2022, 290, 120824. [Google Scholar] [CrossRef]
- Xiong, H.; Qian, R.; Liu, Z.; Zhang, R.; Sun, G.; Guo, B.; Du, F.; Song, S.; Qiao, Z.A.; Dai, S. A Polymer-Assisted Spinodal Decomposition Strategy toward Interconnected Porous Sodium Super Ionic Conductor-Structured Polyanion-Type Materials and Their Application as a High-Power Sodium-Ion Battery Cathode. Adv. Sci. 2021, 8, 2004943. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Zhao, W.; Yuan, S.; Zhang, L.; Yang, Y.; Ge, P.; Ji, X. Designing Rational Interfacial Bonds for Hierarchical Mineral-Type Trogtalite with Double Carbon towards Ultra-Fast Sodium-Ions Storage Properties. Adv. Funct. Mater. 2021, 31, 2100156. [Google Scholar] [CrossRef]
- Sun, S.; Chen, Y.; Cheng, J.; Tian, Z.; Wang, C.; Wu, G.; Liu, C.; Wang, Y.; Guo, L. Constructing Dimensional Gradient Structure of Na3V2(PO4)3/C@CNTs-WC by Wolfram Substitution for Superior Sodium Storage. Chem. Eng. J. 2021, 420, 130453. [Google Scholar] [CrossRef]
- Shin, J.; Choi, M.; Go, C.Y.; Bae, S.; Kim, K.C.; Chon, K. NaOH-Assisted H2O2 Post-Modification as a Novel Approach to Enhance Adsorption Capacity of Residual Coffee Waste Biochars toward Radioactive Strontium: Experimental and Theoretical Studies. J. Hazard. Mater. 2022, 435, 129081. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Wu, G.; Song, J.; Hu, B. Preparation of Chitosan-Based Asymmetric Electrodes by Co-Imprinting Technology for Simultaneous Electro-Adsorption of Multi-Radionuclides. Sep. Purif. Technol. 2022, 297, 121568. [Google Scholar] [CrossRef]
- Zhang, H.; Li, C.; Chen, X.; Fu, H.; Chen, Y.; Ning, S.; Fujita, T.; Wei, Y.; Wang, X. Layered Ammonium Vanadate Nanobelt as Efficient Adsorbents for Removal of Sr2+ and Cs+ from Contaminated Water. J. Colloid Interface Sci. 2022, 615, 110–123. [Google Scholar] [CrossRef] [PubMed]








| Adsorbents | Pseudo-First-Order | Pseudo-Second-Order | E.T. (min) | ||||
|---|---|---|---|---|---|---|---|
| K1 (L/min) | qe | R2 | K2 (g/mmol/min) | qe | R2 | ||
| 1V: 9Mn | 3.01 | 99.23 | 0.97 | 0.05 | 101.83 | 1.00 | 2 |
| 3V: 7Mn | 2.57 | 98.09 | 0.97 | 0.03 | 102.04 | 0.99 | 5 |
| 5V: 5Mn | 1.69 | 98.40 | 0.98 | 0.02 | 102.04 | 0.99 | 20 |
| 7V: 3Mn | 1.34 | 98.20 | 0.98 | 0.02 | 102.14 | 0.99 | 30 |
| 9V: 1Mn | 0.72 | 93.35 | 0.97 | 0.01 | 102.35 | 0.99 | 60 |
| NVP | 1.05 | 97.33 | 0.99 | 0.01 | 102.04 | 0.99 | 40 |
| NMP | 0.83 | 94.78 | 0.98 | 0.01 | 102.14 | 0.99 | 40 |
| Adsorbent | pH | E.T. (min) | qe (mg/g) | Reference |
|---|---|---|---|---|
| Covalent triazine framework-B | 2–9 | 10 | 35.61 | [41] |
| Metakaolin/slag-based zeolite microspheres | 2–8 | 60 | 37.04 | [42] |
| Uranyl and strontium ion-imprinted aerogel | 2–7 | 120 | 160 | [43] |
| Nano ZrO2-MnO2 | 4–6 | 30 | 65.7 | [44] |
| Thiacalixarene-functionalized graphene oxide | 7–9 | 55 | 101.10 | [45] |
| Layered sodium vanadosilicates | 2.5–11 | 5 | 174.30 | [46] |
| Na/Zn/Sn/S | 2–12 | 5 | 32.30 | [12] |
| Sodium manganese silicate material | 2–12 | 1 | 249.00 | [47] |
| 1V: 9Mn NVMP | 2–10 | 2 | 198.88 | This work |
| Temperature (K) | |||
|---|---|---|---|
| 298 | 42.29 | 0.23 | −25.45 |
| 308 | −27.73 | ||
| 318 | −30.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Yin, X.; Fu, H.; Lin, Z.; Ma, G.; Wang, X.; Wang, Q.; Chen, F. Preparation of Porous Composite Phase Na Super Ionic Conductor Adsorbent by In Situ Process for Ultrafast and Efficient Strontium Adsorption from Wastewater. Metals 2023, 13, 677. https://doi.org/10.3390/met13040677
Chen Y, Yin X, Fu H, Lin Z, Ma G, Wang X, Wang Q, Chen F. Preparation of Porous Composite Phase Na Super Ionic Conductor Adsorbent by In Situ Process for Ultrafast and Efficient Strontium Adsorption from Wastewater. Metals. 2023; 13(4):677. https://doi.org/10.3390/met13040677
Chicago/Turabian StyleChen, Yuliang, Xiangbiao Yin, Hao Fu, Zheyang Lin, Guangcan Ma, Xinpeng Wang, Qingsong Wang, and Fangqiang Chen. 2023. "Preparation of Porous Composite Phase Na Super Ionic Conductor Adsorbent by In Situ Process for Ultrafast and Efficient Strontium Adsorption from Wastewater" Metals 13, no. 4: 677. https://doi.org/10.3390/met13040677