A Current Update on the Role of HDL-Based Nanomedicine in Targeting Macrophages in Cardiovascular Disease
Abstract
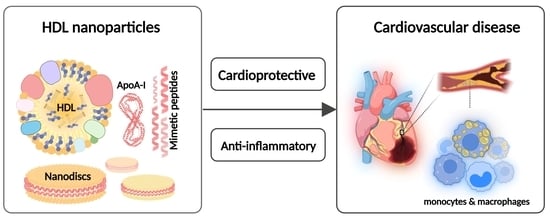
:1. Introduction
2. The Mononuclear Phagocytes Orchestrate Vascular Inflammation and Remodeling
3. High-Density Lipoproteins Are Self-Assembling Nanoparticles
3.1. HDL Composition and Function
3.1.1. Protein Components
Apolipoproteins
HDL Associated Enzymes
Enzymes and Proteins Partially Associated with HDL
3.1.2. HDL-Associated Lipids and Steroids
3.1.3. Other Constituents
4. Anti-Inflammatory Effects of Native HDL: Focus on Mononuclear Phagocytes
4.1. Anti-Inflammatory Mechanisms of HDL and ApoA-I
4.2. Other Anti-Inflammatory Components of HDL
5. HDL Particles Are Remodeled in Cardiovascular Disease and Lose Their Function
6. The Era of HDL Mimetic Nanoparticles: Replenishing HDL Quality and Quantity
6.1. Protein Components of HDL Mimetic Nanoparticles
6.1.1. ApoA-I Full-Length Protein
6.1.2. ApoA-I Mimetic Peptides
6.1.3. Non-ApoA-I Mimetic Peptides
6.2. Lipid Components of HDL Mimetic Nanoparticles
7. Anti-Inflammatory Effects of Synthetic/Reconstituted HDL Mimetic Nanoparticles
7.1. Effects of HDL Nanodiscs Containing Full-Length ApoA-I on Monocytes/Macrophages
7.2. Effects of HDL Nanodiscs Containing ApoA-I Mimetic Peptides on Monocytes/Macrophages
7.3. Anti-Inflammatory Effects of HDL Mimetics on Other Cell Types
8. HDL Nanotherapeutics: Existing Strategies and Clinical Trials
8.1. ApoA-I Full-Length Protein: Reconstituted HDL (rHDL) Nanoparticles
8.1.1. Clinical Trials with Non-Lipidated ApoA-I
8.1.2. Clinical Trials with Lipidated ApoA-I: rHDL Formulations
8.2. Clinical Trials with ApoA-I Mimetics
9. Future Outlook and Perspectives
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Feingold, K.R.; Grunfeld, C. Introduction to Lipids and Lipoproteins. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., Dungan, K., Grossman, A., Hershman, J.M., Kaltsas, G., Koch, C., Kopp, P., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Olson, R.E. Discovery of the Lipoproteins, Their Role in Fat Transport and Their Significance as Risk Factors. J. Nutr. 1998, 128, 439S–443S. [Google Scholar] [CrossRef] [PubMed]
- Gordon, T.; Castelli, W.P.; Hjortland, M.C.; Kannel, W.B.; Dawber, T.R. High Density Lipoprotein as a Protective Factor against Coronary Heart Disease. Am. J. Med. 1977, 62, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Asztalos, B.F.; Demissie, S.; Cupples, L.A.; Collins, D.; Cox, C.E.; Horvath, K.V.; Bloomfield, H.E.; Robins, S.J.; Schaefer, E.J. LpA-I, LpA-I:A-II HDL and CHD-Risk: The Framingham Offspring Study and the Veterans Affairs HDL Intervention Trial. Atherosclerosis 2006, 188, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Freund, K.M.; Belanger, A.J.; D’Agostino, R.B.; Kannel, W.B. The Health Risks of Smoking the Framingham Study: 34 Years of Follow-Up. Ann. Epidemiol. 1993, 3, 417–424. [Google Scholar] [CrossRef]
- Casula, M.; Colpani, O.; Xie, S.; Catapano, A.L.; Baragetti, A. HDL in Atherosclerotic Cardiovascular Disease: In Search of a Role. Cells 2021, 10, 1869. [Google Scholar] [CrossRef]
- Ali, K.M.; Wonnerth, A.; Huber, K.; Wojta, J. Cardiovascular Disease Risk Reduction by Raising HDL Cholesterol—Current Therapies and Future Opportunities. Br. J. Pharmacol. 2012, 167, 1177–1194. [Google Scholar] [CrossRef]
- Riaz, H.; Khan, S.U.; Rahman, H.; Shah, N.P.; Kaluski, E.; Lincoff, A.M.; Nissen, S.E. Effects of High-Density Lipoprotein Targeting Treatments on Cardiovascular Outcomes: A Systematic Review and Meta-Analysis. Eur. J. Prev. Cardiol. 2019, 26, 533–543. [Google Scholar] [CrossRef]
- Kingwell, B.A.; Chapman, M.J.; Kontush, A.; Miller, N.E. HDL-Targeted Therapies: Progress, Failures and Future. Nat. Rev. Drug Discov. 2014, 13, 445–464. [Google Scholar] [CrossRef]
- Cochran, B.J.; Ong, K.-L.; Manandhar, B.; Rye, K.-A. APOA1: A Protein with Multiple Therapeutic Functions. Curr. Atheroscler. Rep. 2021, 23, 11. [Google Scholar] [CrossRef]
- Trakaki, A.; Marsche, G. Current Understanding of the Immunomodulatory Activities of High-Density Lipoproteins. Biomedicines 2021, 9, 587. [Google Scholar] [CrossRef]
- Darabi, M.; Kontush, A. High-Density Lipoproteins (HDL): Novel Function and Therapeutic Applications. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2022, 1867, 159058. [Google Scholar] [CrossRef]
- Reisinger, A.C.; Schuller, M.; Sourij, H.; Stadler, J.T.; Hackl, G.; Eller, P.; Marsche, G. Impact of Sepsis on High-Density Lipoprotein Metabolism. Front. Cell Dev. Biol. 2022, 9, 3753. [Google Scholar] [CrossRef]
- Holzwirth, E.; Fischer-Schaepmann, T.; Obradovic, D.; von Lucadou, M.; Schwedhelm, E.; Daum, G.; Hindricks, G.; Marsche, G.; Trieb, M.; Thiele, H.; et al. Anti-Inflammatory HDL Effects Are Impaired in Atrial Fibrillation. Heart Vessel. 2022, 37, 161–171. [Google Scholar] [CrossRef]
- Trakaki, A.; Sturm, G.J.; Pregartner, G.; Scharnagl, H.; Eichmann, T.O.; Trieb, M.; Knuplez, E.; Holzer, M.; Stadler, J.T.; Heinemann, A.; et al. Allergic Rhinitis Is Associated with Complex Alterations in High-Density Lipoprotein Composition and Function. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2019, 1864, 1280–1292. [Google Scholar] [CrossRef]
- Trieb, M.; Horvath, A.; Birner-Gruenberger, R.; Spindelboeck, W.; Stadlbauer, V.; Taschler, U.; Curcic, S.; Stauber, R.E.; Holzer, M.; Pasterk, L.; et al. Liver Disease Alters High-Density Lipoprotein Composition, Metabolism and Function. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2016, 1861, 630–638. [Google Scholar] [CrossRef]
- Marsche, G.; Saemann, M.D.; Heinemann, A.; Holzer, M. Inflammation Alters HDL Composition and Function: Implications for HDL-Raising Therapies. Pharmacol. Ther. 2013, 137, 341–351. [Google Scholar] [CrossRef]
- Holzer, M.; Wolf, P.; Curcic, S.; Birner-Gruenberger, R.; Weger, W.; Inzinger, M.; El-Gamal, D.; Wadsack, C.; Heinemann, A.; Marsche, G. Psoriasis Alters HDL Composition and Cholesterol Efflux Capacity. J. Lipid Res. 2012, 53, 1618–1624. [Google Scholar] [CrossRef]
- Stadler, J.T.; Marsche, G. Obesity-Related Changes in High-Density Lipoprotein Metabolism and Function. Int. J. Mol. Sci. 2020, 21, 8985. [Google Scholar] [CrossRef]
- Chiesa, S.T.; Charakida, M. High-Density Lipoprotein Function and Dysfunction in Health and Disease. Cardiovasc. Drugs Ther. 2019, 33, 207–219. [Google Scholar] [CrossRef]
- Hui, N.; Barter, P.J.; Ong, K.-L.; Rye, K.-A. Altered HDL Metabolism in Metabolic Disorders: Insights into the Therapeutic Potential of HDL. Clin. Sci. 2019, 133, 2221–2235. [Google Scholar] [CrossRef]
- Kim, S.Y.; Yu, M.; Morin, E.E.; Kang, J.; Kaplan, M.J.; Schwendeman, A. High-Density Lipoprotein in Lupus: Disease Biomarkers and Potential Therapeutic Strategy. Arthritis Rheumatol. 2020, 72, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Deprince, A.; Haas, J.T.; Staels, B. Dysregulated Lipid Metabolism Links NAFLD to Cardiovascular Disease. Mol. Metab. 2020, 42, 101092. [Google Scholar] [CrossRef]
- Zimetti, F.; Adorni, M.P.; Marsillach, J.; Marchi, C.; Trentini, A.; Valacchi, G.; Cervellati, C. Connection between the Altered HDL Antioxidant and Anti-Inflammatory Properties and the Risk to Develop Alzheimer’s Disease: A Narrative Review. Oxid. Med. Cell. Longev. 2021, 2021, 6695796. [Google Scholar] [CrossRef] [PubMed]
- Barker, G.; Leeuwenburgh, C.; Brusko, T.; Moldawer, L.; Reddy, S.; Guirgis, F. Lipid and Lipoprotein Dysregulation in Sepsis: Clinical and Mechanistic Insights into Chronic Critical Illness. J. Clin. Med. 2021, 10, 1693. [Google Scholar] [CrossRef] [PubMed]
- Bonilha, I.; Zimetti, F.; Zanotti, I.; Papotti, B.; Sposito, A.C. Dysfunctional High-Density Lipoproteins in Type 2 Diabetes Mellitus: Molecular Mechanisms and Therapeutic Implications. J. Clin. Med. 2021, 10, 2233. [Google Scholar] [CrossRef]
- Bonizzi, A.; Piuri, G.; Corsi, F.; Cazzola, R.; Mazzucchelli, S. HDL Dysfunctionality: Clinical Relevance of Quality Rather Than Quantity. Biomedicines 2021, 9, 729. [Google Scholar] [CrossRef]
- Ossoli, A.; Cetti, F.; Gomaraschi, M. Air Pollution: Another Threat to HDL Function. Int. J. Mol. Sci. 2022, 24, 317. [Google Scholar] [CrossRef]
- Pirillo, A.; Catapano, A.L.; Norata, G.D. Biological Consequences of Dysfunctional HDL. Curr. Med. Chem. 2019, 26, 1644–1664. [Google Scholar] [CrossRef]
- Begue, F.; Tanaka, S.; Mouktadi, Z.; Rondeau, P.; Veeren, B.; Diotel, N.; Tran-Dinh, A.; Robert, T.; Vélia, E.; Mavingui, P.; et al. Altered high-density lipoprotein composition and functions during severe COVID-19. Sci. Rep. 2021, 11, 2291. [Google Scholar] [CrossRef]
- Mocciaro, G.; D’Amore, S.; Jenkins, B.; Kay, R.; Murgia, A.; Herrera-Marcos, L.V.; Neun, S.; Sowton, A.P.; Hall, Z.; Palma-Duran, S.A.; et al. Lipidomic Approaches to Study HDL Metabolism in Patients with Central Obesity Diagnosed with Metabolic Syndrome. Int. J. Mol. Sci. 2022, 23, 6786. [Google Scholar] [CrossRef]
- Wang, D.; Yu, B.; Li, Q.; Guo, Y.; Koike, T.; Koike, Y.; Wu, Q.; Zhang, J.; Mao, L.; Tang, X.; et al. HDL Quality Features Revealed by Proteome-lipidome Connectivity Are Associated with Atherosclerotic Disease. J. Mol. Cell. Biol. 2022, 14, mjac004. [Google Scholar] [CrossRef]
- Krause, B.R.; Remaley, A.T. Reconstituted HDL for the Acute Treatment of Acute Coronary Syndrome. Curr. Opin. Lipidol. 2013, 24, 480–486. [Google Scholar] [CrossRef]
- Tsujita, M.; Wolska, A.; Gutmann, D.A.; Remaley, A.T. Reconstituted Discoidal High-Density Lipoproteins: Bioinspired Nanodiscs with Many Unexpected Applications. Curr. Atheroscler. Rep. 2018, 20, 59. [Google Scholar] [CrossRef]
- He, H.; Hong, K.; Liu, L.; Schwendeman, A. Artificial High-Density Lipoprotein-Mimicking Nanotherapeutics for the Treatment of Cardiovascular Diseases. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2021, 13, e1737. [Google Scholar] [CrossRef]
- Kuai, R.; Li, D.; Chen, Y.E.; Moon, J.J.; Schwendeman, A. High-Density Lipoproteins: Nature’s Multifunctional Nanoparticles. ACS Nano 2016, 10, 3015–3041. [Google Scholar] [CrossRef]
- Tricoci, P.; D’Andrea, D.M.; Gurbel, P.A.; Yao, Z.; Cuchel, M.; Winston, B.; Schott, R.; Weiss, R.; Blazing, M.A.; Cannon, L.; et al. Infusion of Reconstituted High-Density Lipoprotein, CSL112, in Patients with Atherosclerosis: Safety and Pharmacokinetic Results from a Phase 2a Randomized Clinical Trial. J. Am. Heart Assoc. 2015, 4, e002171. [Google Scholar] [CrossRef]
- Nanjee, M.N.; Doran, J.E.; Lerch, P.G.; Miller, N.E. Acute Effects of Intravenous Infusion of ApoA1/Phosphatidylcholine Discs on Plasma Lipoproteins in Humans. Arter. Thromb. Vasc. Biol. 1999, 19, 979–989. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Puri, R.; Ballantyne, C.M.; Jukema, J.W.; Kastelein, J.J.P.; Koenig, W.; Wright, R.S.; Kallend, D.; Wijngaard, P.; Borgman, M.; et al. Effect of Infusion of High-Density Lipoprotein Mimetic Containing Recombinant Apolipoprotein A-I Milano on Coronary Disease in Patients with an Acute Coronary Syndrome in the MILANO-PILOT Trial. JAMA Cardiol. 2018, 3, 806. [Google Scholar] [CrossRef]
- Park, M.D.; Silvin, A.; Ginhoux, F.; Merad, M. Macrophages in Health and Disease. Cell 2022, 185, 4259–4279. [Google Scholar] [CrossRef]
- Mosser, D.M.; Hamidzadeh, K.; Goncalves, R. Macrophages and the Maintenance of Homeostasis. Cell. Mol. Immunol. 2021, 18, 579–587. [Google Scholar] [CrossRef]
- Shi, C.; Pamer, E.G. Monocyte Recruitment during Infection and Inflammation. Nat. Rev. Immunol. 2011, 11, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Sabir, F.; Farooq, R.K.; Asim.ur.Rehman; Ahmed, N. Monocyte as an Emerging Tool for Targeted Drug Delivery: A Review. Curr. Pharm. Des. 2019, 24, 5296–5312. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Kapate, N.; Shields, C.W.; Mitragotri, S. Drug Delivery to Macrophages: A Review of Targeting Drugs and Drug Carriers to Macrophages for Inflammatory Diseases. Adv. Drug. Deliv. Rev. 2020, 165–166, 15–40. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Guo, M.; Xu, J.; Wu, F.; Fan, J.; Huang, Q.; Yang, G.; Lv, Z.; Wang, X.; Jin, Y. Nanoparticles Targeting Macrophages as Potential Clinical Therapeutic Agents Against Cancer and Inflammation. Front. Immunol. 2019, 10, 1998. [Google Scholar] [CrossRef] [PubMed]
- Cai, D.; Gao, W.; Li, Z.; Zhang, Y.; Xiao, L.; Xiao, Y. Current Development of Nano-Drug Delivery to Target Macrophages. Biomedicines 2022, 10, 1203. [Google Scholar] [CrossRef]
- Swirski, F.K.; Nahrendorf, M. Cardioimmunology: The Immune System in Cardiac Homeostasis and Disease. Nat. Rev. Immunol. 2018, 18, 733–744. [Google Scholar] [CrossRef]
- Murphy, A.J.; Febbraio, M.A. Immune-Based Therapies in Cardiovascular and Metabolic Diseases: Past, Present and Future. Nat. Rev. Immunol. 2021, 21, 669–679. [Google Scholar] [CrossRef]
- Silveira Rossi, J.L.; Barbalho, S.M.; Reverete de Araujo, R.; Bechara, M.D.; Sloan, K.P.; Sloan, L.A. Metabolic Syndrome and Cardiovascular Diseases: Going beyond Traditional Risk Factors. Diabetes Metab. Res. Rev. 2022, 38, e3502. [Google Scholar] [CrossRef]
- Woollard, K.J.; Geissmann, F. Monocytes in Atherosclerosis: Subsets and Functions. Nat. Rev. Cardiol. 2010, 7, 77–86. [Google Scholar] [CrossRef]
- Kim, K.; Shim, D.; Lee, J.S.; Zaitsev, K.; Williams, J.W.; Kim, K.-W.; Jang, M.-Y.; Seok Jang, H.; Yun, T.J.; Lee, S.H.; et al. Transcriptome Analysis Reveals Nonfoamy Rather Than Foamy Plaque Macrophages Are Proinflammatory in Atherosclerotic Murine Models. Circ. Res. 2018, 123, 1127–1142. [Google Scholar] [CrossRef]
- Lin, P.; Ji, H.-H.; Li, Y.-J.; Guo, S.-D. Macrophage Plasticity and Atherosclerosis Therapy. Front. Mol. Biosci. 2021, 8, 679797. [Google Scholar] [CrossRef]
- Adamson, S.; Leitinger, N. Phenotypic Modulation of Macrophages in Response to Plaque Lipids. Curr. Opin. Lipidol. 2011, 22, 335–342. [Google Scholar] [CrossRef]
- Karlinsey, K.; Qu, L.; Matz, A.J.; Zhou, B. A Novel Strategy to Dissect Multifaceted Macrophage Function in Human Diseases. J. Leukoc. Biol. 2022, 112, 1535–1542. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, Y.; Liu, C.; Wu, X.; Wang, S.; Sui, W.; Zhang, Y.; Zhang, C.; Zhang, M. Heterogeneity of Macrophages in Atherosclerosis Revealed by Single-cell RNA Sequencing. FASEB J. 2023, 37, e22810. [Google Scholar] [CrossRef]
- Gisterå, A.; Hansson, G.K. The Immunology of Atherosclerosis. Nat. Rev. Nephrol. 2017, 13, 368–380. [Google Scholar] [CrossRef]
- Gialeli, C.; Shami, A.; Gonçalves, I. Extracellular Matrix: Paving the Way to the Newest Trends in Atherosclerosis. Curr. Opin. Lipidol. 2021, 32, 277–285. [Google Scholar] [CrossRef]
- Nasser, M.I.; Zhu, S.; Huang, H.; Zhao, M.; Wang, B.; Ping, H.; Geng, Q.; Zhu, P. Macrophages: First Guards in the Prevention of Cardiovascular Diseases. Life Sci. 2020, 250, 117559. [Google Scholar] [CrossRef]
- Komal, S.; Han, S.-N.; Cui, L.-G.; Zhai, M.-M.; Zhou, Y.-J.; Wang, P.; Shakeel, M.; Zhang, L.-R. Epigenetic Regulation of Macrophage Polarization in Cardiovascular Diseases. Pharmaceuticals 2023, 16, 141. [Google Scholar] [CrossRef]
- Khoury, M.K.; Yang, H.; Liu, B. Macrophage Biology in Cardiovascular Diseases. Arter. Thromb. Vasc. Biol. 2021, 41, e77–e81. [Google Scholar] [CrossRef]
- Gianazza, E.; Zoanni, B.; Mallia, A.; Brioschi, M.; Colombo, G.I.; Banfi, C. Proteomic Studies on ApoB-containing Lipoprotein in Cardiovascular Research: A Comprehensive Review. Mass Spectrom. Rev. 2021, 1–27. [Google Scholar] [CrossRef]
- Ben-Aicha, S.; Badimon, L.; Vilahur, G. Advances in HDL: Much More than Lipid Transporters. Int. J. Mol. Sci. 2020, 21, 732. [Google Scholar] [CrossRef] [PubMed]
- Kontush, A.; Lindahl, M.; Lhomme, M.; Calabresi, L.; Chapman, M.J.; Davidson, W.S. Structure of HDL: Particle subclasses and molecular components. Handb. Exp. Pharmacol. 2015, 224, 3–51. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Ramirez, D.; Herrington, W.G.; Alegre-Díaz, J.; Staplin, N.; Ramírez-Reyes, R.; Gnatiuc, L.F.; Hill, M.; Romer, F.; Trichia, E.; Bragg, F.; et al. Adiposity and NMR-Measured Lipid and Metabolic Biomarkers among 30,000 Mexican Adults. Commun. Med. 2022, 2, 143. [Google Scholar] [CrossRef] [PubMed]
- Streese, L.; Habisch, H.; Deiseroth, A.; Carrard, J.; Infanger, D.; Schmidt-Trucksäss, A.; Madl, T.; Hanssen, H. Lipoprotein Subclasses Independently Contribute to Subclinical Variance of Microvascular and Macrovascular Health. Molecules 2022, 27, 4760. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, H.; Vincent, V.; Sen, A.; Singh, A.; Roy, A. Changing Perspectives on HDL: From Simple Quantity Measurements to Functional Quality Assessment. J. Lipids 2021, 2021, 5585521. [Google Scholar] [CrossRef]
- Holzer, M.; Ljubojevic-Holzer, S.; Souza Junior, D.R.; Stadler, J.T.; Rani, A.; Scharnagl, H.; Ronsein, G.E.; Marsche, G. HDL Isolated by Immunoaffinity, Ultracentrifugation, or Precipitation Is Compositionally and Functionally Distinct. J. Lipid Res. 2022, 63, 100307. [Google Scholar] [CrossRef]
- Lorkowski, S.W.; Smith, J.D. HDL Is Not Dead Yet. Biomedicines 2022, 10, 128. [Google Scholar] [CrossRef]
- Vandendriessche, L. Changes in Free Cholesterol and Lecithine Content of Horse Serum Natuurwetenschap. Tijdschr 1944, 1944, 62–66. [Google Scholar]
- Rohatgi, A.; Westerterp, M.; von Eckardstein, A.; Remaley, A.; Rye, K.A. HDL in the 21st Century. Circulation 2021, 143, 2293–2309. [Google Scholar] [CrossRef]
- Michell, D.L.; Vickers, K.C. HDL and MicroRNA Therapeutics in Cardiovascular Disease. Pharmacol. Ther. 2016, 168, 43–52. [Google Scholar] [CrossRef]
- Eckardstein, A.V.; Kardassis, D. High Density Lipoproteins: From Biological Understanding to Clinical Exploitation; Cham, S.L., Ed.; Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2015; Volume 224. [Google Scholar]
- Ronsein, G.E.; Vaisar, T. Deepening Our Understanding of HDL Proteome. Expert Rev. Proteom. 2019, 16, 749–760. [Google Scholar] [CrossRef]
- Thomas, M.J.; Bhat, S.; Sorci-Thomas, M.G. Three-Dimensional Models of HDL ApoA-I: Implications for Its Assembly and Function. J. Lipid Res. 2008, 49, 1875–1883. [Google Scholar] [CrossRef]
- Schrijver, D.P.; de Dreu, A.; Hofstraat, S.R.J.; Kluza, E.; Zwolsman, R.; Deckers, J.; Anbergen, T.; de Bruin, K.; Trines, M.M.; Nugraha, E.G.; et al. Nanoengineering Apolipoprotein A1-Based Immunotherapeutics. Adv. Ther. 2021, 4, 2100083. [Google Scholar] [CrossRef]
- Silva, R.A.G.D.; Huang, R.; Morris, J.; Fang, J.; Gracheva, E.O.; Ren, G.; Kontush, A.; Jerome, W.G.; Rye, K.-A.; Davidson, W.S. Structure of Apolipoprotein A-I in Spherical High Density Lipoproteins of Different Sizes. Proc. Natl. Acad. Sci. USA 2008, 105, 12176–12181. [Google Scholar] [CrossRef]
- Iqbal, A.J.; Barrett, T.J.; Taylor, L.; McNeill, E.; Manmadhan, A.; Recio, C.; Carmineri, A.; Brodermann, M.H.; White, G.E.; Cooper, D.; et al. Acute Exposure to Apolipoprotein A1 Inhibits Macrophage Chemotaxis in Vitro and Monocyte Recruitment in Vivo. Elife 2016, 5, e15190. [Google Scholar] [CrossRef]
- Georgila; Vyrla; Drakos Apolipoprotein A-I (ApoA-I), Immunity, Inflammation and Cancer. Cancers 2019, 11, 1097. [CrossRef]
- Javaheri, A.; Rader, D.J. Apolipoprotein A-I and Cholesterol Efflux. Circ. Res. 2014, 114, 1681–1683. [Google Scholar] [CrossRef]
- Garbuzova-Davis, S.; Willing, A.E.; Borlongan, C.V. Apolipoprotein A1 Enhances Endothelial Cell Survival in an In Vitro Model of ALS. eNeuro 2022, 9, ENEURO.0140-22.2022. [Google Scholar] [CrossRef]
- Mahley, R.W.; Innerarity, T.L.; Rall, S.C.; Weisgraber, K.H. Plasma Lipoproteins: Apolipoprotein Structure and Function. J. Lipid Res. 1984, 25, 1277–1294. [Google Scholar] [CrossRef]
- van Linthout, S.; Spillmann, F.; Graiani, G.; Miteva, K.; Peng, J.; van Craeyveld, E.; Meloni, M.; Tölle, M.; Escher, F.; Subasigüller, A.; et al. Down-Regulation of Endothelial TLR4 Signalling after Apo A-I Gene Transfer Contributes to Improved Survival in an Experimental Model of Lipopolysaccharide-Induced Inflammation. J. Mol. Med. 2011, 89, 151–160. [Google Scholar] [CrossRef]
- Xu, X.; Song, Z.; Mao, B.; Xu, G. Apolipoprotein A1-Related Proteins and Reverse Cholesterol Transport in Antiatherosclerosis Therapy: Recent Progress and Future Perspectives. Cardiovasc. Ther. 2022, 2022, 4610834. [Google Scholar] [CrossRef]
- Getz, G.; Reardon, C. Apoprotein E and Reverse Cholesterol Transport. Int. J. Mol. Sci. 2018, 19, 3479. [Google Scholar] [CrossRef] [PubMed]
- Getz, G.S.; Reardon, C.A. Apoprotein E as a Lipid Transport and Signaling Protein in the Blood, Liver, and Artery Wall. J. Lipid Res. 2009, 50, S156–S161. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.H.; Reddick, R.L.; Piedrahita, J.A.; Maeda, N. Spontaneous Hypercholesterolemia and Arterial Lesions in Mice Lacking Apolipoprotein E. Science 1992, 258, 468–471. [Google Scholar] [CrossRef] [PubMed]
- Wilson, H.M.; Patel, J.C.; Russell, D.; Skinner, E.R. Alterations in the Concentration of an Apolipoprotein E-Containing Subfraction of Plasma High Density Lipoprotein in Coronary Heart Disease. Clin. Chim. Acta 1993, 220, 175–187. [Google Scholar] [CrossRef]
- Sacks, F.M.; Alaupovic, P.; Moye, L.A.; Cole, T.G.; Sussex, B.; Stampfer, M.J.; Pfeffer, M.A.; Braunwald, E. VLDL, Apolipoproteins B, CIII, and E, and Risk of Recurrent Coronary Events in the Cholesterol and Recurrent Events (CARE) Trial. Circulation 2000, 102, 1886–1892. [Google Scholar] [CrossRef]
- Wolska, A.; Reimund, M.; Remaley, A.T. Apolipoprotein C-II. Curr. Opin. Lipidol. 2020, 31, 147–153. [Google Scholar] [CrossRef]
- Kei, A.A.; Filippatos, T.D.; Tsimihodimos, V.; Elisaf, M.S. A Review of the Role of Apolipoprotein C-II in Lipoprotein Metabolism and Cardiovascular Disease. Metabolism 2012, 61, 906–921. [Google Scholar] [CrossRef]
- Tian, L.; Xu, Y.; Fu, M.; Jia, L.; Yang, Y. Influence of ApolipoproteinCII Concentrations on HDL Subclass Distribution. J. Atheroscler. Thromb. 2009, 16, 611–620. [Google Scholar] [CrossRef]
- Berberich, A.J.; Hegele, R.A. The Role of Genetic Testing in Dyslipidaemia. Pathology 2019, 51, 184–192. [Google Scholar] [CrossRef]
- Scheffer, P.G.; Teerlink, T.; Dekker, J.M.; Bos, G.; Nijpels, G.; Diamant, M.; Kostense, P.J.; Stehouwer, C.D.A.; Heine, R.J. Increased Plasma Apolipoprotein C-III Concentration Independently Predicts Cardiovascular Mortality: The Hoorn Study. Clin. Chem. 2008, 54, 1325–1330. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, A.B.; Frikke-Schmidt, R.; Nordestgaard, B.G.; Tybjærg-Hansen, A. Loss-of-Function Mutations in APOC3 and Risk of Ischemic Vascular Disease. N. Engl. J. Med. 2014, 371, 32–41. [Google Scholar] [CrossRef]
- Marsche, G.; Heine, G.H.; Stadler, J.T.; Holzer, M. Current Understanding of the Relationship of HDL Composition, Structure and Function to Their Cardioprotective Properties in Chronic Kidney Disease. Biomolecules 2020, 10, 1348. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-C.; Shao, B.; Kanter, J.E.; He, Y.; Vaisar, T.; Witztum, J.L.; Snell-Bergeon, J.; McInnes, G.; Bruse, S.; Gottesman, O.; et al. Apolipoprotein C3 Induces Inflammasome Activation Only in Its Delipidated Form. Nat. Immunol. 2023, 24, 408–411. [Google Scholar] [CrossRef]
- de la Parra Soto, L.G.; Gutiérrez-Uribe, J.A.; Sharma, A.; Ramírez-Jiménez, A.K. Is Apo-CIII the New Cardiovascular Target? An Analysis of Its Current Clinical and Dietetic Therapies. Nutr. Metab. Cardiovasc. Dis. 2022, 32, 295–308. [Google Scholar] [CrossRef]
- Jensen, M.K.; Rimm, E.B.; Furtado, J.D.; Sacks, F.M. Apolipoprotein C-III as a Potential Modulator of the Association Between HDL-Cholesterol and Incident Coronary Heart Disease. J. Am. Heart Assoc. 2012, 1, e000232. [Google Scholar] [CrossRef]
- Morton, A.M.; Koch, M.; Mendivil, C.O.; Furtado, J.D.; Tjønneland, A.; Overvad, K.; Wang, L.; Jensen, M.K.; Sacks, F.M. Apolipoproteins E and CIII Interact to Regulate HDL Metabolism and Coronary Heart Disease Risk. JCI Insight 2018, 3, e98045. [Google Scholar] [CrossRef]
- Ahmed, Z. Paraoxonase-1 Reduces Monocyte Chemotaxis and Adhesion to Endothelial Cells Due to Oxidation of Palmitoyl, Linoleoyl Glycerophosphorylcholine. Cardiovasc. Res. 2003, 57, 225–231. [Google Scholar] [CrossRef]
- Ng, C.J.; Bourquard, N.; Grijalva, V.; Hama, S.; Shih, D.M.; Navab, M.; Fogelman, A.M.; Lusis, A.J.; Young, S.; Reddy, S.T. Paraoxonase-2 Deficiency Aggravates Atherosclerosis in Mice Despite Lower Apolipoprotein-B-Containing Lipoproteins. J. Biol. Chem. 2006, 281, 29491–29500. [Google Scholar] [CrossRef]
- Rosenblat, M.; Gaidukov, L.; Khersonsky, O.; Vaya, J.; Oren, R.; Tawfik, D.S.; Aviram, M. The Catalytic Histidine Dyad of High Density Lipoprotein-Associated Serum Paraoxonase-1 (PON1) Is Essential for PON1-Mediated Inhibition of Low Density Lipoprotein Oxidation and Stimulation of Macrophage Cholesterol Efflux. J. Biol. Chem. 2006, 281, 7657–7665. [Google Scholar] [CrossRef] [PubMed]
- Toma, L.; Barbălată, T.; Sanda, G.M.; Niculescu, L.S.; Sima, A.V.; Stancu, C.S. CRISPR/DCas9 Transcriptional Activation of Endogenous Apolipoprotein AI and Paraoxonase 1 in Enterocytes Alleviates Endothelial Cell Dysfunction. Biomolecules 2021, 11, 1769. [Google Scholar] [CrossRef]
- James, R.W.; Deakin, S.P. The Contribution of High Density Lipoprotein Apolipoproteins and Derivatives to Serum Paraoxonase-1 Activity and Function. In Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2010; pp. 173–181. [Google Scholar]
- James, R.W.; Deakin, S.P. The Importance of High-Density Lipoproteins for Paraoxonase-1 Secretion, Stability, and Activity. Free Radic. Biol. Med. 2004, 37, 1986–1994. [Google Scholar] [CrossRef]
- Hine, D.; Mackness, B.; Mackness, M. Coincubation of PON1, APO A1, and LCAT Increases the Time HDL Is Able to Prevent LDL Oxidation. IUBMB Life 2012, 64, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Mackness, M.; Mackness, B. Paraoxonase 1 and Atherosclerosis: Is the Gene or the Protein More Important? Free Radic. Biol. Med. 2004, 37, 1317–1323. [Google Scholar] [CrossRef]
- Dubois-Deruy, E.; Peugnet, V.; Turkieh, A.; Pinet, F. Oxidative Stress in Cardiovascular Diseases. Antioxidants 2020, 9, 864. [Google Scholar] [CrossRef]
- Lubrano, V. Enzymatic Antioxidant System in Vascular Inflammation and Coronary Artery Disease. World J. Exp. Med. 2015, 5, 218. [Google Scholar] [CrossRef] [PubMed]
- Tall, A.R.; Rader, D.J. Trials and Tribulations of CETP Inhibitors. Circ. Res. 2018, 122, 106–112. [Google Scholar] [CrossRef]
- Pussinen, P.J.; Jauhiainen, M.; Metso, J.; Pyle, L.E.; Marcel, Y.L.; Fidge, N.H.; Ehnholm, C. Binding of Phospholipid Transfer Protein (PLTP) to Apolipoproteins A-I and A-II: Location of a PLTP Binding Domain in the Amino Terminal Region of ApoA-I. J. Lipid Res. 1998, 39, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.-H. Human Serum Amyloid a Impaired Structural Stability of High-Density Lipoproteins (HDL) and Apolipoprotein (Apo) A-I and Exacerbated Glycation Susceptibility of ApoA-I and HDL. Molecules 2022, 27, 4255. [Google Scholar] [CrossRef]
- Nie, J.; Yang, J.; Wei, Y.; Wei, X. The Role of Oxidized Phospholipids in the Development of Disease. Mol. Asp. Med. 2020, 76, 100909. [Google Scholar] [CrossRef] [PubMed]
- Bretscher, P.; Egger, J.; Shamshiev, A.; Trötzmüller, M.; Köfeler, H.; Carreira, E.M.; Kopf, M.; Freigang, S. Phospholipid Oxidation Generates Potent Anti-inflammatory Lipid Mediators That Mimic Structurally Related Pro-resolving Eicosanoids by Activating Nrf2. EMBO Mol. Med. 2015, 7, 593–607. [Google Scholar] [CrossRef] [PubMed]
- Knuplez, E.; Marsche, G. An Updated Review of Pro- and Anti-Inflammatory Properties of Plasma Lysophosphatidylcholines in the Vascular System. Int. J. Mol. Sci. 2020, 21, 4501. [Google Scholar] [CrossRef]
- Knuplez, E.; Curcic, S.; Theiler, A.; Bärnthaler, T.; Trakaki, A.; Trieb, M.; Holzer, M.; Heinemann, A.; Zimmermann, R.; Sturm, E.M.; et al. Lysophosphatidylcholines Inhibit Human Eosinophil Activation and Suppress Eosinophil Migration in Vivo. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 1865, 158686. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Welch, E.J.; Gao, X.-P.; Malik, A.B.; Ye, R.D. Lysophosphatidylcholine Modulates Neutrophil Oxidant Production through Elevation of Cyclic AMP. J. Immunol. 2005, 174, 2981–2989. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.; Frej, C.; Holmér, A.; Guo, L.J.; Tran, S.; Dahlbäck, B. High-Density Lipoprotein–Associated Apolipoprotein M Limits Endothelial Inflammation by Delivering Sphingosine-1-Phosphate to the Sphingosine-1-Phosphate Receptor 1. Arter. Thromb. Vasc. Biol. 2017, 37, 118–129. [Google Scholar] [CrossRef]
- Levkau, B. HDL-S1P: Cardiovascular Functions, Disease-Associated Alterations, and Therapeutic Applications. Front. Pharm. 2015, 6, 243. [Google Scholar] [CrossRef]
- Kontush, A.; Lhomme, M.; Chapman, M.J. Unraveling the Complexities of the HDL Lipidome. J. Lipid Res. 2013, 54, 2950–2963. [Google Scholar] [CrossRef]
- Cui, H.; Lv, K.; Yang, N. HDL and MicroRNAs. Adv. Exp. Med. Biol. 2022, 1377, 153–161. [Google Scholar]
- Morrison, K.R.; Solly, E.L.; Shemesh, T.; Psaltis, P.J.; Nicholls, S.J.; Brown, A.; Bursill, C.A.; Tan, J.T.M. Elevated HDL-Bound MiR-181c-5p Level Is Associated with Diabetic Vascular Complications in Australian Aboriginal People. Diabetologia 2021, 64, 1402–1411. [Google Scholar] [CrossRef]
- Catapano, A.L.; Pirillo, A.; Bonacina, F.; Norata, G.D. HDL in Innate and Adaptive Immunity. Cardiovasc. Res. 2014, 103, 372–383. [Google Scholar] [CrossRef]
- Simons, K.; Toomre, D. Lipid Rafts and Signal Transduction. Nat. Rev. Mol. Cell Biol. 2000, 1, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.J.; Woollard, K.J.; Hoang, A.; Mukhamedova, N.; Stirzaker, R.A.; McCormick, S.P.A.; Remaley, A.T.; Sviridov, D.; Chin-Dusting, J. High-Density Lipoprotein Reduces the Human Monocyte Inflammatory Response. Arter. Thromb. Vasc. Biol. 2008, 28, 2071–2077. [Google Scholar] [CrossRef]
- Diederich, W.; Orsó, E.; Drobnik, W.; Schmitz, G. Apolipoprotein AI and HDL3 Inhibit Spreading of Primary Human Monocytes through a Mechanism That Involves Cholesterol Depletion and Regulation of CDC42. Atherosclerosis 2001, 159, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Barrett, T.J.; Distel, E.; Murphy, A.J.; Hu, J.; Garshick, M.S.; Ogando, Y.; Liu, J.; Vaisar, T.; Heinecke, J.W.; Berger, J.S.; et al. Apolipoprotein AI) Promotes Atherosclerosis Regression in Diabetic Mice by Suppressing Myelopoiesis and Plaque Inflammation. Circulation 2019, 140, 1170–1184. [Google Scholar] [CrossRef] [PubMed]
- Feig, J.E.; Rong, J.X.; Shamir, R.; Sanson, M.; Vengrenyuk, Y.; Liu, J.; Rayner, K.; Moore, K.; Garabedian, M.; Fisher, E.A. HDL Promotes Rapid Atherosclerosis Regression in Mice and Alters Inflammatory Properties of Plaque Monocyte-Derived Cells. Proc. Natl. Acad. Sci. USA 2011, 108, 7166–7171. [Google Scholar] [CrossRef]
- De Nardo, D.; Labzin, L.I.; Kono, H.; Seki, R.; Schmidt, S.V.; Beyer, M.; Xu, D.; Zimmer, S.; Lahrmann, C.; Schildberg, F.A.; et al. High-Density Lipoprotein Mediates Anti-Inflammatory Reprogramming of Macrophages via the Transcriptional Regulator ATF3. Nat. Immunol. 2014, 15, 152–160. [Google Scholar] [CrossRef]
- Suzuki, M.; Pritchard, D.K.; Becker, L.; Hoofnagle, A.N.; Tanimura, N.; Bammler, T.K.; Beyer, R.P.; Bumgarner, R.; Vaisar, T.; de Beer, M.C.; et al. High-Density Lipoprotein Suppresses the Type I Interferon Response, a Family of Potent Antiviral Immunoregulators, in Macrophages Challenged With Lipopolysaccharide. Circulation 2010, 122, 1919–1927. [Google Scholar] [CrossRef]
- Daub, S.; Lutgens, E.; Münzel, T.; Daiber, A. CD40/CD40L and Related Signaling Pathways in Cardiovascular Health and Disease—The Pros and Cons for Cardioprotection. Int. J. Mol. Sci. 2020, 21, 8533. [Google Scholar] [CrossRef]
- Yin, K.; Chen, W.-J.; Zhou, Z.-G.; Zhao, G.-J.; Lv, Y.-C.; Ouyang, X.-P.; Yu, X.-H.; Fu, Y.; Jiang, Z.-S.; Tang, C.-K. Apolipoprotein A-I Inhibits CD40 Proinflammatory Signaling via ATP-Binding Cassette Transporter A1-Mediated Modulation of Lipid Raft in Macrophages. J. Atheroscler. Thromb. 2012, 19, 823–836. [Google Scholar] [CrossRef]
- Kishore, R.; Verma, S.K. Roles of STATs Signaling in Cardiovascular Diseases. JAK-STAT 2012, 1, 118–124. [Google Scholar] [CrossRef]
- Zhao, G.-J.; Yin, K.; Fu, Y.; Tang, C.-K. The Interaction of ApoA-I and ABCA1 Triggers Signal Transduction Pathways to Mediate Efflux of Cellular Lipids. Mol. Med. 2012, 18, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Liu, Y.; Kessler, P.S.; Vaughan, A.M.; Oram, J.F. The Macrophage Cholesterol Exporter ABCA1 Functions as an Anti-Inflammatory Receptor. J. Biol. Chem. 2009, 284, 32336–32343. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Houston, B.A.; Storey, C.; LeBoeuf, R.C. Both STAT3 Activation and Cholesterol Efflux Contribute to the Anti-Inflammatory Effect of ApoA-I/ABCA1 Interaction in Macrophages. J. Lipid Res. 2016, 57, 848–857. [Google Scholar] [CrossRef]
- Murphy, A.J.; Westerterp, M.; Yvan-Charvet, L.; Tall, A.R. Anti-Atherogenic Mechanisms of High Density Lipoprotein: Effects on Myeloid Cells. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2012, 1821, 513–521. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Gelissen, I.C.; Ammit, A.J. Regulation of ATP Binding Cassette Transporter A1 (ABCA1) Expression: Cholesterol-Dependent and—Independent Signaling Pathways with Relevance to Inflammatory Lung Disease. Respir. Res. 2020, 21, 250. [Google Scholar] [CrossRef]
- White, C.R.; Anantharamaiah, G.M. Cholesterol Reduction and Macrophage Function. Curr. Opin. Lipidol. 2017, 28, 397–402. [Google Scholar] [CrossRef]
- Rozenberg, O.; Shiner, M.; Aviram, M.; Hayek, T. Paraoxonase 1 (PON1) Attenuates Diabetes Development in Mice through Its Antioxidative Properties. Free Radic. Biol. Med. 2008, 44, 1951–1959. [Google Scholar] [CrossRef]
- Rozenberg, O. Paraoxonase (PON1) Deficiency Is Associated with Increased Macrophage Oxidative Stress: Studies in PON1-Knockout Mice. Free Radic. Biol. Med. 2003, 34, 774–784. [Google Scholar] [CrossRef]
- Rozenberg, O.; Shih, D.M.; Aviram, M. Human Serum Paraoxonase 1 Decreases Macrophage Cholesterol Biosynthesis. Arter. Thromb. Vasc. Biol. 2003, 23, 461–467. [Google Scholar] [CrossRef]
- Ng, C.J.; Bourquard, N.; Hama, S.Y.; Shih, D.; Grijalva, V.R.; Navab, M.; Fogelman, A.M.; Reddy, S.T. Adenovirus-Mediated Expression of Human Paraoxonase 3 Protects Against the Progression of Atherosclerosis in Apolipoprotein E–Deficient Mice. Arter. Thromb. Vasc. Biol. 2007, 27, 1368–1374. [Google Scholar] [CrossRef]
- Theilmeier, G.; Schmidt, C.; Herrmann, J.; Keul, P.; Schäfers, M.; Herrgott, I.; Mersmann, J.; Larmann, J.; Hermann, S.; Stypmann, J.; et al. High-Density Lipoproteins and Their Constituent, Sphingosine-1-Phosphate, Directly Protect the Heart Against Ischemia/Reperfusion Injury In Vivo via the S1P 3 Lysophospholipid Receptor. Circulation 2006, 114, 1403–1409. [Google Scholar] [CrossRef]
- Orekhov, A.N.; Sobenin, I.A. Modified and Dysfunctional Lipoproteins in Atherosclerosis: Effectors or Biomarkers? Curr. Med. Chem. 2019, 26, 1512–1524. [Google Scholar] [CrossRef]
- Márquez, A.B.; Nazir, S.; van der Vorst, E.P.C. High-Density Lipoprotein Modifications: A Pathological Consequence or Cause of Disease Progression? Biomedicines 2020, 8, 549. [Google Scholar] [CrossRef]
- Vyletelová, V.; Nováková, M.; Pašková, Ľ. Alterations of HDL’s to PiHDL’s Proteome in Patients with Chronic Inflammatory Diseases, and HDL-Targeted Therapies. Pharmaceuticals 2022, 15, 1278. [Google Scholar] [CrossRef]
- Sirtori, C.R.; Corsini, A.; Ruscica, M. The Role of High-Density Lipoprotein Cholesterol in 2022. Curr. Atheroscler. Rep. 2022, 24, 365–377. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Brewer, H.B.; Ansell, B.J.; Barter, P.; Chapman, M.J.; Heinecke, J.W.; Kontush, A.; Tall, A.R.; Webb, N.R. Dysfunctional HDL and Atherosclerotic Cardiovascular Disease. Nat. Rev. Cardiol. 2016, 13, 48–60. [Google Scholar] [CrossRef]
- Khera, A.V.; Cuchel, M.; de la Llera-Moya, M.; Rodrigues, A.; Burke, M.F.; Jafri, K.; French, B.C.; Phillips, J.A.; Mucksavage, M.L.; Wilensky, R.L.; et al. Cholesterol Efflux Capacity, High-Density Lipoprotein Function, and Atherosclerosis. N. Engl. J. Med. 2011, 364, 127–135. [Google Scholar] [CrossRef]
- Saleheen, D.; Scott, R.; Javad, S.; Zhao, W.; Rodrigues, A.; Picataggi, A.; Lukmanova, D.; Mucksavage, M.L.; Luben, R.; Billheimer, J.; et al. Association of HDL Cholesterol Efflux Capacity with Incident Coronary Heart Disease Events: A Prospective Case-Control Study. Lancet Diabetes Endocrinol. 2015, 3, 507–513. [Google Scholar] [CrossRef]
- Ashby, D.T.; Rye, K.-A.; Clay, M.A.; Vadas, M.A.; Gamble, J.R.; Barter, P.J. Factors Influencing the Ability of HDL to Inhibit Expression of Vascular Cell Adhesion Molecule-1 in Endothelial Cells. Arter. Thromb. Vasc. Biol. 1998, 18, 1450–1455. [Google Scholar] [CrossRef]
- Kontush, A.; Chapman, M.J. Antiatherogenic Small, Dense HDL—Guardian Angel of the Arterial Wall? Nat. Clin. Pract. Cardiovasc. Med. 2006, 3, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Du, X.-M.; Kim, M.-J.; Hou, L.; le Goff, W.; Chapman, M.J.; van Eck, M.; Curtiss, L.K.; Burnett, J.R.; Cartland, S.P.; Quinn, C.M.; et al. HDL Particle Size Is a Critical Determinant of ABCA1-Mediated Macrophage Cellular Cholesterol Export. Circ. Res. 2015, 116, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Asztalos, B.F.; Cupples, L.A.; Demissie, S.; Horvath, K.V.; Cox, C.E.; Batista, M.C.; Schaefer, E.J. High-Density Lipoprotein Subpopulation Profile and Coronary Heart Disease Prevalence in Male Participants of the Framingham Offspring Study. Arter. Thromb. Vasc. Biol. 2004, 24, 2181–2187. [Google Scholar] [CrossRef]
- Girona, J.; Amigó, N.; Ibarretxe, D.; Plana, N.; Rodríguez-Borjabad, C.; Heras, M.; Ferré, R.; Gil, M.; Correig, X.; Masana, L. Masana HDL Triglycerides: A New Marker of Metabolic and Cardiovascular Risk. Int. J. Mol. Sci. 2019, 20, 3151. [Google Scholar] [CrossRef] [PubMed]
- Maiocchi, S.L.; Ku, J.; Thai, T.; Chan, E.; Rees, M.D.; Thomas, S.R. Myeloperoxidase: A Versatile Mediator of Endothelial Dysfunction and Therapeutic Target during Cardiovascular Disease. Pharmacol. Ther. 2021, 221, 107711. [Google Scholar] [CrossRef]
- Kattoor, A.J.; Pothineni, N.V.K.; Palagiri, D.; Mehta, J.L. Oxidative Stress in Atherosclerosis. Curr. Atheroscler. Rep. 2017, 19, 42. [Google Scholar] [CrossRef]
- Huang, Y.; DiDonato, J.A.; Levison, B.S.; Schmitt, D.; Li, L.; Wu, Y.; Buffa, J.; Kim, T.; Gerstenecker, G.S.; Gu, X.; et al. An Abundant Dysfunctional Apolipoprotein A1 in Human Atheroma. Nat. Med. 2014, 20, 193–203. [Google Scholar] [CrossRef]
- Zheng, L.; Nukuna, B.; Brennan, M.-L.; Sun, M.; Goormastic, M.; Settle, M.; Schmitt, D.; Fu, X.; Thomson, L.; Fox, P.L.; et al. Apolipoprotein A-I Is a Selective Target for Myeloperoxidase-Catalyzed Oxidation and Functional Impairment in Subjects with Cardiovascular Disease. J. Clin. Investig. 2004, 114, 529–541. [Google Scholar] [CrossRef]
- Holzer, M.; Zangger, K.; El-Gamal, D.; Binder, V.; Curcic, S.; Konya, V.; Schuligoi, R.; Heinemann, A.; Marsche, G. Myeloperoxidase-Derived Chlorinating Species Induce Protein Carbamylation Through Decomposition of Thiocyanate and Urea: Novel Pathways Generating Dysfunctional High-Density Lipoprotein. Antioxid. Redox Signal. 2012, 17, 1043–1052. [Google Scholar] [CrossRef]
- Holzer, M.; Gauster, M.; Pfeifer, T.; Wadsack, C.; Fauler, G.; Stiegler, P.; Koefeler, H.; Beubler, E.; Schuligoi, R.; Heinemann, A.; et al. Protein Carbamylation Renders High-Density Lipoprotein Dysfunctional. Antioxid. Redox Signal. 2011, 14, 2337–2346. [Google Scholar] [CrossRef]
- Bergt, C.; Pennathur, S.; Fu, X.; Byun, J.; O’Brien, K.; McDonald, T.O.; Singh, P.; Anantharamaiah, G.M.; Chait, A.; Brunzell, J.; et al. The Myeloperoxidase Product Hypochlorous Acid Oxidizes HDL in the Human Artery Wall and Impairs ABCA1-Dependent Cholesterol Transport. Proc. Natl. Acad. Sci. USA 2004, 101, 13032–13037. [Google Scholar] [CrossRef]
- DiDonato, J.A.; Huang, Y.; Aulak, K.S.; Even-Or, O.; Gerstenecker, G.; Gogonea, V.; Wu, Y.; Fox, P.L.; Tang, W.H.W.; Plow, E.F.; et al. Function and Distribution of Apolipoprotein A1 in the Artery Wall Are Markedly Distinct From Those in Plasma. Circulation 2013, 128, 1644–1655. [Google Scholar] [CrossRef] [PubMed]
- Marsche, G.; Stadler, J.T.; Kargl, J.; Holzer, M. Understanding Myeloperoxidase-Induced Damage to HDL Structure and Function in the Vessel Wall: Implications for HDL-Based Therapies. Antioxidants 2022, 11, 556. [Google Scholar] [CrossRef]
- Besler, C.; Heinrich, K.; Rohrer, L.; Doerries, C.; Riwanto, M.; Shih, D.M.; Chroni, A.; Yonekawa, K.; Stein, S.; Schaefer, N.; et al. Mechanisms Underlying Adverse Effects of HDL on ENOS-Activating Pathways in Patients with Coronary Artery Disease. J. Clin. Investig. 2011, 121, 2693–2708. [Google Scholar] [CrossRef]
- Shih, C.-M.; Lin, F.-Y.; Yeh, J.-S.; Lin, Y.-W.; Loh, S.-H.; Tsao, N.-W.; Nakagami, H.; Morishita, R.; Sawamura, T.; Li, C.-Y.; et al. Dysfunctional High Density Lipoprotein Failed to Rescue the Function of Oxidized Low Density Lipoprotein-Treated Endothelial Progenitor Cells: A Novel Index for the Prediction of HDL Functionality. Transl. Res. 2019, 205, 17–32. [Google Scholar] [CrossRef]
- Sorrentino, S.A.; Besler, C.; Rohrer, L.; Meyer, M.; Heinrich, K.; Bahlmann, F.H.; Mueller, M.; Horváth, T.; Doerries, C.; Heinemann, M.; et al. Endothelial-Vasoprotective Effects of High-Density Lipoprotein Are Impaired in Patients With Type 2 Diabetes Mellitus but Are Improved After Extended-Release Niacin Therapy. Circulation 2010, 121, 110–122. [Google Scholar] [CrossRef]
- Ben-Aicha, S.; Casaní, L.; Muñoz-García, N.; Joan-Babot, O.; Peña, E.; Aržanauskaitė, M.; Gutierrez, M.; Mendieta, G.; Padró, T.; Badimon, L.; et al. HDL (High-Density Lipoprotein) Remodeling and Magnetic Resonance Imaging–Assessed Atherosclerotic Plaque Burden. Arter. Thromb. Vasc. Biol. 2020, 40, 2481–2493. [Google Scholar] [CrossRef]
- Besler, C.; Lüscher, T.F.; Landmesser, U. Molecular Mechanisms of Vascular Effects of High-density Lipoprotein: Alterations in Cardiovascular Disease. EMBO Mol. Med. 2012, 4, 251–268. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, S.; Gao, Y.; Tong, W.; Sun, S. High-Density Lipoprotein Subfractions Remodeling: A Critical Process for the Treatment of Atherosclerotic Cardiovascular Diseases. Angiology 2023, 000331972311574. [Google Scholar] [CrossRef]
- Cho, K.-H. The Current Status of Research on High-Density Lipoproteins (HDL): A Paradigm Shift from HDL Quantity to HDL Quality and HDL Functionality. Int. J. Mol. Sci. 2022, 23, 3967. [Google Scholar] [CrossRef]
- Stadler, J.T.; Marsche, G. Dietary Strategies to Improve Cardiovascular Health: Focus on Increasing High-Density Lipoprotein Functionality. Front. Nutr. 2021, 8, 761170. [Google Scholar] [CrossRef]
- Segrest, J.P.; De Loof, H.; Dohlman, J.G.; Brouillette, C.G.; Anantharamaiah, G.M. Amphipathic Helix Motif: Classes and Properties. Proteins Struct. Funct. Genet. 1990, 8, 103–117. [Google Scholar] [CrossRef] [PubMed]
- Pourmousa, M.; Pastor, R.W. Molecular Dynamics Simulations of Lipid Nanodiscs. Biochim. Biophys. Acta (BBA) Biomembr. 2018, 1860, 2094–2107. [Google Scholar] [CrossRef]
- Kornmueller; Vidakovic; Prassl Artificial High Density Lipoprotein Nanoparticles in Cardiovascular Research. Molecules 2019, 24, 2829. [CrossRef] [PubMed]
- Bhale, A.S.; Venkataraman, K. Leveraging Knowledge of HDLs Major Protein ApoA1: Structure, Function, Mutations, and Potential Therapeutics. Biomed. Pharmacother. 2022, 154, 113634. [Google Scholar] [CrossRef] [PubMed]
- Melchior, J.T.; Walker, R.G.; Cooke, A.L.; Morris, J.; Castleberry, M.; Thompson, T.B.; Jones, M.K.; Song, H.D.; Rye, K.-A.; Oda, M.N.; et al. A Consensus Model of Human Apolipoprotein A-I in Its Monomeric and Lipid-Free State. Nat. Struct. Mol. Biol. 2017, 24, 1093–1099. [Google Scholar] [CrossRef]
- Anantharamaiah, G.M.; Jones, J.L.; Brouillette, C.G.; Schmidt, C.F.; Chung, B.H.; Hughes, T.A.; Bhown, A.S.; Segrest, J.P. Studies of Synthetic Peptide Analogs of the Amphipathic Helix. Structure of Complexes with Dimyristoyl Phosphatidylcholine. J. Biol. Chem. 1985, 260, 10248–10255. [Google Scholar] [CrossRef]
- Miyazaki, M.; Tajima, Y.; Handa, T.; Nakano, M. Static and Dynamic Characterization of Nanodiscs with Apolipoprotein A-I and Its Model Peptide. J. Phys. Chem. B 2010, 114, 12376–12382. [Google Scholar] [CrossRef]
- Xie, Q.; Li, F.; Zhao, S.-P. Ac-HE-18A-NH2, a Novel Dual-Domain Apolipoprotein Mimetic Peptide, Inhibits Apoptosis in Macrophages by Promoting Cholesterol Efflux. Mol. Med. Rep. 2014, 9, 1851–1856. [Google Scholar] [CrossRef]
- Datta, G.; White, C.R.; Dashti, N.; Chaddha, M.; Palgunachari, M.N.; Gupta, H.; Handattu, S.P.; Garber, D.W.; Anantharamaiah, G.M. Anti-Inflammatory and Recycling Properties of an Apolipoprotein Mimetic Peptide, Ac-HE18A-NH2. Atherosclerosis 2010, 208, 134–141. [Google Scholar] [CrossRef]
- Larsen, A.N.; Sørensen, K.K.; Johansen, N.T.; Martel, A.; Kirkensgaard, J.J.K.; Jensen, K.J.; Arleth, L.; Midtgaard, S.R. Dimeric Peptides with Three Different Linkers Self-Assemble with Phospholipids to Form Peptide Nanodiscs That Stabilize Membrane Proteins. Soft Matter 2016, 12, 5937–5949. [Google Scholar] [CrossRef]
- Osei-Hwedieh, D.O.; Amar, M.; Sviridov, D.; Remaley, A.T. Apolipoprotein Mimetic Peptides: Mechanisms of Action as Anti-Atherogenic Agents. Pharmacol. Ther. 2011, 130, 83–91. [Google Scholar] [CrossRef]
- Wolska, A.; Reimund, M.; Sviridov, D.O.; Amar, M.J.; Remaley, A.T. Apolipoprotein Mimetic Peptides: Potential New Therapies for Cardiovascular Diseases. Cells 2021, 10, 597. [Google Scholar] [CrossRef]
- Sethi, A.A.; Stonik, J.A.; Thomas, F.; Demosky, S.J.; Amar, M.; Neufeld, E.; Brewer, H.B.; Davidson, W.S.; D’Souza, W.; Sviridov, D.; et al. Asymmetry in the Lipid Affinity of Bihelical Amphipathic Peptides. J. Biol. Chem. 2008, 283, 32273–32282. [Google Scholar] [CrossRef]
- Sviridov, D.O.; Andrianov, A.M.; Anishchenko, I.V.; Stonik, J.A.; Amar, M.J.A.; Turner, S.; Remaley, A.T. Hydrophobic Amino Acids in the Hinge Region of the 5A Apolipoprotein Mimetic Peptide Are Essential for Promoting Cholesterol Efflux by the ABCA1 Transporter. J. Pharmacol. Exp. Ther. 2013, 344, 50–58. [Google Scholar] [CrossRef]
- Di Bartolo, B.A.; Nicholls, S.J.; Bao, S.; Rye, K.-A.; Heather, A.K.; Barter, P.J.; Bursill, C. The Apolipoprotein A-I Mimetic Peptide ETC-642 Exhibits Anti-Inflammatory Properties That Are Comparable to High Density Lipoproteins. Atherosclerosis 2011, 217, 395–400. [Google Scholar] [CrossRef]
- Suematsu, Y.; Kawachi, E.; Idemoto, Y.; Matsuo, Y.; Kuwano, T.; Kitajima, K.; Imaizumi, S.; Kawamura, A.; Saku, K.; Uehara, Y.; et al. Anti-Atherosclerotic Effects of an Improved Apolipoprotein A-I Mimetic Peptide. Int. J. Cardiol. 2019, 297, 111–117. [Google Scholar] [CrossRef]
- Uehara, Y.; Ando, S.; Yahiro, E.; Oniki, K.; Ayaori, M.; Abe, S.; Kawachi, E.; Zhang, B.; Shioi, S.; Tanigawa, H.; et al. FAMP, a Novel ApoA-I Mimetic Peptide, Suppresses Aortic Plaque Formation Through Promotion of Biological HDL Function in ApoE-Deficient Mice. J. Am. Heart Assoc. 2013, 2, e000048. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Li, S.; Shi, M.; Li, Z.; Wang, Y.; Li, J.; Li, Q.; Klausen, L.H.; Li, A.; Zhao, H.; et al. Peptide-Based High-Density Lipoprotein Promotes Adipose Tissue Browning and Restrains Development of Atherosclerosis and Type 2 Diabetes. Nano Today 2021, 36, 101054. [Google Scholar] [CrossRef]
- White, C.R.; Goldberg, D.I.; Anantharamaiah, G.M. Recent Developments in Modulating Atherogenic Lipoproteins. Curr. Opin. Lipidol. 2015, 26, 369–375. [Google Scholar] [CrossRef]
- Anantharamaiah, G.M.; Garber, D.W.; Roger White, C. Apolipoprotein Mimetic Peptides as Modulators of Lipoprotein Function. Protein Pept. Lett. 2016, 23, 1024–1031. [Google Scholar] [CrossRef]
- Lin, C.Y.; Duan, H.; Mazzone, T. Apolipoprotein E-Dependent Cholesterol Efflux from Macrophages: Kinetic Study and Divergent Mechanisms for Endogenous versus Exogenous Apolipoprotein E. J. Lipid Res. 1999, 40, 1618–1627. [Google Scholar] [CrossRef] [PubMed]
- VanDusen, K.W.; Eleswarpu, S.; Moretti, E.W.; Devinney, M.J.; Crabtree, D.M.; Laskowitz, D.T.; Woldorff, M.G.; Roberts, K.C.; Whittle, J.; Browndyke, J.N.; et al. The MARBLE Study Protocol: Modulating ApoE Signaling to Reduce Brain Inflammation, DeLirium, and PostopErative Cognitive Dysfunction. J. Alzheimer’s Dis. 2020, 75, 1319–1328. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, B.G. Triglyceride-Rich Lipoproteins and Atherosclerotic Cardiovascular Disease. Circ. Res. 2016, 118, 547–563. [Google Scholar] [CrossRef]
- Wolska, A.; Lo, L.; Sviridov, D.O.; Pourmousa, M.; Pryor, M.; Ghosh, S.S.; Kakkar, R.; Davidson, M.; Wilson, S.; Pastor, R.W.; et al. A Dual Apolipoprotein C-II Mimetic–Apolipoprotein C-III Antagonist Peptide Lowers Plasma Triglycerides. Sci. Transl. Med. 2020, 12, eaaw7905. [Google Scholar] [CrossRef]
- Bricarello, D.A.; Smilowitz, J.T.; Zivkovic, A.M.; German, J.B.; Parikh, A.N. Reconstituted Lipoprotein: A Versatile Class of Biologically-Inspired Nanostructures. ACS Nano 2011, 5, 42–57. [Google Scholar] [CrossRef]
- Schwendeman, A.; Sviridov, D.O.; Yuan, W.; Guo, Y.; Morin, E.E.; Yuan, Y.; Stonik, J.; Freeman, L.; Ossoli, A.; Thacker, S.; et al. The Effect of Phospholipid Composition of Reconstituted HDL on Its Cholesterol Efflux and Anti-Inflammatory Properties. J. Lipid Res. 2015, 56, 1727–1737. [Google Scholar] [CrossRef]
- Patel, H.; Ding, B.; Ernst, K.; Shen, L.; Yuan, W.; Tang, J.; Drake, L.R.; Kang, J.; Li, Y.; Chen, Z.; et al. Characterization of Apolipoprotein A-I Peptide Phospholipid Interaction and Its Effect on HDL Nanodisc Assembly. Int. J. Nanomed. 2019, 14, 3069–3086. [Google Scholar] [CrossRef] [PubMed]
- Fawaz, M.V.; Kim, S.Y.; Li, D.; Ming, R.; Xia, Z.; Olsen, K.; Pogozheva, I.D.; Tesmer, J.J.G.; Schwendeman, A. Phospholipid Component Defines Pharmacokinetic and Pharmacodynamic Properties of Synthetic High-Density Lipoproteins. J. Pharmacol. Exp. Ther. 2020, 372, 193–204. [Google Scholar] [CrossRef]
- Darabi, M.; Lhomme, M.; Dahik, V.D.; Guillas, I.; Frisdal, E.; Tubeuf, E.; Poupel, L.; Patel, M.; Gautier, E.L.; Huby, T.; et al. Phosphatidylserine Enhances Anti-inflammatory Effects of Reconstituted HDL in Macrophages via Distinct Intracellular Pathways. FASEB J. 2022, 36, e22274. [Google Scholar] [CrossRef]
- Didichenko, S.A.; Navdaev, A.V.; Cukier, A.M.O.; Gille, A.; Schuetz, P.; Spycher, M.O.; Thérond, P.; Chapman, M.J.; Kontush, A.; Wright, S.D. Enhanced HDL Functionality in Small HDL Species Produced Upon Remodeling of HDL by Reconstituted HDL, CSL112. Circ. Res. 2016, 119, 751–763. [Google Scholar] [CrossRef]
- Wu, B.J.; Ong, K.L.; Shrestha, S.; Chen, K.; Tabet, F.; Barter, P.J.; Rye, K.-A. Inhibition of Arthritis in the Lewis Rat by Apolipoprotein A-I and Reconstituted High-Density Lipoproteins. Arter. Thromb. Vasc. Biol. 2014, 34, 543–551. [Google Scholar] [CrossRef]
- Guo, L.; Morin, E.E.; Yu, M.; Mei, L.; Fawaz, M.V.; Wang, Q.; Yuan, Y.; Zhan, C.-G.; Standiford, T.J.; Schwendeman, A.; et al. Replenishing HDL with Synthetic HDL Has Multiple Protective Effects against Sepsis in Mice. Sci. Signal. 2022, 15, eabl9322. [Google Scholar] [CrossRef] [PubMed]
- Smythies, L.E.; White, C.R.; Maheshwari, A.; Palgunachari, M.N.; Anantharamaiah, G.M.; Chaddha, M.; Kurundkar, A.R.; Datta, G. Apolipoprotein A-I Mimetic 4F Alters the Function of Human Monocyte-Derived Macrophages. Am. J. Physiol. Cell Physiol. 2010, 298, C1538–C1548. [Google Scholar] [CrossRef] [PubMed]
- White, C.R.; Smythies, L.E.; Crossman, D.K.; Palgunachari, M.N.; Anantharamaiah, G.M.; Datta, G. Regulation of Pattern Recognition Receptors by the Apolipoprotein A-I Mimetic Peptide 4F. Arter. Thromb. Vasc. Biol. 2012, 32, 2631–2639. [Google Scholar] [CrossRef]
- Ross, D.J.; Hough, G.; Hama, S.; Aboulhosn, J.; Belperio, J.A.; Saggar, R.; Van Lenten, B.J.; Ardehali, A.; Eghbali, M.; Reddy, S.; et al. Proinflammatory High-Density Lipoprotein Results from Oxidized Lipid Mediators in the Pathogenesis of Both Idiopathic and Associated Types of Pulmonary Arterial Hypertension. Pulm. Circ. 2015, 5, 640–648. [Google Scholar] [CrossRef]
- Amar, M.J.A.; D’Souza, W.; Turner, S.; Demosky, S.; Sviridov, D.; Stonik, J.; Luchoomun, J.; Voogt, J.; Hellerstein, M.; Sviridov, D.; et al. 5A Apolipoprotein Mimetic Peptide Promotes Cholesterol Efflux and Reduces Atherosclerosis in Mice. J. Pharmacol. Exp. Ther. 2010, 334, 634–641. [Google Scholar] [CrossRef]
- Shah, P.K.; Nilsson, J.; Kaul, S.; Fishbein, M.C.; Ageland, H.; Hamsten, A.; Johansson, J.; Karpe, F.; Cercek, B. Effects of Recombinant Apolipoprotein A-IMilano on Aortic Atherosclerosis in Apolipoprotein E–Deficient Mice. Circulation 1998, 97, 780–785. [Google Scholar] [CrossRef]
- Ameli, S.; Hultgardh-Nilsson, A.; Cercek, B.; Shah, P.K.; Forrester, J.S.; Ageland, H.; Nilsson, J. Recombinant Apolipoprotein A-I Milano Reduces Intimal Thickening after Balloon Injury in Hypercholesterolemic Rabbits. Circulation 1994, 90, 1935–1941. [Google Scholar] [CrossRef]
- Tardy, C.; Goffinet, M.; Boubekeur, N.; Ackermann, R.; Sy, G.; Bluteau, A.; Cholez, G.; Keyserling, C.; Lalwani, N.; Paolini, J.F.; et al. CER-001, a HDL-Mimetic, Stimulates the Reverse Lipid Transport and Atherosclerosis Regression in High Cholesterol Diet-Fed LDL-Receptor Deficient Mice. Atherosclerosis 2014, 232, 110–118. [Google Scholar] [CrossRef]
- Richart, A.L.; Reddy, M.; Khalaji, M.; Natoli, A.L.; Heywood, S.E.; Siebel, A.L.; Lancaster, G.L.; Murphy, A.J.; Carey, A.L.; Drew, B.G.; et al. Apo AI Nanoparticles Delivered Post Myocardial Infarction Moderate Inflammation. Circ. Res. 2020, 127, 1422–1436. [Google Scholar] [CrossRef]
- Gille, A.; Easton, R.; D’Andrea, D.; Wright, S.D.; Shear, C.L. CSL112 Enhances Biomarkers of Reverse Cholesterol Transport After Single and Multiple Infusions in Healthy Subjects. Arter. Thromb. Vasc. Biol. 2014, 34, 2106–2114. [Google Scholar] [CrossRef] [PubMed]
- Michael Gibson, C.; Korjian, S.; Tricoci, P.; Daaboul, Y.; Yee, M.; Jain, P.; Alexander, J.H.; Steg, P.G.; Lincoff, A.M.; Kastelein, J.J.P.; et al. Safety and Tolerability of CSL112, a Reconstituted, Infusible, Plasma-Derived Apolipoprotein A-I, After Acute Myocardial Infarction. Circulation 2016, 134, 1918–1930. [Google Scholar] [CrossRef] [PubMed]
- Gille, A.; D’Andrea, D.; Tortorici, M.A.; Hartel, G.; Wright, S.D. CSL112 (Apolipoprotein A-I [Human]) Enhances Cholesterol Efflux Similarly in Healthy Individuals and Stable Atherosclerotic Disease Patients. Arter. Thromb. Vasc. Biol. 2018, 38, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Gille, A.; Duffy, D.; Tortorici, M.A.; Wright, S.D.; Deckelbaum, L.I.; D’Andrea, D.M. Moderate Renal Impairment Does Not Impact the Ability of CSL112 (Apolipoprotein A-I [Human]) to Enhance Cholesterol Efflux Capacity. J. Clin. Pharmacol. 2019, 59, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Duffy, D.; Tricoci, P.; Kastrissios, H.; Pfister, M.; Wright, S.D.; Gille, A.; Tortorici, M.A. Pharmacometric Analyses to Characterize the Effect of CSL112 on Apolipoprotein A-I and Cholesterol Efflux Capacity in Acute Myocardial Infarction Patients. Br. J. Clin. Pharm. 2021, 87, 2558–2571. [Google Scholar] [CrossRef]
- Navab, M.; Anantharamaiah, G.M.; Hama, S.; Hough, G.; Reddy, S.T.; Frank, J.S.; Garber, D.W.; Handattu, S.; Fogelman, A.M. D-4F and Statins Synergize to Render HDL Antiinflammatory in Mice and Monkeys and Cause Lesion Regression in Old Apolipoprotein E–Null Mice. Arter. Thromb. Vasc. Biol. 2005, 25, 1426–1432. [Google Scholar] [CrossRef]
- Tabet, F.; Remaley, A.T.; Segaliny, A.I.; Millet, J.; Yan, L.; Nakhla, S.; Barter, P.J.; Rye, K.-A.; Lambert, G. The 5A Apolipoprotein A-I Mimetic Peptide Displays Antiinflammatory and Antioxidant Properties In Vivo and In Vitro. Arter. Thromb. Vasc. Biol. 2010, 30, 246–252. [Google Scholar] [CrossRef]
- Nowacki, T.M.; Remaley, A.T.; Bettenworth, D.; Eisenblätter, M.; Vowinkel, T.; Becker, F.; Vogl, T.; Roth, J.; Tietge, U.J.; Lügering, A.; et al. The 5A Apolipoprotein A-I (ApoA-I) Mimetic Peptide Ameliorates Experimental Colitis by Regulating Monocyte Infiltration. Br. J. Pharm. 2016, 173, 2780–2792. [Google Scholar] [CrossRef]
- Ortega-Paz, L.; Giordano, S.; Capodanno, D.; Mehran, R.; Gibson, C.M.; Angiolillo, D.J. Clinical Pharmacokinetics and Pharmacodynamics of CSL112. Clin. Pharm. 2023, 62, 541–558. [Google Scholar] [CrossRef]
- Jin, X.; Sviridov, D.; Liu, Y.; Vaisman, B.; Addadi, L.; Remaley, A.T.; Kruth, H.S. ABCA1 (ATP-Binding Cassette Transporter A1) Mediates ApoA-I (Apolipoprotein A-I) and ApoA-I Mimetic Peptide Mobilization of Extracellular Cholesterol Microdomains Deposited by Macrophages. Arter. Thromb. Vasc. Biol. 2016, 36, 2283–2291. [Google Scholar] [CrossRef]
- Calabresi, L.; Gomaraschi, M.; Franceschini, G. Endothelial Protection by High-Density Lipoproteins. Arter. Thromb. Vasc. Biol. 2003, 23, 1724–1731. [Google Scholar] [CrossRef]
- Muñoz-Vega, M.; Massó, F.; Páez, A.; Vargas-Alarcón, G.; Coral-Vázquez, R.; Mas-Oliva, J.; Carreón-Torres, E.; Pérez-Méndez, Ó. HDL-Mediated Lipid Influx to Endothelial Cells Contributes to Regulating Intercellular Adhesion Molecule (ICAM)-1 Expression and ENOS Phosphorylation. Int. J. Mol. Sci. 2018, 19, 3394. [Google Scholar] [CrossRef]
- Gupta, H.; Dai, L.; Datta, G.; Garber, D.W.; Grenett, H.; Li, Y.; Mishra, V.; Palgunachari, M.N.; Handattu, S.; Gianturco, S.H.; et al. Inhibition of Lipopolysaccharide-Induced Inflammatory Responses by an Apolipoprotein AI Mimetic Peptide. Circ. Res. 2005, 97, 236–243. [Google Scholar] [CrossRef]
- Patel, R.S.; Li, Q.; Ghasemzadeh, N.; Eapen, D.J.; Moss, L.D.; Janjua, A.U.; Manocha, P.; Al Kassem, H.; Veledar, E.; Samady, H.; et al. Circulating CD34 + Progenitor Cells and Risk of Mortality in a Population With Coronary Artery Disease. Circ. Res. 2015, 116, 289–297. [Google Scholar] [CrossRef]
- Gebhard, C.; Rhéaume, E.; Berry, C.; Brand, G.; Kernaleguen, A.-E.; Théberge-Julien, G.; Alam, M.A.; Lee, C.Y.W.; Boileau, L.; Chabot-Blanchet, M.; et al. Beneficial Effects of Reconstituted High-Density Lipoprotein (RHDL) on Circulating CD34+ Cells in Patients after an Acute Coronary Syndrome. PLoS ONE 2017, 12, e0168448. [Google Scholar] [CrossRef]
- Yang, N.; Tian, H.; Zhan, E.; Zhai, L.; Jiao, P.; Yao, S.; Lu, G.; Mu, Q.; Wang, J.; Zhao, A.; et al. Reverse-D-4F Improves Endothelial Progenitor Cell Function and Attenuates LPS-Induced Acute Lung Injury. Respir. Res. 2019, 20, 131. [Google Scholar] [CrossRef]
- Guo, Y.; Li, W.; Qian, M.; Jiang, T.; Guo, P.; Du, Q.; Lin, N.; Xie, X.; Wu, Z.; Lin, D.; et al. D-4F Ameliorates Contrast Media–Induced Oxidative Injuries in Endothelial Cells via the AMPK/PKC Pathway. Front. Pharm. 2021, 11, 556074. [Google Scholar] [CrossRef]
- He, D.; Zhao, M.; Wu, C.; Zhang, W.; Niu, C.; Yu, B.; Jin, J.; Ji, L.; Willard, B.; Mathew, A.V.; et al. Apolipoprotein A-1 Mimetic Peptide 4F Promotes Endothelial Repairing and Compromises Reendothelialization Impaired by Oxidized HDL through SR-B1. Redox Biol. 2018, 15, 228–242. [Google Scholar] [CrossRef]
- Calkin, A.C.; Drew, B.G.; Ono, A.; Duffy, S.J.; Gordon, M.V.; Schoenwaelder, S.M.; Sviridov, D.; Cooper, M.E.; Kingwell, B.A.; Jackson, S.P. Reconstituted High-Density Lipoprotein Attenuates Platelet Function in Individuals With Type 2 Diabetes Mellitus by Promoting Cholesterol Efflux. Circulation 2009, 120, 2095–2104. [Google Scholar] [CrossRef]
- Gurbel, P.A.; Tantry, U.S.; D’Andrea, D.; Chung, T.; Alexander, J.H.; Bliden, K.P.; Wright, S.D.; Tricoci, P. Evaluation of Potential Antiplatelet Effects of CSL112 (Apolipoprotein A-I [Human]) in Patients with Atherosclerosis: Results from a Phase 2a Study. J. Thromb. Thrombolysis 2018, 45, 469–476. [Google Scholar] [CrossRef]
- Plump, A.S.; Scott, C.J.; Breslow, J.L. Human Apolipoprotein A-I Gene Expression Increases High Density Lipoprotein and Suppresses Atherosclerosis in the Apolipoprotein E-Deficient Mouse. Proc. Natl. Acad. Sci. USA 1994, 91, 9607–9611. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.C.; Lawn, R.M.; Verstuyft, J.G.; Rubin, E.M. Human Apolipoprotein A-I Prevents Atherosclerosis Associated with Apolipoprotein[a] in Transgenic Mice. J. Lipid Res. 1994, 35, 2263–2267. [Google Scholar] [CrossRef] [PubMed]
- Duverger, N.; Kruth, H.; Emmanuel, F.; Caillaud, J.-M.; Viglietta, C.; Castro, G.; Tailleux, A.; Fievet, C.; Fruchart, J.C.; Houdebine, L.M.; et al. Inhibition of Atherosclerosis Development in Cholesterol-Fed Human Apolipoprotein A-I–Transgenic Rabbits. Circulation 1996, 94, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zanotti, I.; Reilly, M.P.; Glick, J.M.; Rothblat, G.H.; Rader, D.J. Overexpression of Apolipoprotein A-I Promotes Reverse Transport of Cholesterol from Macrophages to Feces In Vivo. Circulation 2003, 108, 661–663. [Google Scholar] [CrossRef]
- Valenta, D.T.; Bulgrien, J.J.; Banka, C.L.; Curtiss, L.K. Overexpression of Human ApoAI Transgene Provides Long-Term Atheroprotection in LDL Receptor-Deficient Mice. Atherosclerosis 2006, 189, 255–263. [Google Scholar] [CrossRef]
- Nanjee, M.N.; Crouse, J.R.; King, J.M.; Hovorka, R.; Rees, S.E.; Carson, E.R.; Morgenthaler, J.-J.; Lerch, P.; Miller, N.E. Effects of Intravenous Infusion of Lipid-Free Apo A-I in Humans. Arter. Thromb. Vasc. Biol. 1996, 16, 1203–1214. [Google Scholar] [CrossRef]
- Franceschini, G.; Sirtori, C.R.; Capurso, A.; Weisgraber, K.H.; Mahley, R.W. A-IMilano Apoprotein. Decreased High Density Lipoprotein Cholesterol Levels with Significant Lipoprotein Modifications and without Clinical Atherosclerosis in an Italian Family. J. Clin. Investig. 1980, 66, 892–900. [Google Scholar] [CrossRef]
- Sirtori, C.R.; Calabresi, L.; Franceschini, G.; Baldassarre, D.; Amato, M.; Johansson, J.; Salvetti, M.; Monteduro, C.; Zulli, R.; Muiesan, M.L.; et al. Cardiovascular Status of Carriers of the Apolipoprotein A-I Milano Mutant. Circulation 2001, 103, 1949–1954. [Google Scholar] [CrossRef]
- Nissen, S.E.; Tsunoda, T.; Tuzcu, E.M.; Schoenhagen, P.; Cooper, C.J.; Yasin, M.; Eaton, G.M.; Lauer, M.A.; Sheldon, W.S.; Grines, C.L.; et al. Effect of Recombinant ApoA-I Milano on Coronary Atherosclerosis in Patients with Acute Coronary Syndromes. JAMA 2003, 290, 2292. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Tuzcu, E.M.; Sipahi, I.; Schoenhagen, P.; Crowe, T.; Kapadia, S.; Nissen, S.E. Relationship Between Atheroma Regression and Change in Lumen Size After Infusion of Apolipoprotein A-I Milano. J. Am. Coll. Cardiol. 2006, 47, 992–997. [Google Scholar] [CrossRef]
- Bisgaier, C.L.; Ackermann, R.; Rea, T.; Rodrigueza, W.V.; Hartman, D. ApoA-IMilano Phospholipid Complex (ETC-216) Infusion in Human Volunteers. Insights into the Phenotypic Characteristics of ApoA-IMilano Carriers. Pharm. Res 2016, 111, 86–99. [Google Scholar] [CrossRef]
- Kallend, D.G.; Reijers, J.A.A.; Bellibas, S.E.; Bobillier, A.; Kempen, H.; Burggraaf, J.; Moerland, M.; Wijngaard, P.L.J. A Single Infusion of MDCO-216 (ApoA-1 Milano/POPC) Increases ABCA1-Mediated Cholesterol Efflux and Pre-Beta 1 HDL in Healthy Volunteers and Patients with Stable Coronary Artery Disease. Eur. Heart J. Cardiovasc. Pharm. 2016, 2, 23–29. [Google Scholar] [CrossRef]
- Kempen, H.J.; Gomaraschi, M.; Simonelli, S.; Calabresi, L.; Moerland, M.; Otvos, J.; Jeyarajah, E.; Kallend, D.; Wijngaard, P.L.J. Persistent Changes in Lipoprotein Lipids after a Single Infusion of Ascending Doses of MDCO-216 (ApoA-IMilano/POPC) in Healthy Volunteers and Stable Coronary Artery Disease Patients. Atherosclerosis 2016, 255, 17–24. [Google Scholar] [CrossRef]
- Kempen, H.J.; Asztalos, B.F.; Moerland, M.; Jeyarajah, E.; Otvos, J.; Kallend, D.G.; Bellibas, S.E.; Wijngaard, P.L.J. High-Density Lipoprotein Subfractions and Cholesterol Efflux Capacities After Infusion of MDCO-216 (Apolipoprotein A-IMilano/Palmitoyl-Oleoyl-Phosphatidylcholine) in Healthy Volunteers and Stable Coronary Artery Disease Patients. Arter. Thromb. Vasc. Biol. 2016, 36, 736–742. [Google Scholar] [CrossRef]
- Reijers, J.A.A.; Kallend, D.G.; Malone, K.E.; Jukema, J.W.; Wijngaard, P.L.J.; Burggraaf, J.; Moerland, M. MDCO-216 Does Not Induce Adverse Immunostimulation, in Contrast to Its Predecessor ETC-216. Cardiovasc. Drugs 2017, 31, 381–389. [Google Scholar] [CrossRef]
- Tardy, C.; Goffinet, M.; Boubekeur, N.; Cholez, G.; Ackermann, R.; Sy, G.; Keyserling, C.; Lalwani, N.; Paolini, J.F.; Dasseux, J.-L.; et al. HDL and CER-001 Inverse-Dose Dependent Inhibition of Atherosclerotic Plaque Formation in ApoE-/- Mice: Evidence of ABCA1 Down-Regulation. PLoS ONE 2015, 10, e0137584. [Google Scholar] [CrossRef]
- Tardif, J.-C.; Ballantyne, C.M.; Barter, P.; Dasseux, J.-L.; Fayad, Z.A.; Guertin, M.-C.; Kastelein, J.J.P.; Keyserling, C.; Klepp, H.; Koenig, W.; et al. Effects of the High-Density Lipoprotein Mimetic Agent CER-001 on Coronary Atherosclerosis in Patients with Acute Coronary Syndromes: A Randomized Trial. Eur. Heart J. 2014, 35, 3277–3286. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Andrews, J.; Kastelein, J.J.P.; Merkely, B.; Nissen, S.E.; Ray, K.K.; Schwartz, G.G.; Worthley, S.G.; Keyserling, C.; Dasseux, J.-L.; et al. Effect of Serial Infusions of CER-001, a Pre-β High-Density Lipoprotein Mimetic, on Coronary Atherosclerosis in Patients Following Acute Coronary Syndromes in the CER-001 Atherosclerosis Regression Acute Coronary Syndrome Trial. JAMA Cardiol. 2018, 3, 815. [Google Scholar] [CrossRef]
- Keyserling, C.H.; Barbaras, R.; Benghozi, R.; Dasseux, J.-L. Development of CER-001: Preclinical Dose Selection Through to Phase I Clinical Findings. Clin. Drug Investig. 2017, 37, 483–491. [Google Scholar] [CrossRef]
- Hovingh, G.K.; Smits, L.P.; Stefanutti, C.; Soran, H.; Kwok, S.; de Graaf, J.; Gaudet, D.; Keyserling, C.H.; Klepp, H.; Frick, J.; et al. The Effect of an Apolipoprotein A-I–Containing High-Density Lipoprotein–Mimetic Particle (CER-001) on Carotid Artery Wall Thickness in Patients with Homozygous Familial Hypercholesterolemia. Am. Heart J. 2015, 169, 736–742.e1. [Google Scholar] [CrossRef]
- Kootte, R.S.; Smits, L.P.; van der Valk, F.M.; Dasseux, J.-L.; Keyserling, C.H.; Barbaras, R.; Paolini, J.F.; Santos, R.D.; van Dijk, T.H.; Dallinga-van Thie, G.M.; et al. Effect of Open-Label Infusion of an ApoA-I-Containing Particle (CER-001) on RCT and Artery Wall Thickness in Patients with FHA. J. Lipid Res. 2015, 56, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.H.; Kaiser, Y.; van Olden, C.C.; Santos, R.D.; Dasseux, J.-L.; Genest, J.; Gaudet, D.; Westerink, J.; Keyserling, C.; Verberne, H.J.; et al. No Benefit of HDL Mimetic CER-001 on Carotid Atherosclerosis in Patients with Genetically Determined Very Low HDL Levels. Atherosclerosis 2020, 311, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Tardif, J.-C.; Grégoire, J.; L’Allier, P.L.; Ibrahim, R.; Lespérance, J.; Heinonen, T.M.; Kouz, S.; Berry, C.; Basser, R.; Lavoie, M.-A.; et al. Effects of reconstituted high-density lipoprotein infusions on coronary atherosclerosis: A randomized controlled trial. JAMA 2007, 297, 1675–1682. [Google Scholar] [CrossRef]
- Diditchenko, S.; Gille, A.; Pragst, I.; Stadler, D.; Waelchli, M.; Hamilton, R.; Leis, A.; Wright, S.D. Novel Formulation of a Reconstituted High-Density Lipoprotein (CSL112) Dramatically Enhances ABCA1-Dependent Cholesterol Efflux. Arter. Thromb. Vasc. Biol. 2013, 33, 2202–2211. [Google Scholar] [CrossRef] [PubMed]
- Easton, R.; Gille, A.; D’Andrea, D.; Davis, R.; Wright, S.D.; Shear, C. A Multiple Ascending Dose Study of CSL112, an Infused Formulation of ApoA-I. J. Clin. Pharmacol. 2014, 54, 301–310. [Google Scholar] [CrossRef]
- Bauer, L.; Kern, S.; Rogacev, K.S.; Emrich, I.E.; Zawada, A.; Fliser, D.; Heinemann, A.; Heine, G.H.; Marsche, G. HDL Cholesterol Efflux Capacity and Cardiovascular Events in Patients With Chronic Kidney Disease. J. Am. Coll. Cardiol. 2017, 69, 246–247. [Google Scholar] [CrossRef]
- Beyerle, A.; Greene, B.; Dietrich, B.; Kingwell, B.A.; Panjwani, P.; Wright, S.D.; Herzog, E. Co-Administration of CSL112 (Apolipoprotein A-I [Human]) with Atorvastatin and Alirocumab Is Not Associated with Increased Hepatotoxic or Toxicokinetic Effects in Rats. Toxicol. Appl. Pharm. 2021, 422, 115557. [Google Scholar] [CrossRef]
- Gibson, C.M.; Kastelein, J.J.P.; Phillips, A.T.; Aylward, P.E.; Yee, M.K.; Tendera, M.; Nicholls, S.J.; Pocock, S.; Goodman, S.G.; Alexander, J.H.; et al. Rationale and Design of ApoA-I Event Reducing in Ischemic Syndromes II (AEGIS-II): A Phase 3, Multicenter, Double-Blind, Randomized, Placebo-Controlled, Parallel-Group Study to Investigate the Efficacy and Safety of CSL112 in Subjects after Acute Myocardial Infarction. Am. Heart J. 2021, 231, 121–127. [Google Scholar] [CrossRef]
- Chung, B.H.; Anatharamaiah, G.M.; Brouillette, C.G.; Nishida, T.; Segrest, J.P. Studies of Synthetic Peptide Analogs of the Amphipathic Helix. Correlation of Structure with Function. J. Biol. Chem. 1985, 260, 10256–10262. [Google Scholar] [CrossRef]
- Iwata, A.; Miura, S.; Zhang, B.; Imaizumi, S.; Uehara, Y.; Shiomi, M.; Saku, K. Antiatherogenic Effects of Newly Developed Apolipoprotein A-I Mimetic Peptide/Phospholipid Complexes against Aortic Plaque Burden in Watanabe-Heritable Hyperlipidemic Rabbits. Atherosclerosis 2011, 218, 300–307. [Google Scholar] [CrossRef]
- di Bartolo, B.A.; Vanags, L.Z.; Tan, J.T.; Bao, S.; Rye, K.-A.; Barter, P.J.; Bursill, C.A. The Apolipoprotein A-I Mimetic Peptide, ETC-642, Reduces Chronic Vascular Inflammation in the Rabbit. Lipids Health Dis. 2011, 10, 224. [Google Scholar] [CrossRef]
- Khan, M.L.N.; Drake, S.L.; Crockatt, J.G.; Dasseux, J.L.H. Single-dose intravenous infusion of ETC-642, a 22-Mer ApoA-I analogue and phospholipids complex, elevates HDL-C in atherosclerosis patients. Circulation 2003, 108, 563–564. [Google Scholar]
- Smith, C.K.; Seto, N.L.; Vivekanandan-Giri, A.; Yuan, W.; Playford, M.P.; Manna, Z.; Hasni, S.A.; Kuai, R.; Mehta, N.N.; Schwendeman, A.; et al. Lupus High-Density Lipoprotein Induces Proinflammatory Responses in Macrophages by Binding Lectin-like Oxidised Low-Density Lipoprotein Receptor 1 and Failing to Promote Activating Transcription Factor 3 Activity. Ann. Rheum. Dis. 2017, 76, 602–611. [Google Scholar] [CrossRef]
- Taylor, M.J.; Sanjanwala, A.R.; Morin, E.E.; Rowland-Fisher, E.; Anderson, K.; Schwendeman, A.; Rainey, W.E. Synthetic High-Density Lipoprotein (SHDL) Inhibits Steroid Production in HAC15 Adrenal Cells. Endocrinology 2016, 157, 3122–3129. [Google Scholar] [CrossRef]
- Navab, M.; Anantharamaiah, G.M.; Reddy, S.T.; Hama, S.; Hough, G.; Grijalva, V.R.; Wagner, A.C.; Frank, J.S.; Datta, G.; Garber, D.; et al. Oral D-4F Causes Formation of Pre-β High-Density Lipoprotein and Improves High-Density Lipoprotein–Mediated Cholesterol Efflux and Reverse Cholesterol Transport from Macrophages in Apolipoprotein E–Null Mice. Circulation 2004, 109, 3215–3220. [Google Scholar] [CrossRef]
- Ou, Z.; Ou, J.; Ackerman, A.W.; Oldham, K.T.; Pritchard, K.A. L-4F, an Apolipoprotein A-1 Mimetic, Restores Nitric Oxide and Superoxide Anion Balance in Low-Density Lipoprotein-Treated Endothelial Cells. Circulation 2003, 107, 1520–1524. [Google Scholar] [CrossRef]
- Wool, G.D.; Cabana, V.G.; Lukens, J.; Shaw, P.X.; Binder, C.J.; Witztum, J.L.; Reardon, C.A.; Getz, G.S. 4F Peptide Reduces Nascent Atherosclerosis and Induces Natural Antibody Production in Apolipoprotein E-null Mice. FASEB J. 2011, 25, 290–300. [Google Scholar] [CrossRef]
- Datta, G. HDL Mimetic Peptide Administration Improves Left Ventricular Filling and Cardiac Output in Lipopolysaccharide-Treated Rats. J. Clin. Exp. Cardiol. 2011, 2, 1000172. [Google Scholar] [CrossRef]
- Moreira, R.S.; Irigoyen, M.C.; Capcha, J.M.C.; Sanches, T.R.; Gutierrez, P.S.; Garnica, M.R.; de Noronha, I.L.; Andrade, L. Synthetic Apolipoprotein A–I Mimetic Peptide 4F Protects Hearts and Kidneys after Myocardial Infarction. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2020, 318, R529–R544. [Google Scholar] [CrossRef]
- Bloedon, L.T.; Dunbar, R.; Duffy, D.; Pinell-Salles, P.; Norris, R.; DeGroot, B.J.; Movva, R.; Navab, M.; Fogelman, A.M.; Rader, D.J. Safety, Pharmacokinetics, and Pharmacodynamics of Oral ApoA-I Mimetic Peptide D-4F in High-Risk Cardiovascular Patients. J. Lipid Res. 2008, 49, 1344–1352. [Google Scholar] [CrossRef]
- Dunbar, R.L.; Movva, R.; Bloedon, L.T.; Duffy, D.; Norris, R.B.; Navab, M.; Fogelman, A.M.; Rader, D.J. Oral Apolipoprotein A-I Mimetic D-4F Lowers HDL-Inflammatory Index in High-Risk Patients: A First-in-Human Multiple-Dose, Randomized Controlled Trial. Clin. Transl. Sci. 2017, 10, 455–469. [Google Scholar] [CrossRef] [PubMed]
- Watson, C.E.; Weissbach, N.; Kjems, L.; Ayalasomayajula, S.; Zhang, Y.; Chang, I.; Navab, M.; Hama, S.; Hough, G.; Reddy, S.T.; et al. Treatment of Patients with Cardiovascular Disease with L-4F, an Apo-A1 Mimetic, Did Not Improve Select Biomarkers of HDL Function. J. Lipid Res. 2011, 52, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Troutt, J.S.; Alborn, W.E.; Mosior, M.K.; Dai, J.; Murphy, A.T.; Beyer, T.P.; Zhang, Y.; Cao, G.; Konrad, R.J. An Apolipoprotein A-I Mimetic Dose-Dependently Increases the Formation of Preβ1 HDL in Human Plasma. J. Lipid Res. 2008, 49, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Bourdi, M.; Amar, M.; Remaley, A.T.; Terse, P.S. Intravenous Toxicity and Toxicokinetics of an HDL Mimetic, Fx-5A Peptide Complex, in Cynomolgus Monkeys. Regul. Toxicol. Pharmacol. 2018, 100, 59–67. [Google Scholar] [CrossRef]
- Fotakis, P.; Kothari, V.; Thomas, D.G.; Westerterp, M.; Molusky, M.M.; Altin, E.; Abramowicz, S.; Wang, N.; He, Y.; Heinecke, J.W.; et al. Anti-Inflammatory Effects of HDL (High-Density Lipoprotein) in Macrophages Predominate Over Proinflammatory Effects in Atherosclerotic Plaques. Arter. Thromb. Vasc. Biol. 2019, 39, e253–e272. [Google Scholar] [CrossRef]
- Li, D.; Fawaz, M.V.; Morin, E.E.; Ming, R.; Sviridov, D.; Tang, J.; Ackermann, R.; Olsen, K.; Remaley, A.T.; Schwendeman, A. Effect of Synthetic High Density Lipoproteins Modification with Polyethylene Glycol on Pharmacokinetics and Pharmacodynamics. Mol. Pharm. 2018, 15, 83–96. [Google Scholar] [CrossRef]
- Sanchez-Gaytan, B.L.; Fay, F.; Lobatto, M.E.; Tang, J.; Ouimet, M.; Kim, Y.; van der Staay, S.E.M.; van Rijs, S.M.; Priem, B.; Zhang, L.; et al. HDL-Mimetic PLGA Nanoparticle to Target Atherosclerosis Plaque Macrophages. Bioconjug. Chem. 2015, 26, 443–451. [Google Scholar] [CrossRef]
- Marrache, S.; Dhar, S. Biodegradable Synthetic High-Density Lipoprotein Nanoparticles for Atherosclerosis. Proc. Natl. Acad. Sci. USA 2013, 110, 9445–9450. [Google Scholar] [CrossRef]
- Lai, C.-T.; Sun, W.; Palekar, R.U.; Thaxton, C.S.; Schatz, G.C. Molecular Dynamics Simulation and Experimental Studies of Gold Nanoparticle Templated HDL-like Nanoparticles for Cholesterol Metabolism Therapeutics. ACS Appl. Mater. Interfaces 2017, 9, 1247–1254. [Google Scholar] [CrossRef]
- Yu, M.; Hong, K.; Adili, R.; Mei, L.; Liu, L.; He, H.; Guo, Y.; Chen, Y.E.; Holinstat, M.; Schwendeman, A. Development of Activated Endothelial Targeted High-Density Lipoprotein Nanoparticles. Front. Pharm. 2022, 13, 3224. [Google Scholar] [CrossRef]
- Sharifov, O.F.; Xu, X.; Gaggar, A.; Grizzle, W.E.; Mishra, V.K.; Honavar, J.; Litovsky, S.H.; Palgunachari, M.N.; White, C.R.; Anantharamaiah, G.M.; et al. Anti-Inflammatory Mechanisms of Apolipoprotein A-I Mimetic Peptide in Acute Respiratory Distress Syndrome Secondary to Sepsis. PLoS ONE 2013, 8, e64486. [Google Scholar] [CrossRef]
- Pajkrt, D.; Doran, J.E.; Koster, F.; Lerch, P.G.; Arnet, B.; van der Poll, T.; ten Cate, J.W.; van Deventer, S.J. Antiinflammatory Effects of Reconstituted High-Density Lipoprotein during Human Endotoxemia. J. Exp. Med. 1996, 184, 1601–1608. [Google Scholar] [CrossRef]
- Pajkrt, D.; Lerch, P.G.; van der Poll, T.; Levi, M.; Illi, M.; Doran, J.E.; Arnet, B.; van den Ende, A.; ten Cate, J.W.; van Deventer, S.J. Differential Effects of Reconstituted High-Density Lipoprotein on Coagulation, Fibrinolysis and Platelet Activation during Human Endotoxemia. Thromb. Haemost. 1997, 77, 303–307. [Google Scholar] [CrossRef]
- Lapergue, B.; Moreno, J.-A.; Dang, B.Q.; Coutard, M.; Delbosc, S.; Raphaeli, G.; Auge, N.; Klein, I.; Mazighi, M.; Michel, J.-B.; et al. Protective Effect of High-Density Lipoprotein-Based Therapy in a Model of Embolic Stroke. Stroke 2010, 41, 1536–1542. [Google Scholar] [CrossRef]
- Khirfan, G.; Tejwani, V.; Wang, X.; Li, M.; DiDonato, J.; Dweik, R.A.; Smedira, N.; Heresi, G.A. Plasma Levels of High Density Lipoprotein Cholesterol and Outcomes in Chronic Thromboembolic Pulmonary Hypertension. PLoS ONE 2018, 13, e0197700. [Google Scholar] [CrossRef]
- Avci, A.; Biricik, S.; Avci, B.S.; Yesiloglu, O.; Sumbul, H.E.; Icme, F.; Koca, H.; Cinar, H.; Koc, M.; Satar, S. The New Prognostic Factor for Pulmonary Embolism: The Ratio of Monocyte Count to HDL Cholesterol. Am. J. Emerg. Med. 2021, 46, 212–216. [Google Scholar] [CrossRef]
- Faguer, S.; Del Bello, A.; Danet, C.; Renaudineau, Y.; Izopet, J.; Kamar, N. Apolipoprotein-A-I for Severe COVID-19-Induced Hyperinflammatory States: A Prospective Case Study. Front. Pharm. 2022, 13, 3925. [Google Scholar] [CrossRef]
- Tanaka, S.; Begue, F.; Veeren, B.; Tran-Dinh, A.; Robert, T.; Tashk, P.; Lortat-Jacob, B.; Faille, D.; de Chaisemartin, L.; Zappella, N.; et al. First Recombinant High-Density Lipoprotein Particles Administration in a Severe ICU COVID-19 Patient, a Multi-Omics Exploratory Investigation. Biomedicines 2022, 10, 754. [Google Scholar] [CrossRef]
- Lin, Y.-J.; Lin, J.-L.; Peng, Y.-C.; Li, S.-L.; Chen, L.-W. TG/HDL-C Ratio Predicts in-Hospital Mortality in Patients with Acute Type A Aortic Dissection. BMC Cardiovasc. Disord. 2022, 22, 346. [Google Scholar] [CrossRef]
- Adorni, M.P.; Palumbo, M.; Marchi, C.; Zimetti, F.; Ossoli, A.; Turri, M.; Bernini, F.; Hollan, I.; Moláček, J.; Treska, V.; et al. HDL Metabolism and Functions Impacting on Cell Cholesterol Homeostasis Are Specifically Altered in Patients with Abdominal Aortic Aneurysm. Front. Immunol. 2022, 13, 5249. [Google Scholar] [CrossRef]
- Rodríguez-Carrio, J.; Cerro-Pardo, I.; Lindholt, J.S.; Bonzon-Kulichenko, E.; Martínez-López, D.; Roldán-Montero, R.; Escolà-Gil, J.-C.; Michel, J.-B.; Blanco-Colio, L.M.; Vázquez, J.; et al. Malondialdehyde-Modified HDL Particles Elicit a Specific IgG Response in Abdominal Aortic Aneurysm. Free Radic. Biol. Med. 2021, 174, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Burillo, E.; Lindholt, J.S.; Molina-Sánchez, P.; Jorge, I.; Martinez-Pinna, R.; Blanco-Colio, L.M.; Tarin, C.; Torres-Fonseca, M.M.; Esteban, M.; Laustsen, J.; et al. ApoA-I/HDL-C Levels Are Inversely Associated with Abdominal Aortic Aneurysm Progression. Thromb. Haemost. 2015, 113, 1335–1346. [Google Scholar] [CrossRef] [PubMed]
- Martínez-López, D.; Camafeita, E.; Cedó, L.; Roldan-Montero, R.; Jorge, I.; García-Marqués, F.; Gómez-Serrano, M.; Bonzon-Kulichenko, E.; Blanco-Vaca, F.; Blanco-Colio, L.M.; et al. APOA1 Oxidation Is Associated to Dysfunctional High-Density Lipoproteins in Human Abdominal Aortic Aneurysm. EBioMedicine 2019, 43, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Delbosc, S.; Rouer, M.; Alsac, J.-M.; Louedec, L.; Al Shoukr, F.; Rouzet, F.; Michel, J.-B.; Meilhac, O. High-Density Lipoprotein Therapy Inhibits Porphyromonas Gingivalis-Induced Abdominal Aortic Aneurysm Progression. Thromb. Haemost. 2016, 115, 789–799. [Google Scholar] [CrossRef]



| HDL Mimetic | Protein Type | Peptide Sequence (P) | Phospholipid (L) | Nanoparticle Size | Ratio (P/L) | Administration | Patients Recruited | Clinical Trial | Major Findings | Reason to Discontinue Clinical Trial |
|---|---|---|---|---|---|---|---|---|---|---|
| ETC-216 | Recombinant ApoA-IM | ApoA-I Milano | DPPC | 7–30 nm | 1:2.7 (w/w) | 5 weekly, intravenous, 15 mg/kg and 45 mg/kg | 57 | Milano, Phase II | Significantly decreased atheroma volume | Change of company ownership; protein production and quality constrain |
| MDCO-206 | Recombinant ApoA-IM | ApoA-I Milano | POPC | 7–30 nm | 1:1.1 (w/w) | 5 weekly, intravenous, 20 mg/kg | 126 | MILANO-PILOT, Phase III, NCT02678923 | 80.4% increase in cholesterol efflux, no increment in plaque regression | Change of company ownership; protein production and quality constrain |
| CER-001 | Recombinant ApoA-I | ApoA-I native | SM, DPPG | 7–13 nm | 1:2.7 (w/w) | 10 weekly, intravenous, 3 mg/kg with statins and 6 weekly, intravenous, 12 mg/kg | 507 293 | CHI SQUARE, Phase II NCT01201837 and CARAT, Phase II NCT2484378 | Failed to promote coronary atherosclerosis regression | Insufficient beneficial effects in CAD |
| CSL111 | Plasma purified human ApoA-I | ApoA-I native | Soy PC | 7–30 nm | 1:150 molar ratio | 4 weekly, intravenous, 40 mg/kg and 80 mg/kg | 111 | ERASE Phase II, NCT00225719 | Significant improvement in the plaque characterization index, elevation in liver enzymes in 80 mg/kg group | Modified and improved to CSL112 |
| CSL112 | Plasma purified human ApoA-I | ApoA-I native | Soy PC | 7–13 nm | 1:55 molar ratio | 4 weekly, intravenous, 2 g and 6 g | 1258 | AEGIS-I, Phase II, NCT02108262 | No renal/hepatic toxicity, well tolerated | _ |
| CSL112 | Plasma purified human ApoA-I | ApoA-I native | Soy PC | 7–13 nm | 1:55 molar ratio | 4 weekly intravenous, 6 g | 17400 | AEGIS-II, Phase III, NCT03473223 | Results yet to be concluded | _ |
| ETC-642 | ApoA-I mimetic- 22 amino acids, single helix | P-V-L-D-L-F-R-E-L-L-NE-L-L-E-AL-K-Q-K-L-K | DPPC and SM | 7–13 nm | 1:1:1 molar ratio | 4 weekly intravenous, up to 30 mg/kg | 28 | Phase I | Safe and well tolerated, LCAT activation | Change of company ownership |
| D-4F | ApoA-I mimetic peptide, 18 amino acids, bihelical | Ac-D-W-F-K-A-F-Y-D-KV-A-E-K-F-K-E-A-FNH 2 | Non- lipidated | - | - | Three separate groups, sequential, ascending doses 100 mg, 300 mg, or 500 mg, oral | 104 | Phase I, NCT00907998 | Increased hydrophobicity, blocks absorption of production of oxidized lipids, anti-inflammatory | Insufficient bioavailability |
| L-4F | ApoA-I mimetic peptide, 18 amino acids long, bihelical | 4F enantiomer, L-amino acids | Non- lipidated | - | - | Single and 7-daily infusions, Intravenous, subcutaneous | 176 | Phase I, NCT00568594 | Similar to 4F but shorter circulating time due to proteolysis | Insufficient HDL-function-biomarker improvements |
| 5A | ApoA-I mimetic peptide, 37 amino acids, bihelical | D-W-L-K-A-F-Y-D-K-V-A-E-K-L-K-E-A-F-P-D-W-A-K-A-A-Y-D-K-A-A-E-K-A-K-E-A-A | SM | - | 1:8 (primate study) | 5 weekly, intravenous, 2.5, 5.0, 10.0 and 20.0 mg/kg | 64, currently recruiting | Phase I, NCT04216342 | ABCA-1 specificity, results yet to be concluded | _ |
| HDL Mimetic | In-Vitro | Primary Effects | References |
|---|---|---|---|
| CSL111 | Human monocyte-derived macrophages | Cholesterol efflux, TLR2 suppression, NF-κB inactivation | [204] |
| ETC-642 | THP-1 monocytes | Reduction in monocyte adhesion, MCP-1 and NF-κB activation | [188] |
| ETC-642 | RAW264.7 macrophages | Reduction in TLR4 targeting to lipid rafts | [205] |
| PS-ApoA-I | THP-1 monocytes and human monocyte derived macrophages | Cholesterol efflux, reduction in inflammatory cytokine production and lipid raft abundance | [202] |
| 4F peptide | Human monocyte-derived macrophages, THP-1 monocytes | Cholesterol efflux, M2 polarization, impaired phosphorylation and nuclear translocation of NF-κBp65, phagocytosis, inflammatory surface markers, CD14 expression, TLR4 assembly and recycling, reduced monocyte adhesion and migration | [206,207] |
| 4F peptide | Human monocytes | Reduced chemotaxis | [208] |
| 18 A peptide | THP-1 macrophages, human umbilical vein endothelial cells | Cholesterol efflux, M2 polarization, reduction in reduction in inflammatory cytokine production and MCP-1 secretion | [182] |
| 5A-POPC/5A-SM-DPCC | RAW 264.7 macrophage | Cholesterol efflux | [209] |
| HDL Mimetic | in-vivo | Primary Effects | References |
| ApoA-IM-PC | ApoE-deficient mice | Cholesterol efflux, reduced macrophage content in plaques | [210] |
| ApoA-IM | Hypercholesteremic rabbits | Cholesterol efflux, reduced intimal thickening and macrophage counts | [211] |
| CER-001 | LDL receptor deficient mice | Cholesterol efflux, reduction in MCP-1 and inflammatory cytokine production | [212] |
| CSL-111 | Murine model of myocardial infarction | Cholesterol efflux, reduced CD11b on circulating monocytes | [213] |
| CSL-112 | Clinical trials | Cholesterol efflux, reduced plaque instability markers and anti-inflammatory cytokines, increased cholesterol esterification by LCAT | [204,214,215,216,217,218] |
| 4F peptide | Dyslipidemic mice model of CAD | Cholesterol efflux, reduction in macrophage content in lipid-rich lesions | [206,219] |
| 5A-PLPC | Collared arteries in rabbit | Cholesterol efflux, reduced oxidative stress | [220] |
| 5A-POPC/ 5A-SM-DPCC | ApoE-deficient mice | Cholesterol efflux, reduction in plaque coverage | [209] |
| 5A peptide | Murine colitis model | Reduced monocyte infiltration | [221] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rani, A.; Marsche, G. A Current Update on the Role of HDL-Based Nanomedicine in Targeting Macrophages in Cardiovascular Disease. Pharmaceutics 2023, 15, 1504. https://doi.org/10.3390/pharmaceutics15051504
Rani A, Marsche G. A Current Update on the Role of HDL-Based Nanomedicine in Targeting Macrophages in Cardiovascular Disease. Pharmaceutics. 2023; 15(5):1504. https://doi.org/10.3390/pharmaceutics15051504
Chicago/Turabian StyleRani, Alankrita, and Gunther Marsche. 2023. "A Current Update on the Role of HDL-Based Nanomedicine in Targeting Macrophages in Cardiovascular Disease" Pharmaceutics 15, no. 5: 1504. https://doi.org/10.3390/pharmaceutics15051504