Recent Developments in Photocatalytic Nanotechnology for Purifying Air Polluted with Volatile Organic Compounds: Effect of Operating Parameters and Catalyst Deactivation
Abstract
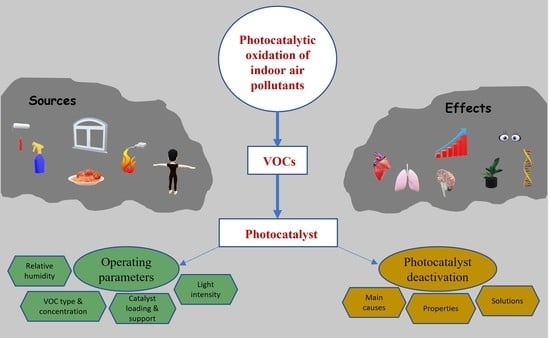
:1. Introduction
2. Air Pollutants and VOCs
3. Types of Nano Photocatalysts
4. Influence of Environmental Conditions
4.1. Effect of Relative Humidity
4.2. Effect of VOC Type and Concentration
4.3. Effect of Catalyst Loading and Support
4.4. Effect of Light Intensity
5. Catalyst Deactivation and Possible Solutions
5.1. Main Reasons for Catalyst Deactivation
5.2. Properties of Deactivated Photocatalysts
5.3. Solutions for Reducing the Catalyst Deactivation
6. Conclusions and Outlook
- (1)
- The photocatalytic efficiency of VOC oxidation depends on various operational parameters (airflow, reactor type, residence time, wavelength and intensity of light, humidity, temperature, etc.). To understand how the degradation efficiency depends on various external factors and to achieve the maximum conversion rate, it is always recommended to perform the PCO reaction in a standard reactor system with different operational parameters.
- (2)
- The stability of the investigated photocatalyst is demonstrated by how long it lasts until the activity starts reducing during continuous operation. The important questions are why the deactivation occurs and how we can reduce the deactivation.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Huang, G.; Liu, L.; Chen, L.; Gao, L.; Zhu, J.; Fu, H. Unique Insights into Photocatalytic VOCs Oxidation over WO3/Carbon Dots Nanohybrids Assisted by Water Activation and Electron Transfer at Interfaces. J. Hazard. Mater. 2022, 423, 127134. [Google Scholar] [CrossRef]
- Li, Y.W.; Ma, W.L. Photocatalytic Oxidation Technology for Indoor Air Pollutants Elimination: A Review. Chemosphere 2021, 280, 130667. [Google Scholar] [CrossRef]
- Mamaghani, A.H.; Haghighat, F.; Lee, C.S. Effect of Titanium Dioxide Properties and Support Material on Photocatalytic Oxidation of Indoor Air Pollutants. Build. Environ. 2021, 189, 107518. [Google Scholar] [CrossRef]
- Shah, K.W.; Li, W. A Review on Catalytic Nanomaterials for Volatile Organic Compounds VOC Removal and Their Applications for Healthy Buildings. Nanomaterials 2019, 9, 910. [Google Scholar] [CrossRef]
- Schiffman, S.S.; Williams, C.M. Science of Odor as a Potential Health Issue. J. Environ. Qual. 2005, 34, 129–138. [Google Scholar] [PubMed]
- Guo, Y.; Wen, M.; Li, G.; An, T. Recent Advances in VOC Elimination by Catalytic Oxidation Technology onto Various Nanoparticles Catalysts: A Critical Review. Appl. Catal. B Environ. 2021, 281, 119447. [Google Scholar] [CrossRef]
- Louis, J.; Padmanabhan, N.T.; John, H. Commercial Applications and Future Trends of Photocatalytic Materials. In Photocatalysis; Walter de Gruyter GmbH: Berlin, Germany, 2021; pp. 343–380. [Google Scholar] [CrossRef]
- Geng, Q.; Wang, H.; Chen, R.; Chen, L.; Li, K.; Dong, F. Advances and challenges of photocatalytic technology for air purification. Natl. Sci. Open 2022, 1, 20220025. [Google Scholar] [CrossRef]
- Liu, B.; Zhan, Y.; Xie, R.; Huang, H.; Li, K.; Zeng, Y.; Shrestha, R.P.; Kim Oanh, N.T.; Winijkul, E. Efficient Photocatalytic Oxidation of Gaseous Toluene in a Bubbling Reactor of Water. Chemosphere 2019, 233, 754–761. [Google Scholar] [CrossRef]
- Zou, W.; Gao, B.; Ok, Y.S.; Dong, L. Integrated Adsorption and Photocatalytic Degradation of Volatile Organic Compounds (VOCs) Using Carbon-Based Nanocomposites: A Critical Review. Chemosphere 2019, 218, 845–859. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Adeel, M.; Zhang, P.; Zhou, P.; Huang, L.; Zhao, Y.; Ahmad, M.A.; Shakoor, N.; Lou, B.; Jiang, Y.; et al. A Critical Review on Surface-Modified Nano-Catalyst Application for the Photocatalytic Degradation of Volatile Organic Compounds. Environ. Sci. Nano 2022, 9, 61–80. [Google Scholar] [CrossRef]
- Chen, J.; Chen, L.; Wang, X.; Sun, J.; Chen, A.; Xie, X. Er Single Atoms Decorated TiO2 and Er3+ Ions Modified TiO2 for Photocatalytic Oxidation of Mixed VOCs. Appl. Surf. Sci. 2022, 596, 153655. [Google Scholar] [CrossRef]
- Mo, J.; Zhang, Y.; Xu, Q.; Zhu, Y.; Lamson, J.J.; Zhao, R. Determination and Risk Assessment of By-Products Resulting from Photocatalytic Oxidation of Toluene. Appl. Catal. B Environ. 2009, 89, 570–576. [Google Scholar] [CrossRef]
- Hodgson, A.T.; Destaillats, H.; Sullivan, D.P.; Fisk, W.J. Performance of Ultraviolet Photocatalytic Oxidation for Indoor Air Cleaning Applications. Indoor Air 2007, 17, 305–316. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-Light Photocatalysis in Nitrogen-Doped Titanium Oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ouyang, S.; Xu, H.; Wang, X.; Bi, Y.; Zhang, Y.; Ye, J. Constructing Solid-Gas-Interfacial Fenton Reaction over Alkalinized-C3N4 Photocatalyst to Achieve Apparent Quantum Yield of 49% at 420 Nm. J. Am. Chem. Soc. 2016, 138, 13289–13297. [Google Scholar] [CrossRef] [PubMed]
- Mo, J.; Zhang, Y.; Xu, Q.; Lamson, J.J.; Zhao, R. Photocatalytic Purification of Volatile Organic Compounds in Indoor Air: A Literature Review. Atmos. Environ. 2009, 43, 2229–2246. [Google Scholar] [CrossRef]
- Weon, S.; Choi, W. TiO2 Nanotubes with Open Channels as Deactivation-Resistant Photocatalyst for the Degradation of Volatile Organic Compounds. Environ. Sci. Technol. 2016, 50, 2556–2563. [Google Scholar] [CrossRef]
- Mamaghani, A.H.; Haghighat, F.; Lee, C.S. Photocatalytic Degradation of VOCs on Various Commercial Titanium Dioxides: Impact of Operating Parameters on Removal Efficiency and by-Products Generation. Build. Environ. 2018, 138, 275–282. [Google Scholar] [CrossRef]
- Saoud, W.A.; Kane, A.; Le Cann, P.; Gerard, A.; Lamaa, L.; Peruchon, L.; Brochier, C.; Bouzaza, A.; Wolbert, D.; Assadi, A.A. Innovative Photocatalytic Reactor for the Degradation of VOCs and Microorganism under Simulated Indoor Air Conditions: Cu-Ag/TiO2-Based Optical Fibers at a Pilot Scale. Chem. Eng. J. 2021, 411, 128622. [Google Scholar] [CrossRef]
- Guo, D.; Feng, D.; Zhang, Y.; Zhang, Z.; Wu, J.; Zhao, Y.; Sun, S. Synergistic Mechanism of Biochar-Nano TiO2 Adsorption-Photocatalytic Oxidation of Toluene. Fuel Process. Technol. 2022, 229, 107200. [Google Scholar] [CrossRef]
- Weitekamp, C.A.; Stevens, T.; Stewart, M.J.; Bhave, P.; Gilmour, M.I. Health Effects from Freshly Emitted versus Oxidatively or Photochemically Aged Air Pollutants. Sci. Total Environ. 2020, 704, 135772. [Google Scholar] [CrossRef]
- Ajmal, Z.; ul Haq, M.; Naciri, Y.; Djellabi, R.; Hassan, N.; Zaman, S.; Murtaza, A.; Kumar, A.; Al-Sehemi, A.G.; Algarni, H.; et al. Recent Advancement in Conjugated Polymers Based Photocatalytic Technology for Air Pollutants Abatement: Cases of CO2, NOx, and VOCs. Chemosphere 2022, 308, 136358. [Google Scholar] [CrossRef]
- Tsai, W.T. An Overview of Health Hazards of Volatile Organic Compounds Regulated as Indoor Air Pollutants. Rev. Environ. Health 2019, 34, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Wolkoff, P.; Nielsen, G.D. Organic Compounds in Indoor Air-Their Relevance for Perceived Indoor Air Quality? Atmos. Environ. 2001, 35, 4407–4417. [Google Scholar] [CrossRef]
- Guieysse, B.; Hort, C.; Platel, V.; Munoz, R.; Ondarts, M.; Revah, S. Biological Treatment of Indoor Air for VOC Removal: Potential and Challenges. Biotechnol. Adv. 2008, 26, 398–410. [Google Scholar] [CrossRef]
- David, E.; Niculescu, V.C. Volatile Organic Compounds (Vocs) as Environmental Pollutants: Occurrence and Mitigation Using Nanomaterials. Int. J. Environ. Res. Public Health 2021, 18, 13147. [Google Scholar] [CrossRef]
- Wang, H.; Lou, S.; Huang, C.; Qiao, L.; Tang, X.; Chen, C.; Zeng, L.; Wang, Q.; Zhou, M.; Lu, S.; et al. Source Profiles of Volatile Organic Compounds from Biomass Burning in Yangtze River Delta, China. Aerosol Air Qual. Res. 2014, 14, 818–828. [Google Scholar] [CrossRef]
- Kamal, M.S.; Razzak, S.A.; Hossain, M.M. Catalytic Oxidation of Volatile Organic Compounds (VOCs)—A Review. Atmos. Environ. 2016, 140, 117–134. [Google Scholar] [CrossRef]
- Özçelik, Z.; Soylu, G.S.P.; Boz, I. Catalytic Combustion of Toluene over Mn, Fe and Co-Exchanged Clinoptilolite Support. Chem. Eng. J. 2009, 155, 94–100. [Google Scholar] [CrossRef]
- Patdhanagul, N.; Rangsriwatananon, K.; Siriwong, K.; Hengrasmee, S. Combined Modification of Zeolite NaY by Phenyl Trimethyl Ammonium Bromide and Potassium for Ethylene Gas Adsorption. Microporous Mesoporous Mater. 2012, 153, 30–34. [Google Scholar] [CrossRef]
- Bakheet, A.A.; Mohd Zain, M.F.; Kadhum, A.A.; Abdalla, Z. Photocatalytic Oxidation Performance to Removal of Volatile Organic Compounds in Indoor Environment. Environ. Res. Eng. Manag. 2011, 58, 27–33. [Google Scholar] [CrossRef]
- Halios, C.H.; Landeg-Cox, C.; Lowther, S.D.; Middleton, A.; Marczylo, T.; Dimitroulopoulou, S. Chemicals in European Residences–Part I: A Review of Emissions, Concentrations and Health Effects of Volatile Organic Compounds (VOCs). Sci. Total Environ. 2022, 839, 156201. [Google Scholar] [CrossRef]
- Wan, Y.; Zhao, D. On the Controllable Soft-Templating Approach to Mesoporous Silicates. Chem. Rev. 2007, 107, 2821–2860. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.; Ouyang, S.; Bi, Y.; Umezawa, N.; Oshikiri, M.; Ye, J. Nano-Photocatalytic Materials: Possibilities and Challenges. Adv. Mater. 2012, 24, 229–251. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Yang, C.; Lv, K.; Li, X.; Li, Q.; Fan, J. Single Atomic Au Induced Dramatic Promotion of the Photocatalytic Activity of TiO2 Hollow Microspheres. Chem. Commun. 2020, 56, 1745–1748. [Google Scholar] [CrossRef]
- He, F.; Weon, S.; Jeon, W.; Chung, M.W.; Choi, W. Self-Wetting Triphase Photocatalysis for Effective and Selective Removal of Hydrophilic Volatile Organic Compounds in Air. Nat. Commun. 2021, 12, 6259. [Google Scholar] [CrossRef] [PubMed]
- Sydorenko, J.; Mere, A.; Krunks, M.; Krichevskaya, M.; Acik, I.O. Transparent TiO2 Thin Films with High Photocatalytic Activity for Indoor Air Purification. RSC Adv. 2022, 12, 35531–35542. [Google Scholar] [CrossRef]
- Yamashita, H.; Mori, K.; Kuwahara, Y.; Kamegawa, T.; Wen, M.; Verma, P.; Che, M. Single-Site and Nano-Confined Photocatalysts Designed in Porous Materials for Environmental Uses and Solar Fuels. Chem. Soc. Rev. 2018, 47, 8072–8096. [Google Scholar] [CrossRef]
- Verma, P.; Kondo, Y.; Kuwahara, Y.; Kamegawa, T.; Mori, K.; Raja, R.; Yamashita, H. Design and Application of Photocatalysts Using Porous Materials. Catal. Rev. Sci. Eng. 2021, 63, 165–233. [Google Scholar] [CrossRef]
- Chu, S.; Wang, Y.; Guo, Y.; Feng, J.; Wang, C.; Luo, W.; Fan, X.; Zou, Z. Band Structure Engineering of Carbon Nitride: In Search of a Polymer Photocatalyst with High Photooxidation Property. ACS Catal. 2013, 3, 912–919. [Google Scholar] [CrossRef]
- Sun, R.; Shi, Q.; Zhang, M.; Xie, L.; Chen, J.; Yang, X.; Chen, M.; Zhao, W. Enhanced Photocatalytic Oxidation of Toluene with a Coral-like Direct Z-Scheme BiVO4/g-C3N4 Photocatalyst. J. Alloys Compd. 2017, 714, 619–626. [Google Scholar] [CrossRef]
- Zhong, D.K.; Sun, J.; Inumaru, H.; Gamelin, D.R. Solar Water Oxidation by Composite Catalyst/α-Fe2O3 Photoanodes. J. Am. Chem. Soc. 2009, 131, 6086–6087. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Mallouk, K.; Emamipour, H.; Rood, M.J.; Liu, X.; Yan, Z. Isobutane Adsorption with Carrier Gas Recirculation at Different Relative Humidities Using Activated Carbon Fiber Cloth and Electrothermal Regeneration. Chem. Eng. J. 2019, 360, 1011–1019. [Google Scholar] [CrossRef]
- Yue, Z.; Vakili, A.; Wang, J. Activated Carbon Fibers from Meltblown Isotropic Pitch Fiber Webs for Vapor Phase Adsorption of Volatile Organic Compounds. Chem. Eng. J. 2017, 330, 183–190. [Google Scholar] [CrossRef]
- Ahmad, A.; Ali, M.; Al-Sehemi, A.G.; Al-Ghamdi, A.A.; Park, J.W.; Algarni, H.; Anwer, H. Carbon-Integrated Semiconductor Photocatalysts for Removal of Volatile Organic Compounds in Indoor Environments. Chem. Eng. J. 2023, 452, 139436. [Google Scholar] [CrossRef]
- Yan, S.; Song, H.; Li, Y.; Yang, J.; Jia, X.; Wang, S.; Yang, X. Integrated Reduced Graphene Oxide/Polypyrrole Hybrid Aerogels for Simultaneous Photocatalytic Decontamination and Water Evaporation. Appl. Catal. B Environ. 2022, 301, 120820. [Google Scholar] [CrossRef]
- Enesca, A.; Cazan, C. Volatile Organic Compounds (VOCS) Removal from Indoor Air by Heterostructures/Composites/Doped Photocatalysts: A Mini-Review. Nanomaterials 2020, 10, 1965. [Google Scholar] [CrossRef] [PubMed]
- Akerdi, A.G.; Bahrami, S.H. Application of Heterogeneous Nano-Semiconductors for Photocatalytic Advanced Oxidation of Organic Compounds: A Review. J. Environ. Chem. Eng. 2019, 7, 103283. [Google Scholar] [CrossRef]
- Chen, H.; Nanayakkara, C.E.; Grassian, V.H. Titanium Dioxide Photocatalysis in Atmospheric Chemistry. Chem. Rev. 2012, 112, 5919–5948. [Google Scholar] [CrossRef]
- Mamaghani, A.H.; Haghighat, F.; Lee, C.S. Photocatalytic Oxidation Technology for Indoor Environment Air Purification: The State-of-the-Art. Appl. Catal. B Environ. 2017, 203, 247–269. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, X. Photocatalytic Oxidation for Indoor Air Purification: A Literature Review. Build. Environ. 2003, 38, 645–654. [Google Scholar] [CrossRef]
- Quici, N.; Vera, M.L.; Choi, H.; Puma, G.L.; Dionysiou, D.D.; Litter, M.I.; Destaillats, H. Effect of Key Parameters on the Photocatalytic Oxidation of Toluene at Low Concentrations in Air under 254 + 185 nm UV Irradiation. Appl. Catal. B Environ. 2010, 95, 312–319. [Google Scholar] [CrossRef]
- Van Durme, J.; Dewulf, J.; Sysmans, W.; Leys, C.; Van Langenhove, H. Abatement and Degradation Pathways of Toluene in Indoor Air by Positive Corona Discharge. Chemosphere 2007, 68, 1821–1829. [Google Scholar] [CrossRef] [PubMed]
- Korologos, C.A.; Philippopoulos, C.J.; Poulopoulos, S.G. The Effect of Water Presence on the Photocatalytic Oxidation of Benzene, Toluene, Ethylbenzene and m-Xylene in the Gas-Phase. Atmos. Environ. 2011, 45, 7089–7095. [Google Scholar] [CrossRef]
- Park, E.J.; Seo, H.O.; Kim, Y.D. Influence of Humidity on the Removal of Volatile Organic Compounds Using Solid Surfaces. Catal. Today 2017, 295, 3–13. [Google Scholar] [CrossRef]
- Talaiekhozani, A.; Rezania, S.; Kim, K.; Sanaye, R.; Mohammad, A. Recent Advances in Photocatalytic Removal of Organic and Inorganic Pollutants in Air. J. Clean. Prod. 2021, 278, 123895. [Google Scholar] [CrossRef]
- Assadi, A.A.; Bouzaza, A.; Wolbert, D. Photocatalytic Oxidation of Trimethylamine and Isovaleraldehyde in an Annular Reactor: Influence of the Mass Transfer and the Relative Humidity. J. Photochem. Photobiol. A Chem. 2012, 236, 61–69. [Google Scholar] [CrossRef]
- Vildozo, D.; Portela, R.; Ferronato, C.; Chovelon, J.M. Photocatalytic Oxidation of 2-Propanol/Toluene Binary Mixtures at Indoor Air Concentration Levels. Appl. Catal. B Environ. 2011, 107, 347–354. [Google Scholar] [CrossRef]
- Vincent, G.; Marquaire, P.M.; Zahraa, O. Photocatalytic Degradation of Gaseous 1-Propanol Using an Annular Reactor: Kinetic Modelling and Pathways. J. Hazard. Mater. 2009, 161, 1173–1181. [Google Scholar] [CrossRef]
- Debono, O.; Thévenet, F.; Gravejat, P.; Héquet, V.; Raillard, C.; Le Coq, L.; Locoge, N. Gas Phase Photocatalytic Oxidation of Decane at ppb Levels: Removal Kinetics, Reaction Intermediates and Carbon Mass Balance. J. Photochem. Photobiol. A Chem. 2013, 258, 17–29. [Google Scholar] [CrossRef]
- Kibanova, D.; Sleiman, M.; Cervini-Silva, J.; Destaillats, H. Adsorption and Photocatalytic Oxidation of Formaldehyde on a Clay-TiO2 Composite. J. Hazard. Mater. 2012, 211–212, 233–239. [Google Scholar] [CrossRef]
- Yu, K.P.; Whei-May Lee, G.; Huang, W.M.; Wu, C.C.; Lou, C.I.; Yang, S. Effectiveness of Photocatalytic Filter for Removing Volatile Organic Compounds in the Heating, Ventilation, and Air Conditioning System. J. Air Waste Manag. Assoc. 2006, 56, 666–674. [Google Scholar] [CrossRef]
- Mamaghani, A.H.; Haghighat, F.; Lee, C.S. Photocatalytic Oxidation of MEK over Hierarchical TiO2 Catalysts: Effect of Photocatalyst Features and Operating Conditions. Appl. Catal. B Environ. 2019, 251, 1–16. [Google Scholar] [CrossRef]
- Vildozo, D.; Ferronato, C.; Sleiman, M.; Chovelon, J.M. Photocatalytic Treatment of Indoor Air: Optimization of 2-Propanol Removal Using a Response Surface Methodology (RSM). Appl. Catal. B Environ. 2010, 94, 303–310. [Google Scholar] [CrossRef]
- Boulamanti, A.K.; Philippopoulos, C.J. Photocatalytic Degradation of C5-C7 Alkanes in the Gas-Phase. Atmos. Environ. 2009, 43, 3168–3174. [Google Scholar] [CrossRef]
- Shaw, J.T.; Rickard, A.R.; Newland, M.J.; Dillon, T.J. Rate Coefficients for Reactions of OH with Aromatic and Aliphatic Volatile Organic Compounds Determined by the Multivariate Relative Rate Technique. Atmos. Chem. Phys. 2020, 20, 9725–9736. [Google Scholar] [CrossRef]
- Ao, C.H.; Lee, S.C. Combination Effect of Activated Carbon with TiO2 for the Photodegradation of Binary Pollutants at Typical Indoor Air Level. J. Photochem. Photobiol. A Chem. 2004, 161, 131–140. [Google Scholar] [CrossRef]
- Li, Y.; Liu, F.; Li, M.; Li, W.; Qi, X.; Xue, M.; Wang, Y.; Han, F. Study on Adsorption Coupling Photodegradation on Hierarchical Nanostructured G-C3N4/TiO2/Activated Carbon Fiber Composites for Toluene Removal. J. Sol-Gel Sci. Technol. 2020, 93, 402–418. [Google Scholar] [CrossRef]
- Huang, H.; Xu, Y.; Feng, Q.; Leung, D.Y.C. Low Temperature Catalytic Oxidation of Volatile Organic Compounds: A Review. Catal. Sci. Technol. 2015, 5, 2649–2669. [Google Scholar] [CrossRef]
- Malayeri, M.; Haghighat, F.; Lee, C.S. Modeling of Volatile Organic Compounds Degradation by Photocatalytic Oxidation Reactor in Indoor Air: A Review. Build. Environ. 2019, 154, 309–323. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, D.; Ma, C.; Yang, H. The Effect of Activated Carbon Adsorption on the Photocatalytic Removal of Formaldehyde. Build. Environ. 2010, 45, 615–621. [Google Scholar] [CrossRef]
- Li, X.; Qian, X.; An, X.; Huang, J. Preparation of a Novel Composite Comprising Biochar Skeleton and “Chrysanthemum” g-C3N4 for Enhanced Visible Light Photocatalytic Degradation of Formaldehyde. Appl. Surf. Sci. 2019, 487, 1262–1270. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Matsushima, S.; Hojo, H.; Einaga, H. Photocatalytic Oxidation Process for Treatment of Gas Phase Benzene Using Ti3+ Self-Doped TiO2 Microsphere with Sea Urchin-like Structure. Chem. Eng. J. 2020, 402, 126220. [Google Scholar] [CrossRef]
- Verbruggen, S.W.; Ribbens, S.; Tytgat, T.; Hauchecorne, B.; Smits, M.; Meynen, V.; Cool, P.; Martens, J.A.; Lenaerts, S. The Benefit of Glass Bead Supports for Efficient Gas Phase Photocatalysis: Case Study of a Commercial and a Synthesised Photocatalyst. Chem. Eng. J. 2011, 174, 318–325. [Google Scholar] [CrossRef]
- Thevenet, F.; Guaïtella, O.; Herrmann, J.M.; Rousseau, A.; Guillard, C. Photocatalytic Degradation of Acetylene over Various Titanium Dioxide-Based Photocatalysts. Appl. Catal. B Environ. 2005, 61, 58–68. [Google Scholar] [CrossRef]
- Mo, J.; Zhang, Y.; Xu, Q.; Yang, R. Effect of TiO2/Adsorbent Hybrid Photocatalysts for Toluene Decomposition in Gas Phase. J. Hazard. Mater. 2009, 168, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Liu, Z. Study on Light Intensity in the Process of Photocatalytic Degradation of Indoor Gaseous Formaldehyde for Saving Energy. Energy Convers. Manag. 2007, 48, 882–889. [Google Scholar] [CrossRef]
- Kibanova, D.; Cervini-Silva, J.; Destaillats, H. Efficiency of Clay-TiO2 Nanocomposites on the Photocatalytic Elimination of a Model Hydrophobic Air Pollutant. Environ. Sci. Technol. 2009, 43, 1500–1506. [Google Scholar] [CrossRef]
- Jeong, J.; Sekiguchi, K.; Lee, W.; Sakamoto, K. Photodegradation of Gaseous Volatile Organic Compounds (VOCs) Using TiO2 Photoirradiated by an Ozone-Producing UV Lamp: Decomposition Characteristics, Identification of by-Products and Water-Soluble Organic Intermediates. J. Photochem. Photobiol. A Chem. 2005, 169, 279–287. [Google Scholar] [CrossRef]
- Krichevskaya, M.; Preis, S.; Moiseev, A.; Pronina, N.; Deubener, J. Gas-Phase Photocatalytic Oxidation of Refractory VOCs Mixtures: Through the Net of Process Limitations. Catal. Today 2017, 280, 93–98. [Google Scholar] [CrossRef]
- Zhang, G.; Peyravi, A.; Hashisho, Z.; Sun, Z.; Liu, Y.; Zheng, S.; Zhong, L. Integrated Adsorption and Photocatalytic Degradation of VOCs Using a TiO2/Diatomite Composite: Effects of Relative Humidity and Reaction Atmosphere. Catal. Sci. Technol. 2020, 10, 2378–2388. [Google Scholar] [CrossRef]
- Liang, W.J.; Li, J.; Jin, Y.Q. Photocatalytic Degradation of Gaseous Acetone, Toluene, and p-Xylene Using a TiO2 Thin Film. J. Environ. Sci. Health Part A Toxic/Hazardous Subst. Environ. Eng. 2010, 45, 1384–1390. [Google Scholar] [CrossRef]
- Dong, F.; Guo, S.; Wang, H.; Li, X.; Wu, Z. Enhancement of the Visible Light Photocatalytic Activity of C-Doped TiO2 Nanomaterials Prepared by a Green Synthetic Approach. J. Phys. Chem. C 2011, 115, 13285–13292. [Google Scholar] [CrossRef]
- Dong, X.; Cui, Z.; Sun, Y.; Dong, F. Humidity-Independent Photocatalytic Toluene Mineralization Benefits from the Utilization of Edge Hydroxyls in Layered Double Hydroxides (LDHs): A Combined Operando and Theoretical Investigation. ACS Catal. 2021, 11, 8132–8139. [Google Scholar] [CrossRef]
- You, J.-W.; Vikrant, K.; Lim, D.-H.; Verma, S.; Kukkar, D.; Anwer, H.; Kim, K.-H. Photocatalytic Potential of a Titanium Dioxide–Supported Platinum Catalyst against VOCs with Complicated Composition under Varying Humidity Conditions. J. Clean. Prod. 2022, 371, 133487. [Google Scholar] [CrossRef]
- An, T.; Chen, J.; Nie, X.; Li, G.; Zhang, H.; Liu, X.; Zhao, H. Synthesis of Carbon Nanotube-Anatase TiO2 Sub-Micrometer-Sized Sphere Composite Photocatalyst for Synergistic Degradation of Gaseous Styrene. ACS Appl. Mater. Interfaces 2012, 4, 5988–5996. [Google Scholar] [CrossRef]
- Zhang, J.; Vikrant, K.; Kim, K.H.; Dong, F.; Won Chung, M.; Weon, S. Unveiling the Collective Effects of Moisture and Oxygen on the Photocatalytic Degradation of M-Xylene Using a Titanium Dioxide Supported Platinum Catalyst. Chem. Eng. J. 2022, 439, 135747. [Google Scholar] [CrossRef]
- Sidheswaran, M.; Tavlarides, L.L. Visible Light Photocatalytic Oxidation of Toluene Using a Cerium-Doped Titania Catalyst. Ind. Eng. Chem. Res. 2008, 47, 3346–3357. [Google Scholar] [CrossRef]
- Arai, T.; Horiguchi, M.; Yanagida, M.; Gunji, T.; Sugihara, H.; Sayama, K. Complete Oxidation of Acetaldehyde and Toluene over a Pd/WO3 Photocatalyst under Fluorescent or Visible-Light Irradiation. Chem. Commun. 2008, 5565–5567. [Google Scholar] [CrossRef]
- Gao, Z.; Wang, J.; Muhammad, Y.; Zhang, Y.; Jalil, S.; Hu, Y. Enhanced Moisture-Resistance and Excellent Photocatalytic Performance of Synchronous N/Zn-Decorated MIL-125(Ti) for Vaporous Acetaldehyde Degradation. Chem. Eng. J. 2020, 388, 124389. [Google Scholar] [CrossRef]
- Zhong, L.; Haghighat, F. Modeling of By-Products from Photocatalytic Oxidation (PCO) Indoor Air Purifiers: A Case Study of Ethanol. Build. Environ. 2018, 144, 427–436. [Google Scholar] [CrossRef]
- Whyte, H.E.; Raillard, C.; Subrenat, A.; Héquet, V. Photocatalytic Oxidation of Isoflurane, an Anesthetic Gas: The Influence of Operating Parameters. Chem. Eng. J. 2018, 352, 441–449. [Google Scholar] [CrossRef]
- Kamaei, M.; Rashedi, H.; Dastgheib, S.M.M.; Tasharrofi, S. Comparing Photocatalytic Degradation of Gaseous Ethylbenzene Using N-Doped and Pure TiO2 Nano-Catalysts Coated on Glass Beads under Both UV and Visible Light Irradiation. Catalysts 2018, 8, 466. [Google Scholar] [CrossRef]
- Xie, R.; Lei, D.; Zhan, Y.; Liu, B.; Tsang, C.H.A.; Zeng, Y.; Li, K.; Leung, D.Y.C.; Huang, H. Efficient Photocatalytic Oxidation of Gaseous Toluene over F-Doped TiO2 in a Wet Scrubbing Process. Chem. Eng. J. 2020, 386, 121025. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, S.; Li, K.; Wang, J.; Chen, P.; Wang, H.; Li, J.; Dong, F. Promote the Activation and Ring Opening of Intermediates for Stable Photocatalytic Toluene Degradation over Zn-Ti-LDH. J. Colloid Interface Sci. 2022, 606, 1435–1444. [Google Scholar] [CrossRef]
- Chen, L.; Wang, X.; Rao, Z.; Tang, Z.; Wang, Y.; Shi, G.; Lu, G.; Xie, X.; Chen, D.; Sun, J. In-Situ Synthesis of Z-Scheme MIL-100(Fe)/α-Fe2O3 Heterojunction for Enhanced Adsorption and Visible-Light Photocatalytic Oxidation of O-Xylene. Chem. Eng. J. 2021, 416, 129112. [Google Scholar] [CrossRef]
- Jafari, A.J.; Kalantary, R.R.; Esrafili, A.; Arfaeinia, H. Synthesis of Silica-Functionalized Graphene Oxide/ZnO Coated on Fiberglass and Its Application in Photocatalytic Removal of Gaseous Benzene. Process Saf. Environ. Prot. 2018, 116, 377–387. [Google Scholar] [CrossRef]
- Lei, B.; Cui, W.; Chen, P.; Chen, R.; Sun, Y.; Kim, K.H.; Dong, F. Rational Design of LDH/Zn2SnO4 Heterostructures for Efficient Mineralization of Toluene Through Boosted Interfacial Charge Separation. Energy Environ. Mater. 2022, 6, e12291. [Google Scholar] [CrossRef]
- Liqiang, J.; Baifu, X.; Fulong, Y.; Baiqi, W.; Keying, S.; Weimin, C.; Honggang, F. Deactivation and Regeneration of ZnO and TiO2 Nanoparticles in the Gas Phase Photocatalytic Oxidation of N-C7H16 or SO2. Appl. Catal. A Gen. 2004, 275, 49–54. [Google Scholar] [CrossRef]
- Einaga, H.; Futamura, S.; Ibusuki, T. Heterogeneous Photocatalytic Oxidation of Benzene, Toluene, Cyclohexene and Cyclohexane in Humidified Air: Comparison of Decomposition Behavior on Photoirradiated TiO2 Catalyst. Appl. Catal. B Environ. 2002, 38, 215–225. [Google Scholar] [CrossRef]
- Taranto, J.; Frochot, D.; Pichat, P. Photocatalytic Air Purification: Comparative Efficacy and Pressure Drop of a TiO2-Coated Thin Mesh and a Honeycomb Monolith at High Air Velocities Using a 0.4 M3 Close-Loop Reactor. Sep. Purif. Technol. 2009, 67, 187–193. [Google Scholar] [CrossRef]
- Thevenet, F.; Guillard, C.; Rousseau, A. Acetylene Photocatalytic Oxidation Using Continuous Flow Reactor: Gas Phase and Adsorbed Phase Investigation, Assessment of the Photocatalyst Deactivation. Chem. Eng. J. 2014, 244, 50–58. [Google Scholar] [CrossRef]
- Hay, S.O.; Obee, T.N.; Thibaud-Erkey, C. The Deactivation of Photocatalytic Based Air Purifiers by Ambient Siloxanes. Appl. Catal. B Environ. 2010, 99, 435–441. [Google Scholar] [CrossRef]
- Cha, B.J.; Woo, T.G.; Han, S.W.; Saqlain, S.; Seo, H.O.; Cho, H.K.; Kim, J.Y.; Kim, Y.D. Surface Modification of TiO2 for Obtaining High Resistance against Poisoning during Photocatalytic Decomposition of Toluene. Catalysts 2018, 8, 500. [Google Scholar] [CrossRef]
- Tripathy, J.; Lee, K.; Schmuki, P. Tuning the Selectivity of Photocatalytic Synthetic Reactions Using Modified TiO2 Nanotubes. Angew. Chemie 2014, 126, 12813–12816. [Google Scholar] [CrossRef]
- Weon, S.; He, F.; Choi, W. Status and Challenges in Photocatalytic Nanotechnology for Cleaning Air Polluted with Volatile Organic Compounds: Visible Light Utilization and Catalyst Deactivation. Environ. Sci. Nano 2019, 6, 3185–3214. [Google Scholar] [CrossRef]
- Chen, P.; Cui, W.; Wang, H.; Dong, X.; Li, J.; Sun, Y.; Zhou, Y.; Zhang, Y.; Dong, F. The Importance of Intermediates Ring-Opening in Preventing Photocatalyst Deactivation during Toluene Decomposition. Appl. Catal. B Environ. 2020, 272, 118977. [Google Scholar] [CrossRef]
- Verbruggen, S.W.; Masschaele, K.; Moortgat, E.; Korany, T.E.; Hauchecorne, B.; Martens, J.A.; Lenaerts, S. Factors Driving the Activity of Commercial Titanium Dioxide Powders towards Gas Phase Photocatalytic Oxidation of Acetaldehyde. Catal. Sci. Technol. 2012, 2, 2311–2318. [Google Scholar] [CrossRef]
- Taranto, J.; Frochot, D.; Pichat, P. Photocatalytic Treatment of Air: Comparison of Various TiO2, Coating Methods, and Supports Using Methanol or n-Octane as Test Pollutant. Ind. Eng. Chem. Res. 2009, 48, 6229–6236. [Google Scholar] [CrossRef]
- Dhada, I.; Nagar, P.K.; Sharma, M. Challenges of TiO2-Based Photooxidation of Volatile Organic Compounds: Designing, Coating, and Regenerating Catalyst. Ind. Eng. Chem. Res. 2015, 54, 5381–5387. [Google Scholar] [CrossRef]
- Chemweno, M.K.; Cernohlavek, L.G.; Jacoby, W.A. Deactivation of Titanium Dioxide Photocatalyst by Oxidation of Polydimethylsiloxane and Silicon Sealant Off-Gas in a Recirculating Batch Reactor. J. Air Waste Manag. Assoc. 2008, 58, 12–18. [Google Scholar] [CrossRef]
- De Sun, R.; Nakajima, A.; Watanabe, T.; Hashimoto, K. Decomposition of Gas-Phase Octamethyltrisiloxane on TiO2 Thin Film Photocatalysts-Catalytic Activity, Deactivation, and Regeneration. J. Photochem. Photobiol. A Chem. 2003, 154, 203–209. [Google Scholar] [CrossRef]
- Ajmal, A.; Majeed, I.; Malik, R.N.; Idriss, H.; Nadeem, M.A. Principles and Mechanisms of Photocatalytic Dye Degradation on TiO2 Based Photocatalysts: A Comparative Overview. RSC Adv. 2014, 4, 37003–37026. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, M.; Lei, L. Preparation of Photocatalytic TiO2 Coatings of Nanosized Particles on Activated Carbon by AP-MOCVD. Carbon N. Y. 2005, 43, 1700–1708. [Google Scholar] [CrossRef]
- Araña, J.; Doña-Rodríguez, J.M.; Cabo, C.G.I.; González-Díaz, O.; Herrera-Melián, J.A.; Pérez-Peña, J. FTIR Study of Gas-Phase Alcohols Photocatalytic Degradation with TiO2 and AC-TiO2. Appl. Catal. B Environ. 2004, 53, 221–232. [Google Scholar] [CrossRef]
- Yao, P.; Liu, H.; Wang, D.; Chen, J.; Li, G.; An, T. Enhanced Visible-Light Photocatalytic Activity to Volatile Organic Compounds Degradation and Deactivation Resistance Mechanism of Titania Confined inside a Metal-Organic Framework. J. Colloid Interface Sci. 2018, 522, 174–182. [Google Scholar] [CrossRef]
- Suárez, S.; Coronado, J.M.; Portela, R.; Martín, J.C.; Yates, M.; Avila, P.; Sánchez, B. On the Preparation of TiO2-Sepiolite Hybrid Materials for the Photocatalytic Degradation of TCE: Influence of TiO2 Distribution in the Mineralization. Environ. Sci. Technol. 2008, 42, 5892–5896. [Google Scholar] [CrossRef]
- Qian, X.; Ren, M.; Yue, D.; Zhu, Y.; Han, Y.; Bian, Z.; Zhao, Y. Mesoporous TiO2 Films Coated on Carbon Foam Based on Waste Polyurethane for Enhanced Photocatalytic Oxidation of VOCs. Appl. Catal. B Environ. 2017, 212, 1–6. [Google Scholar] [CrossRef]
- Yan, T.; Long, J.; Shi, X.; Wang, D.; Li, Z.; Wang, X. Efficient Photocatalytic Degradation of Volatile Organic Compounds by Porous Indium Hydroxide Nanocrystals. Environ. Sci. Technol. 2010, 44, 1380–1385. [Google Scholar] [CrossRef]








| VOCs | Major Indoor Sources |
|---|---|
| Toluene | Paints, gasoline, varnishes, solvents, polishes, anti-freezing materials, pesticides |
| Benzene | Glues, adhesives, detergents, tobacco smoke, gypsum board |
| Naphthalene | Mothballs, dyes, smoke, insulating materials |
| Ethylbenzene | Paints, gasoline, inks, glues, candles, insecticides |
| Chloroform | Solvents, glues, chlorinated water |
| Ethylene | Food products, gasoline |
| Formaldehyde | Wood products, wallpaper and paints, flooring items, foam, wallpaper paste, hair conditioners |
| Tetrachloroethylene | Water repellents, wood cleaners, adhesives, dry-cleaned clothes |
| Acetaldehyde | Wood products, deodorants, building materials and furnishings, carpets, gypsum |
| Trichloroethylene | Metal cleaners, adhesives, flooring materials, paint removers |
| Styrene | Paints, detergents, plasters, adhesives, modelling clay |
| Acetone | Solvents, sprays, cosmetics, floor coverings, resins, disinfectants, air fresheners |
| Catalyst | VOC Type | Concentration | RH (%) | Light Source | Removal Efficiency (%) | Ref. |
|---|---|---|---|---|---|---|
| TiO2/diatomite composite | Acetone, MEK | 10 ppm | 15 | UVA lamp | ~42–61 | [82] |
| TiO2 thin film | Acetone, toluene, p-xylene | 0.1–0.3 mol/m3 | 35 | UV lamp | ~55–77 | [83] |
| C-doped TiO2 | Toluene | 150 mg/m3 | 60 | Visible light | ~60 | [84] |
| TiO2/Mg-Al LDH | Toluene | 100 ppm | 50 | UV lamp | ~74 | [85] |
| Pt-TiO2 | MEK | 1 ppm | 100 | UV lamp | ~73 | [86] |
| CNT-TiO2 | Gaseous styrene | 25 ppm | 0 | UV-LED light | ~50 | [87] |
| Pt-TiO2-R | m-xylene | 1 ppm | 50 | UV lamp | ~73 | [88] |
| Ce-TiO2 | Toluene | 150–600 ppb | <3 | Visible light | ~22 | [89] |
| Pd/WO3 | Acetaldehyde | 5 ppm | 50 | Fluorescent-visible light | ~99 | [90] |
| Pt-TiO2 | BuAc | 1 ppm | 100 | UV lamp | ~98 | [86] |
| TiO2/diatomite composite | 2-Heptanone | 10 ppm | 15 | UVA lamp | ~38 | [82] |
| MOF(Ti) | Acetaldehyde | 200 ppm | 80 | Visible light | ~98 | [91] |
| TiO2 | Ethanol | 200–2000 ppb | 9–60 | UVC lamp | ~4–44 | [92] |
| SiO2 coated TiO2 | Isoflurane | 500 ppb | 50 | UVC fluorescent lamp | ~100 | [93] |
| N-doped TiO2 | Ethyl benzene | 20–140 ppm | 50 | TUV lamp | ~25–100 | [94] |
| F-TiO2 | Toluene | 30 ppm | 50 | VUV lamp | ~80 | [95] |
| Zn-Ti-LDH | Toluene | 500 ppm | 50 | UV lamp | ~75 | [96] |
| MOF(Fe)/Fe2O3 | o-xylene | 25 ppm | 50 | Xenon lamp | ~100 | [97] |
| GO/ZnO | Benzene | 100 mg/L | 35–45 | UV lamp | ~87 | [98] |
| Zn2SO4/LDH | Toluene | 500 ppm | 60 | UV lamp | ~90 | [99] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaison, A.; Mohan, A.; Lee, Y.-C. Recent Developments in Photocatalytic Nanotechnology for Purifying Air Polluted with Volatile Organic Compounds: Effect of Operating Parameters and Catalyst Deactivation. Catalysts 2023, 13, 407. https://doi.org/10.3390/catal13020407
Jaison A, Mohan A, Lee Y-C. Recent Developments in Photocatalytic Nanotechnology for Purifying Air Polluted with Volatile Organic Compounds: Effect of Operating Parameters and Catalyst Deactivation. Catalysts. 2023; 13(2):407. https://doi.org/10.3390/catal13020407
Chicago/Turabian StyleJaison, Augustine, Anandhu Mohan, and Young-Chul Lee. 2023. "Recent Developments in Photocatalytic Nanotechnology for Purifying Air Polluted with Volatile Organic Compounds: Effect of Operating Parameters and Catalyst Deactivation" Catalysts 13, no. 2: 407. https://doi.org/10.3390/catal13020407