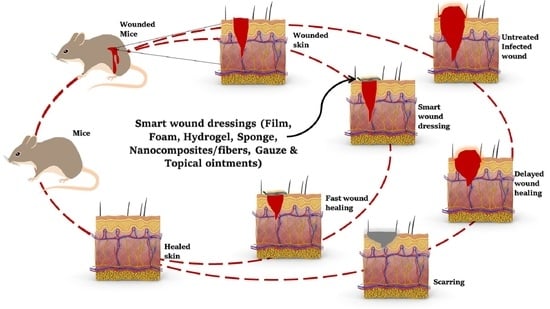
Multifunctional and Smart Wound Dressings—A Review on Recent Research Advancements in Skin Regenerative Medicine
Abstract
:1. Introduction
2. Current Challenges and Perspective
3. Characteristics of Multifunctional Wound Dressing
3.1. pH Responsive Wound Dressings
3.2. Temperature Responsive Wound Dressings
3.3. Pressure Responsive Wound Dressings
3.4. Moisture Responsive Wound Dressings
3.5. Sustained Drug Releasing Wound Dressings
4. Articles Search for Narrative Review-Inclusion & Exclusion Criteria
5. Wound Dressing
5.1. Hydrogels
5.2. Films
| Dressing Composition | Dressing Material Evaluated/ Group of Subjects | Key Findings | Reference |
|---|---|---|---|
| Sodium alginate and pectin loaded with simvastatin (SIM) | Control, Saline, Hydrogel film, Kaltostat® commercial dressing, SIM-hydrogel film | Better angiogenic effect contributed accelerated healing, quicker re-epithelialization and improved collagen deposition | [78] |
| Hammada scoparia leaf extract (PSP) and poly (vinyl alcohol) (PVA) | Saline, Cytol centella cream, PVA film (100%), (70:30) PSP/PVA film | Hastened wound closure and reepithelialisation | [79] |
| Xyloglucan dressing (XG) and Concanavalin A © | Saline, XG, XGC | Non-toxic, homogenous, angiogenesis, remodelling, early epithelialization | [80] |
| Chitosan film (CF) loaded with Vancomycin (V) | Saline, Burn, Burn vancomycin, Burn + CF, Burn + VCF2 | Controlled drug release, remarkable antimicrobial effect and enhanced wound recovery | [81] |
| Polycaprolactone (PCL), Gelatin, poly (perfluoro decyl methacrylate) + poly (dimethyl siloxane) + poly (perfluoro decyl methacrylate (PMFA) | PCL-Gelatin, PCL-PMFA, PCL G + F | Non-adherent ability and constant drainage performance | [82] |
| Ofloxacin (O), tea tree (T) and lavender oil (L) in gellan gum hydrogel film | Blank, O, L, T, OL, OT | Antibacterial and wound-healing properties are notable | [83] |
5.3. Sponges
5.4. Nanofibers/Nanocomposite
5.5. Foams
5.6. Gauzes
5.7. Others
5.8. Plant Derived Bioactive Compounds and Biopolymers in Wound Therapy
5.9. Polymers
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, R.; Hu, J.; Qian, W.; Chen, L.; Zhang, D. Recent advances in nanotherapeutics for the treatment of burn wounds. Burns Trauma 2021, 9, tkab026. [Google Scholar] [CrossRef] [PubMed]
- Silina, E.V.; Khokhlov, N.V.; Stupin, V.A.; Manturova, N.E.; Vasin, V.I.; Velikanov, E.V.; Popov, A.L.; Gavrilyuk, V.B.; Artyushkova, E.B.; Gladchenko, M.P.; et al. Multicomponent Polysaccharide Essential Formula of Wound Healing Medicines Enriched with Fibroblast Growth Factor. Int. J. Biomed. 2019, 9, 247–250. [Google Scholar] [CrossRef]
- Wild, H.; Stewart, B.T.; LeBoa, C.; Stave, C.D.; Wren, S.M. Epidemiology of Injuries Sustained by Civilians and Local Combatants in Contemporary Armed Conflict: An Appeal for a Shared Trauma Registry Among Humanitarian Actors. World J. Surg. 2020, 44, 1863–1873. [Google Scholar] [CrossRef]
- Toussaint, J.; Singer, A.J. The evaluation and management of thermal injuries: 2014 update. Clin. Exp. Emerg. Med. 2014, 1, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Yang, Y.; He, J.; Li, M.; Guo, B. Novel supramolecular self-healing silk fibroin-based hydrogel via host–guest interaction as wound dressing to enhance wound healing. Chem. Eng. J. 2021, 417, 128278. [Google Scholar] [CrossRef]
- De Luca, I.; Pedram, P.; Moeini, A.; Cerruti, P.; Peluso, G.; Di Salle, A.; Germann, N. Nanotechnology Development for Formulating Essential Oils in Wound Dressing Materials to Promote the Wound-Healing Process: A Review. Appl. Sci. 2021, 11, 1713. [Google Scholar] [CrossRef]
- Wallace, H.A.; Basehore, B.M.; Zito, P.M. Wound Healing Phases. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: http://www.ncbi.nlm.nih.gov/books/NBK470443/ (accessed on 4 February 2022).
- Irfan-Maqsood, M. Classification of Wounds: Know before Research and Clinical Practice. Cell Ther. Regen. Med. J. 2016, 1, 79. [Google Scholar] [CrossRef]
- Sharma, R.K.; John, J.R. Role of stem cells in the management of chronic wounds. Indian J. Plast. Surg. 2012, 45, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Shedoeva, A.; Leavesley, D.; Upton, Z.; Fan, C. Wound Healing and the Use of Medicinal Plants. Evid. Based Complement. Alternat. Med. 2019, 2019, 1–30. [Google Scholar] [CrossRef]
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin Wound Healing Process and New Emerging Technologies for Skin Wound Care and Regeneration. Pharmaceutics 2020, 12, 735. [Google Scholar] [CrossRef]
- Karadag, A.; Sengul, T. Challenges faced by doctors and nurses in wound care management during the COVID-19 pandemic in Turkey and their views on telehealth. J. Tissue Viability 2021, 30, 484–488. [Google Scholar] [CrossRef] [PubMed]
- Deufert, D.; Graml, R. Disease-specific, health-related quality of life (HRQoL) of people with chronic wounds—A descriptive cross-sectional study using the Wound-QoL. Wound Med. 2017, 16, 29–33. [Google Scholar] [CrossRef]
- Guo, S.; DiPietro, L.A. Factors Affecting Wound Healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Halim, A.S.; Khoo, T.L.; Mat Saad, A.Z. Wound bed preparation from a clinical perspective. Indian J. Plast. Surg. 2012, 45, 193–202. [Google Scholar] [CrossRef]
- Dhivya, S.; Padma, V.V.; Santhini, E. Wound dressings—A review. BioMedicine 2015, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Politis, C.; Schoenaers, J.; Jacobs, R.; Agbaje, J.O. Wound Healing Problems in the Mouth. Front. Physiol. 2016, 7, 507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karner, L.; Drechsler, S.; Metzger, M.; Slezak, P.; Zipperle, J.; Pinar, G.; Sterflinger, K.; Leisch, F.; Grillari, J.; Osuchowski, M.; et al. Contamination of wounds with fecal bacteria in immuno-suppressed mice. Sci. Rep. 2020, 10, 11494. [Google Scholar] [CrossRef]
- Sen, C.K. Human Wound and Its Burden: Updated 2020 Compendium of Estimates. Adv. Wound Care 2021, 10, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Nussbaum, S.R.; Carter, M.J.; Fife, C.E.; DaVanzo, J.; Haught, R.; Nusgart, M.; Cartwright, D. An Economic Evaluation of the Impact, Cost, and Medicare Policy Implications of Chronic Nonhealing Wounds. Value Health 2018, 21, 27–32. [Google Scholar] [CrossRef] [Green Version]
- Rani, N.; Chinnasamy, T.; Swamy, V.H.; Deshpande, G.; Achar, R.R. Evidence of new calcium-dependent cysteine protease of Melia dubia latex with fibrinolytic potential. J. Proteins Proteom. 2021, 12, 115–123. [Google Scholar] [CrossRef]
- Tan, W.S.; Arulselvan, P.; Ng, S.-F.; Taib, C.N.M.; Sarian, M.N.; Fakurazi, S. Improvement of diabetic wound healing by topical application of Vicenin-2 hydrocolloid film on Sprague Dawley rats. BMC Complement. Altern. Med. 2019, 19, 20. [Google Scholar] [CrossRef]
- Adamu, B.F.; Gao, J.; Jhatial, A.K.; Kumelachew, D.M. A review of medicinal plant-based bioactive electrospun nano fibrous wound dressings. Mater. Des. 2021, 209, 109942. [Google Scholar] [CrossRef]
- Arafa, A.A.; Nada, A.A.; Ibrahim, A.Y.; Sajkiewicz, P.; Zahran, M.K.; Hakeim, O.A. Preparation and characterization of smart therapeutic pH-sensitive wound dressing from red cabbage extract and chitosan hydrogel. Int. J. Biol. Macromol. 2021, 182, 1820–1831. [Google Scholar] [CrossRef] [PubMed]
- Montaser, A.S.; Rehan, M.; El-Senousy, W.M.; Zaghloul, S. Designing strategy for coating cotton gauze fabrics and its application in wound healing. Carbohydr. Polym. 2020, 244, 116479. [Google Scholar] [CrossRef] [PubMed]
- Omidi, M.; Yadegari, A.; Tayebi, L. Wound dressing application of pH-sensitive carbon dots/chitosan hydrogel. RSC Adv. 2017, 7, 10638–10649. [Google Scholar] [CrossRef] [Green Version]
- Tang, N.; Zheng, Y.; Jiang, X.; Zhou, C.; Jin, H.; Jin, K.; Wu, W.; Haick, H. Wearable Sensors and Systems for Wound Healing-Related pH and Temperature Detection. Micromachines 2021, 12, 430. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.-H.; Samandari, M.; Li, C.; Li, H.; Song, D.; Zhang, Y.; Tamayol, A.; Wang, X. Multimodal sensing and therapeutic systems for wound healing and management: A review. Sens. Actuators Rep. 2022, 4, 100075. [Google Scholar] [CrossRef]
- Cutting, K.F.; White, R.J.; Legerstee, R. Evidence and practical wound care—An all-inclusive approach. Wound Med. 2017, 16, 40–45. [Google Scholar] [CrossRef]
- Deng, W.-J.; Wang, L.-F.; Dong, L.; Huang, Q.-A. LC Wireless Sensitive Pressure Sensors With Microstructured PDMS Dielectric Layers for Wound Monitoring. IEEE Sens. J. 2018, 18, 4886–4892. [Google Scholar] [CrossRef]
- Lu, L.; Zhao, N.; Liu, J.; Yang, B. Coupling piezoelectric and piezoresistive effects in flexible pressure sensors for human motion detection from zero to high frequency. J. Mater. Chem. C 2021, 9, 9309–9318. [Google Scholar] [CrossRef]
- Luo, Z.; Chen, J.; Zhu, Z.; Li, L.; Su, Y.; Tang, W.; Omisore, O.M.; Wang, L.; Li, H. High-Resolution and High-Sensitivity Flexible Capacitive Pressure Sensors Enhanced by a Transferable Electrode Array and a Micropillar–PVDF Film. ACS Appl. Mater. Interfaces 2021, 13, 7635–7649. [Google Scholar] [CrossRef] [PubMed]
- Messaoud, M.; Marsiquet, C.; Revol-Cavalier, F.; Rat, V.; Marchand, G. Flexible sensors for real-time monitoring of moisture levels in wound dressings. J. Wound Care 2018, 27, 385–391. [Google Scholar] [CrossRef]
- Cutting, K.F.; White, R.J. Maceration of the skin and wound bed 1: Its nature and causes. J. Wound Care 2002, 11, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Tessarolo, M.; Possanzini, L.; Gualandi, I.; Mariani, F.; Torchia, L.D.; Arcangeli, D.; Melandri, F.; Scavetta, E.; Fraboni, B. Wireless Textile Moisture Sensor for Wound Care. Front. Phys. 2021, 9, 722173. [Google Scholar] [CrossRef]
- Kiaee, G.; Mostafalu, P.; Samandari, M.; Sonkusale, S. A pH-mediated electronic wound dressing for controlled drug delivery. Adv. Healthc. Mater. 2018, 7, 1800396. [Google Scholar] [CrossRef] [PubMed]
- Gámez-Herrera, E.; García-Salinas, S.; Salido, S.; Sancho-Albero, M.; Andreu, V.; Perez, M.; Lujan, L.; Irusta, S.; Arruebo, M.; Mendoza, G. Drug-eluting wound dressings having sustained release of antimicrobial compounds. Eur. J. Pharm. Biopharm. 2020, 152, 327–339. [Google Scholar] [CrossRef]
- Maver, U.; Gradišnik, L.; Smrke, D.M.; Kleinschek, K.S.; Maver, T. Impact of growth factors on wound healing in polysaccharide blend thin films. Appl. Surf. Sci. 2019, 489, 485–493. [Google Scholar] [CrossRef]
- Ghomi, E.R.; Khalili, S.; Khorasani, S.N.; Neisiany, R.E.; Ramakrishna, S. Wound dressings: Current advances and future directions. J. Appl. Polym. Sci. 2019, 136, 47738. [Google Scholar] [CrossRef] [Green Version]
- Shah, J.B. The history of wound care. J. Am. Coll. Certif. Wound Spec. 2011, 3, 65–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallner, C.; Moormann, E.; Lulof, P.; Drysch, M.; Lehnhardt, M.; Behr, B. Burn Care in the Greek and Roman Antiquity. Medicina 2020, 56, 657. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, S.; Klar, A.S. Advanced Hydrogels as Wound Dressings. Biomolecules 2020, 10, 1169. [Google Scholar] [CrossRef] [PubMed]
- van Vlierberghe, S.; Graulus, G.-J.; Samal, S.K.; van Nieuwenhove, I.; Dubruel, P. Porous hydrogel biomedical foam scaffolds for tissue repair. In Biomedical Foams for Tissue Engineering Applications; Elsevier: Cambridge, UK, 2014; pp. 335–390. [Google Scholar] [CrossRef]
- Tang, Y.; Zhang, X.; Li, X.; Ma, C.; Chu, X.; Wang, L.; Xu, W. A review on recent advances of Protein-Polymer hydrogels. Eur. Polym. J. 2022, 162, 110881. [Google Scholar] [CrossRef]
- Ullah, F.; Othman, M.B.H.; Javed, F.; Ahmad, Z.; Akil, H.M. Classification, processing and application of hydrogels: A review. Mater. Sci. Eng. C 2015, 57, 414–433. [Google Scholar] [CrossRef]
- Jones, A.; Vaughan, D. Hydrogel dressings in the management of a variety of wound types: A review. J. Orthop. Nurs. 2005, 9, S1–S11. [Google Scholar] [CrossRef]
- Zhang, A.; Liu, Y.; Qin, D.; Sun, M.; Wang, T.; Chen, X. Research status of self-healing hydrogel for wound management: A review. Int. J. Biol. Macromol. 2020, 164, 2108–2123. [Google Scholar] [CrossRef] [PubMed]
- Sabzevari, R.; Roushandeh, A.M.; Mehdipour, A.; Alini, M.; Roudkenar, M.H. SA/G hydrogel containing hCAP-18/LL-37-engineered WJ-MSCs-derived conditioned medium promoted wound healing in rat model of excision injury. Life Sci. 2020, 261, 118381. [Google Scholar] [CrossRef] [PubMed]
- Atefyekta, S.; Blomstrand, E.; Rajasekharan, A.K.; Svensson, S.; Trobos, M.; Hong, J.; Webster, T.J.; Thomsen, P.; Andersson, M. Antimicrobial Peptide-Functionalized Mesoporous Hydrogels. ACS Biomater. Sci. Eng. 2021, 7, 1693–1702. [Google Scholar] [CrossRef]
- Afami, M.E.; el Karim, I.; About, I.; Coulter, S.M.; Laverty, G.; Lundy, F.T. Ultrashort Peptide Hydrogels Display Antimicrobial Activity and Enhance Angiogenic Growth Factor Release by Dental Pulp Stem/Stromal Cells. Materials 2021, 14, 2237. [Google Scholar] [CrossRef]
- Liang, J.-Y.; Li, Q.; Feng, L.B.; Hu, S.X.; Zhang, S.Q.; Li, C.X.; Zhang, X.B. Injectable antimicrobial hydrogels with antimicrobial peptide and sanguinarine controlled release ability for preventing bacterial infections. Am. J. Transl. Res. 2021, 13, 12614–12625. [Google Scholar] [PubMed]
- Sabzevari, R.; Roushandeh, A.M.; Alijani-Ghazyani, Z.; Jahanian-Najafabadi, A.; Roudkenar, M.H. SA/G hydrogel containing NRF2-engineered HEK-293-derived CM improves wound healing efficacy of WJ-MSCs in a rat model of excision injury. J. Tissue Viability 2021, 30, 527–536. [Google Scholar] [CrossRef]
- Soleimanpour, M.; Mirhaji, S.S.; Jafari, S.; Derakhshankhah, H.; Mamashli, F.; Nedaei, H.; Karimi, M.R.; Motasadizadeh, H.; Fatahi, Y.; Ghasemi, A.; et al. Designing a new alginate-fibrinogen biomaterial composite hydrogel for wound healing. Sci. Rep. 2022, 12, 7213. [Google Scholar] [CrossRef]
- Su, J.; Li, J.; Liang, J.; Zhang, K.; Li, J. Hydrogel Preparation Methods and Biomaterials for Wound Dressing. Life 2021, 11, 1016. [Google Scholar] [CrossRef] [PubMed]
- Haidari, H.; Bright, R.; Strudwick, X.L.; Garg, S.; Vasilev, K.; Cowin, A.J.; Kopecki, Z. Multifunctional ultrasmall AgNP hydrogel accelerates healing of S. aureus infected wounds. Acta Biomater. 2021, 128, 420–434. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Sun, X.; Wang, J.; Zhang, Y.; Dong, M.; Bu, T.; Li, L.; Liu, Y.; Wang, L. Multifunctional Injectable Hydrogel Dressings for Effectively Accelerating Wound Healing: Enhancing Biomineralization Strategy. Adv. Funct. Mater. 2021, 31, 2100093. [Google Scholar] [CrossRef]
- Cheng, H.; Shi, Z.; Yue, K.; Huang, X.; Xu, Y.; Gao, C.; Yao, Z.; Zhang, Y.S.; Wang, J. Sprayable hydrogel dressing accelerates wound healing with combined reactive oxygen species-scavenging and antibacterial abilities. Acta Biomater. 2021, 124, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Alven, S.; Nqoro, X.; Aderibigbe, B.A. Polymer-Based Materials Loaded with Curcumin for Wound Healing Applications. Polymers 2020, 12, 2286. [Google Scholar] [CrossRef] [PubMed]
- Shanmugapriya, K.; Kim, H.; Kang, H.W. Fucoidan-loaded hydrogels facilitates wound healing using photodynamic therapy by in vitro and in vivo evaluation. Carbohydr. Polym. 2020, 247, 116624. [Google Scholar] [CrossRef]
- Lu, K.; Li, K.; Zhang, M.; Fang, Z.; Wu, P.; Feng, L.; Deng, K.; Yu, C.; Deng, Y.; Xiao, Y.; et al. Adipose-derived stem cells (ADSCs) and platelet-rich plasma (PRP) loaded gelatin/silk fibroin hydrogels for improving healing in a murine pressure ulcer model. Chem. Eng. J. 2021, 424, 130429. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, Y.; Liu, H.; Ren, M.; Wang, Z.; Wang, X.; Liu, H.; Feng, Y.; Lin, Q.; Wang, C.; et al. pH-responsive hydrogel loaded with insulin as a bioactive dressing for enhancing diabetic wound healing. Mater. Des. 2021, 210, 110104. [Google Scholar] [CrossRef]
- Liu, P.; Jin, K.; Wong, W.; Wang, Y.; Liang, T.; He, M.; Li, H.; Lu, C.; Tang, X.; Zong, Y.; et al. Ionic liquid functionalized non-releasing antibacterial hydrogel dressing coupled with electrical stimulation for the promotion of diabetic wound healing. Chem. Eng. J. 2021, 415, 129025. [Google Scholar] [CrossRef]
- Lan, G.; Zhu, S.; Chen, D.; Zhang, H.; Zou, L.; Zeng, Y. Highly Adhesive Antibacterial Bioactive Composite Hydrogels With Controllable Flexibility and Swelling as Wound Dressing for Full-Thickness Skin Healing. Front. Bioeng. Biotechnol. 2021, 9, 785302. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Qi, J.; Hu, L.; Ouyang, D.; Wang, H.; Sun, Q.; Lin, L.; You, L.; Tang, B. A cannabidiol-containing alginate based hydrogel as novel multifunctional wound dressing for promoting wound healing. Mater. Sci. Eng. C 2021, 24, 112560. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wu, H.; Wang, H.; Zaldivar-Silva, D.; Agüero, L.; Liu, Y.; Zhang, Z.; Yin, Y.; Qiu, B.; Zhao, J.; et al. An injectable anti-microbial and adhesive hydrogel for the effective noncompressible visceral hemostasis and wound repair. Mater. Sci. Eng. C 2021, 129, 112422. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, J.; Yang, Y.; Shi, J.; Zhang, H.; Yao, X.; Chen, W.; Zhang, X. A rose bengal/graphene oxide/PVA hybrid hydrogel with enhanced mechanical properties and light-triggered antibacterial activity for wound treatment. Mater. Sci. Eng. C 2021, 118, 111447. [Google Scholar] [CrossRef]
- Xie, Y.-Y.; Zhang, Y.W.; Liu, X.Z.; Ma, X.F.; Qin, X.T.; Jia, S.R.; Zhong, C. Aggregation-induced emission-active amino acid/berberine hydrogels with enhanced photodynamic antibacterial and anti-biofilm activity. Chem. Eng. J. 2021, 413, 127542. [Google Scholar] [CrossRef]
- Ahmed, H.E.; Iqbal, Y.; Aziz, M.H.; Atif, M.; Batool, Z.; Hanif, A.; Yaqub, N.; Farooq, W.A.; Ahmad, S.; Fatehmulla, A.; et al. Green Synthesis of CeO2 Nanoparticles from the Abelmoschus esculentus Extract: Evaluation of Antioxidant, Anticancer, Antibacterial, and Wound-Healing Activities. Molecules 2021, 26, 4659. [Google Scholar] [CrossRef] [PubMed]
- Bloom, H. ’Cellophane’ Dressing for Second-Degree burns. Lancet 1945, 246, 559. [Google Scholar] [CrossRef]
- Bull, J.P.; Squire, J.R.; Topley, E. Experiments with occlusive dressings of a new plastic. Lancet 1948, 2, 213–215. [Google Scholar] [CrossRef]
- Meuleneire, F. A vapour-permeable film dressing used on superficial wounds. Br. J. Nurs. 2014, 23, S36–S43. [Google Scholar] [CrossRef] [PubMed]
- Kus, K.J.B.; Ruiz, E.S. Wound Dressings—A Practical Review. Curr. Dermatol. Rep. 2020, 9, 298–308. [Google Scholar] [CrossRef]
- Savina, I.N.; Zoughaib, M.; Yergeshov, A.A. Design and Assessment of Biodegradable Macroporous Cryogels as Advanced Tissue Engineering and Drug Carrying Materials. Gels 2021, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- Holloway, S.; Harding, K.G. Wound dressings. Surg. Oxf. 2022, 40, 25–32. [Google Scholar] [CrossRef]
- Sanchez, M.F.; Guzman, M.L.; Apas, A.L.; Alovero, F.d.; Olivera, M.E. Sustained dual release of ciprofloxacin and lidocaine from ionic exchange responding film based on alginate and hyaluronate for wound healing. Eur. J. Pharm. Sci. 2021, 161, 105789. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Nagate, T.; Matsuda, H. Acceleration of Wound Healing by Gelatin Film Dressings with Epidermal Growth Factor. J. Vet. Med. Sci. 2005, 67, 909–913. [Google Scholar] [CrossRef] [Green Version]
- Kamoun, E.A.; Kenawy, E.-R.S.; Chen, X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J. Adv. Res. 2017, 8, 217–233. [Google Scholar] [CrossRef] [PubMed]
- Rezvanian, M.; Ng, S.-F.; Alavi, T.; Ahmad, W. In-vivo evaluation of Alginate-Pectin hydrogel film loaded with Simvastatin for diabetic wound healing in Streptozotocin-induced diabetic rats. Int. J. Biol. Macromol. 2021, 171, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Eleroui, M.; Feki, A.; Hamzaoui, A.; Kammoun, I.; Bouhamed, M.; Boudawara, O.; Ayed, I.B.; Amara, I.B. Preparation and characterization of a novel hamada scoparia polysaccharide composite films and evaluation of their effect on cutaneous wound healing in rat. Int. J. Pharm. 2021, 608, 121056. [Google Scholar] [CrossRef] [PubMed]
- Arruda, I.R.S.; Souza, M.P.; Soares, P.A.; Albuquerque, P.B.; Silva, T.D.; Medeiros, P.L.; Silva, M.V.; Correia, M.T.S.; Vicente, A.A.; Carneiro-da-Cunha, M.G. Xyloglucan and Concanavalin A based dressings in the topical treatment of mice wound healing process. Carbohydr. Polym. Technol. Appl. 2021, 2, 100136. [Google Scholar] [CrossRef]
- Kausar, R.; Khan, A.; Jamil, B.; Shahzad, Y.; ul-Haq, I. Development and pharmacological evaluation of vancomycin loaded chitosan films. Carbohydr. Polym. 2021, 256, 117565. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Jiang, L.; Xu, C.; Kai, D.; Fan, X.; You, M.; Hui, C.M.; Wu, C.; Wu, Y.-L.; Li, Z. Engineered Janus amphipathic polymeric fiber films with unidirectional drainage and anti-adhesion abilities to accelerate wound healing. Chem. Eng. J. 2021, 421, 127725. [Google Scholar] [CrossRef]
- Mahmood, H.; Khan, I.U.; Asif, M.; Khan, R.U.; Asghar, S.; Khalid, I.; Khalid, S.H.; Irfan, M.; Rehman, F.; Shahzad, Y.; et al. In vitro and in vivo evaluation of gellan gum hydrogel films: Assessing the co impact of therapeutic oils and ofloxacin on wound healing. Int. J. Biol. Macromol. 2021, 166, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Severinov, D.A.; Lazarenko, S.V.; Sotnikov, K.A.; Pohozhay, V.V.; Ansimova, P.V.; Lipatov, V.A. In vitro Evaluation of Performance Properties of Sponge Hemostatic Dressings (Review). Sovrem. Tehnol. V Med. 2020, 12, 139. [Google Scholar] [CrossRef] [PubMed]
- Rojas Codina, S.; Herance, J.R.; Centeno, A.; Valero, J.; Arias, B.; Miquel, I.; Sanchez, P.; Rincon, E.; Lopez, R.; Murat, J. Nonclinical Evaluation of the New Topical Hemostatic Agent TT-173 for Skin Grafting Procedures. J. Burn Care Res. 2017, 38, e824–e833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartinger, J.M.; Lukáč, P.; Mitáš, P.; Mlček, M.; Popková, M.; Suchý, T.; Šupová, M.; Závora, J.; Adámková, V.; Benáková, H.; et al. Vancomycin-releasing cross-linked collagen sponges as wound dressings. Bosn. J. Basic Med. Sci. 2019, 21, 61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Zhao, W.; Dong, Z.; Ji, Y.; Li, M.; Hao, Y.; Zhang, D.; Yuan, C.; Deng, J.; Zhao, P.; et al. A biodegradable antibacterial alginate/carboxymethyl chitosan/Kangfuxin sponges for promoting blood coagulation and full-thickness wound healing. Int. J. Biol. Macromol. 2021, 167, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Wang, Y.; Qi, H.; Shi, C.; Wei, G.; Xiao, L.; Huang, Z.; Liu, S.; Yu, H.; Teng, C.; et al. Nanocomposite sponges of sodium alginate/graphene oxide/polyvinyl alcohol as potential wound dressing: In vitro and in vivo evaluation. Compos. Part B Eng. 2019, 167, 396–405. [Google Scholar] [CrossRef]
- Tamer, T.M.; Sabet, M.M.; Omer, A.M.; Abbas, E.; Eid, A.I.; Mohy-Eldin, M.S.; Hassan, M.A. Hemostatic and antibacterial PVA/Kaolin composite sponges loaded with penicillin–streptomycin for wound dressing applications. Sci. Rep. 2021, 11, 3428. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Jiang, L.; Wu, J.; Su, C.; Huang, C.; Liu, X.; Shao, W. Flexible Amoxicillin-Grafted Bacterial Cellulose Sponges for Wound Dressing: In Vitro and in Vivo Evaluation. ACS Appl. Mater. Interfaces 2018, 10, 5862–5870. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Lu, Y.; Gao, Y.; Cheng, S.; Tian, F.; Xiao, Y.; Li, F. Chitosan/alginate/hyaluronic acid polyelectrolyte composite sponges crosslinked with genipin for wound dressing application. Int. J. Biol. Macromol. 2021, 182, 512–523. [Google Scholar] [CrossRef]
- Yang, W.; Song, J.; Zhu, Y.; Ye, Z.; Wang, M.; Fang, Y.; Wu, W.; Chen, D.; Wang, Y. Application of chain-based sponge dressing for gunshot wounds in the groin. Am. J. Emerg. Med. 2021, 39, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Chen, C.; Zhang, H.; Chen, G.; Wang, Y.; Zhao, Y. Janus medical sponge dressings with anisotropic wettability for wound healing. Appl. Mater. Today 2021, 23, 101068. [Google Scholar] [CrossRef]
- Zheng, Z.; Li, M.; Shi, P.; Gao, Y.; Ma, J.; Li, Y.; Huang, L.; Yang, Z.; Yang, L. Polydopamine-modified collagen sponge scaffold as a novel dermal regeneration template with sustained release of platelet-rich plasma to accelerate skin repair: A one-step strategy. Bioact. Mater. 2021, 6, 2613–2628. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Luo, B.; Wu, Z.; Gu, B.; Xu, C.; Li, X.; Wang, X. Corn stalk/AgNPs modified chitin composite hemostatic sponge with high absorbency, rapid shape recovery and promoting wound healing ability. Chem. Eng. J. 2021, 421, 129815. [Google Scholar] [CrossRef]
- Bakr, R.O.; Amer, R.I.; Attia, D.; Abdelhafez, M.M.; Al-Mokaddem, A.K.; El-Gendy, A.E.N.G.; El-Fishawy, A.M.; Fayed, M.A.; Gad, S.S. In-vivo wound healing activity of a novel composite sponge loaded with mucilage and lipoidal matter of Hibiscus species. Biomed. Pharmacother. 2021, 135, 111225. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, J.; Si, Y.; Wang, X.; Deng, H.; Sheng, Z.; Li, Y.; Liu, J.; Zhao, J. A novel gene recombinant collagen hemostatic sponge with excellent biocompatibility and hemostatic effect. Int. J. Biol. Macromol. 2021, 178, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Wang, Q.; Zhang, Y.S.; Zhang, H.; Tang, Y.; Zhang, Y.; Zhang, W.; Zhang, X. A hemostatic sponge derived from skin secretion of Andrias davidianus and nanocellulose. Chem. Eng. J. 2021, 416, 129136. [Google Scholar] [CrossRef]
- Liu, X.; Xu, H.; Zhang, M.; Yu, D.-G. Electrospun Medicated Nanofibers for Wound Healing: Review. Membranes 2021, 11, 770. [Google Scholar] [CrossRef] [PubMed]
- Venkataprasanna, K.S.; Prakash, J.; Mathapati, S.S.; Bharath, G.; Banat, F.; Venkatasubbu, G.D. Development of chitosan/poly (vinyl alcohol)/graphene oxide loaded with vanadium doped titanium dioxide patch for visible light driven antibacterial activity and accelerated wound healing application. Int. J. Biol. Macromol. 2021, 193, 1430–1448. [Google Scholar] [CrossRef]
- Keshvardoostchokami, M.; Majidi, S.S.; Huo, P.; Ramachandran, R.; Chen, M.; Liu, B. Electrospun Nanofibers of Natural and Synthetic Polymers as Artificial Extracellular Matrix for Tissue Engineering. Nanomaterials 2020, 11, 21. [Google Scholar] [CrossRef]
- L-Jbour, N.D.A.; Beg, M.D.; Gimbun, J.; Alam, A.K.M.M. An Overview of Chitosan Nanofibers and their Applications in the Drug Delivery Process. Curr. Drug Deliv. 2019, 16, 272–294. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Chen, D.; Yang, H.; Lai, C.; Xuan, C.; Chen, Y.; Shi, X. Antibacterial peptide-modified collagen nanosheet for infected wound repair. Smart Mater. Med. 2021, 2, 172–181. [Google Scholar] [CrossRef]
- Salami, M.S.; Bahrami, G.; Arkan, E.; Izadi, Z.; Miraghaee, S.; Samadian, H. Co-electrospun nanofibrous mats loaded with bitter gourd (Momordica charantia) extract as the wound dressing materials: In Vitro and in vivo study. BMC Complement. Med. Ther. 2021, 21, 111. [Google Scholar] [CrossRef] [PubMed]
- Milanesi, G.; Vigani, B.; Rossi, S.; Sandri, G.; Mele, E. Chitosan-Coated Poly(lactic acid) Nanofibres Loaded with Essential Oils for Wound Healing. Polymers 2021, 13, 2582. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Zhou, M.; Dong, W.; Zhao, S.; Wang, Y.; Yao, J.; Liu, Z.; Han, H.; Sun, D.; Zhang, M. A bi-layered scaffold of a poly(lactic-co-glycolic acid) nanofiber mat and an alginate–gelatin hydrogel for wound healing. J. Mater. Chem. B 2021, 9, 7492–7505. [Google Scholar] [CrossRef]
- Bonferoni, M.C.; Rossi, S.; Sandri, G.; Caramella, C.; Del Fante, C.; Perotti, C.; Miele, D.; Vigani, B.; Ferrari, F. Bioactive Medications for the Delivery of Platelet Derivatives to Skin Wounds. Curr. Drug Deliv. 2019, 16, 472–483. [Google Scholar] [CrossRef] [PubMed]
- Lan, X.; Liu, Y.; Wang, Y.; Tian, F.; Miao, X.; Wang, H.; Tang, Y. Coaxial electrospun PVA/PCL nanofibers with dual release of tea polyphenols and ε-poly (L-lysine) as antioxidant and antibacterial wound dressing materials. Int. J. Pharm. 2021, 601, 120525. [Google Scholar] [CrossRef] [PubMed]
- Bacakova, L.; Zikmundova, M.; Pajorova, J.; Broz, A.; Filova, E.; Blanquer, A.; Matejka, R.; Stepanovska, J.; Mikes, P.; Jencova, V.; et al. Nanofibrous Scaffolds for Skin Tissue Engineering and Wound Healing Based on Synthetic Polymers. In Applications of Nanobiotechnology; Stoytcheva, M., Zlatev, M., Eds.; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef] [Green Version]
- Li, A.; Han, Z.; Li, Z.; Li, J.; Li, X.; Zhang, Z. A PTHrP-2 loaded adhesive cellulose acetate nanofiber mat as wound dressing accelerates wound healing. Mater. Des. 2021, 212, 110241. [Google Scholar] [CrossRef]
- Augustine, R.; Hasan, A.; Dalvi, Y.B.; Rehman, S.R.U.; Varghese, R.; Unni, R.N.; Yalcin, H.C.; Alfkey, R.; Thomas, S.; Al Moustafa, A.-E. Growth factor loaded in situ photocrosslinkable poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/gelatin methacryloyl hybrid patch for diabetic wound healing. Mater. Sci. Eng. C 2021, 118, 111519. [Google Scholar] [CrossRef] [PubMed]
- Mehwish, H.M.; Liu, G.; Rajoka, M.S.R.; Cai, H.; Zhong, J.; Song, X.; Xia, L.; Wang, M.; Aadil, R.M.; Inam-Ur-Raheem, M.; et al. Therapeutic potential of Moringa oleifera seed polysaccharide embedded silver nanoparticles in wound healing. Int. J. Biol. Macromol. 2021, 184, 144–158. [Google Scholar] [CrossRef]
- Ehsani, P.; Farahpour, M.R.; Mohammadi, M.; Mahmazi, S.; Jafarirad, S. Green fabrication of ZnO/magnetite-based nanocomposite—Using Salvia officinalis extract with antibacterial properties enhanced infected full-thickness wound. Colloids Surf. Physicochem. Eng. Asp. 2021, 628, 127362. [Google Scholar] [CrossRef]
- Andrabi, S.M.; Singh, P.; Majumder, S.; Kumar, A. A compositionally synergistic approach for the development of a multifunctional bilayer scaffold with antibacterial property for infected and chronic wounds. Chem. Eng. J. 2021, 423, 130219. [Google Scholar] [CrossRef]
- Beebe, M.; Gist, S.; Tio-Matos, I.; Cameron, S.; Falzgraf, S. Wound care in the geriatric client. Clin. Interv. Aging 2009, 4, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaganti, P.; Gordon, I.; Chao, J.H.; Zehtabchi, S. A systematic review of foam dressings for partial thickness burns. Am. J. Emerg. Med. 2019, 37, 1184–1190. [Google Scholar] [CrossRef] [PubMed]
- Fogh, K.; Nielsen, J. Clinical utility of foam dressings in wound management: A review. Chronic Wound Care Manag. Res. 2015, 2, 31. [Google Scholar] [CrossRef] [Green Version]
- He, M.; Ou, F.; Wu, Y.; Sun, X.; Chen, X.; Li, H.; Sun, D.; Zhang, L. Smart multi-layer PVA foam/CMC mesh dressing with integrated multi-functions for wound management and infection monitoring. Mater. Des. 2020, 194, 108913. [Google Scholar] [CrossRef]
- Xia, D.; Wang, X.; Wang, Y.; Wang, Y.; Meng, H.; Li, L.; Zhou, P.; Xu, S. Silver-decorated mesostructured cellular silica foams as excellent antibacterial hemostatic agents for rapid and effective treatment of hemorrhage. Mater. Sci. Eng. C 2020, 115, 111105. [Google Scholar] [CrossRef] [PubMed]
- Namviriyachote, N.; Muangman, P.; Chinaroonchai, K.; Chuntrasakul, C.; Ritthidej, G.C. Polyurethane-biomacromolecule combined foam dressing containing asiaticoside: Fabrication, characterization and clinical efficacy for traumatic dermal wound treatment. Int. J. Biol. Macromol. 2020, 143, 510–520. [Google Scholar] [CrossRef]
- Pietrangelo, L.; Magnifico, I.; Guerrera, A.; Cutuli, M.A.; Petronio, G.P.; Venditti, N.; Covelli, M.; Buccieri, N.; Garofalo, S.; Di Marco, R. LimpiAD foam and the potential control of the pressure ulcers onset. Biomed. Pharmacother. 2021, 144, 112327. [Google Scholar] [CrossRef]
- Jones, V.J. The use of gauze: Will it ever change? Int. Wound J. 2006, 3, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Chen, C.; Zhang, X.; Wen, X.; Xiao, Y.; Li, L.; Xu, Q.; Fu, F.; Diao, H.; Liu, X. Layer-by-layer coating of carboxymethyl chitosan-gelatin-alginate on cotton gauze for hemostasis and wound healing. Surf. Coat. Technol. 2021, 406, 126644. [Google Scholar] [CrossRef]
- Xiang, J.; Zhu, R.; Lang, S.; Yan, H.; Liu, G.; Peng, B. Mussel-inspired immobilization of zwitterionic silver nanoparticles toward antibacterial cotton gauze for promoting wound healing. Chem. Eng. J. 2021, 409, 128291. [Google Scholar] [CrossRef]
- Li, S.; Chen, A.; Chen, Y.; Yang, Y.; Zhang, Q.; Luo, S.; Ye, M.; Zhou, Y.; An, Y.; Huang, W.; et al. Lotus leaf inspired antiadhesive and antibacterial gauze for enhanced infected dermal wound regeneration. Chem. Eng. J. 2020, 402, 126202. [Google Scholar] [CrossRef]
- Valizadeh, R.; Hemmati, A.A.; Houshmand, G.; Bayat, S.; Bahadoram, M. Wound healing potential of Althaea officinalis flower mucilage in rabbit full thickness wounds. Asian Pac. J. Trop. Biomed. 2015, 5, 937–943. [Google Scholar] [CrossRef] [Green Version]
- Asumang, P.; Boakye, Y.D.; Agana, T.A.; Yakubu, J.; Entsie, P.; Akanwariwiak, W.G.; Adu, F.; Agyare, C. Antimicrobial, antioxidant and wound healing activities of methanol leaf extract of Bridelia micrantha (Hochst.) Baill. Sci. Afr. 2021, 14, e00980. [Google Scholar] [CrossRef]
- Kour, H.; Raina, R.; Verma, P.K.; Khan, A.M.; Bhat, M.A.; Nashiruddullah, N. Evaluation of the wound healing activity of ethanolic extract of Bergenia ciliata (Haw.) Sternb. rhizome with excision wound model in Wistar rats. J. Ethnopharmacol. 2021, 281, 114527. [Google Scholar] [CrossRef] [PubMed]
- Samrat, K.; Chandraprabha, M.N.; Krishna, R.H.; Sharath, R.; Harish, B.G. Biogenic synthesis of nano-sulfur using Punica granatum fruit peel extract with enhanced antimicrobial activities for accelerating wound healing. Nano Futures 2021, 5, 045003. [Google Scholar] [CrossRef]
- Li, M.; Zhao, Y.; Hao, H.; Han, W.; Fu, X. Mesenchymal stem cell-based therapy for nonhealing wounds: Today and tomorrow: Mesenchymal stem cell-based therapy. Wound Repair Regen. 2015, 23, 465–482. [Google Scholar] [CrossRef]
- Silina, E.; Manturova, N.; Stupin, V. Mesenchymal Stem Cells Application in Wound Tissue Healing in Old Animals. Stem Cells Cloning Adv. Appl. 2020, 13, 103–116. [Google Scholar] [CrossRef]
- Silina, E.V.; Manturova, N.E.; Litvitskiy, P.F.; Stupin, V.A. Comparative Analysis of the Effectiveness of Some Biological Injected Wound Healing Stimulators and Criteria for Its Evaluation. Drug Des. Devel. Ther. 2020, 14, 4869–4883. [Google Scholar] [CrossRef] [PubMed]
- Rudresha, G.V.; Manjuprasanna, V.N.; Urs, A.P.; Choudhury, M.; Rajaiah, R.; Vishwanath, B.S. Serine protease from Tricosanthus tricuspidata accelerates healing of Echis carinatus venom-induced necrotic wound. Toxicon 2020, 183, 1–10. [Google Scholar] [CrossRef]
- Das, S.; Baker, A.B. Biomaterials and Nanotherapeutics for Enhancing Skin Wound Healing. Front. Bioeng. Biotechnol. 2016, 4, 82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tabata, Y. Biomaterial technology for tissue engineering applications. J. R. Soc. Interface 2009, 6, S311–S324. [Google Scholar] [CrossRef] [Green Version]
- Bombin, A.D.J.; Dunne, N.J.; McCarthy, H.O. Electrospinning of natural polymers for the production of nanofibres for wound healing applications. Mater. Sci. Eng. C 2020, 114, 110994. [Google Scholar] [CrossRef]
- Herman, I.M.; Niranjan, P.; Grover, K. Protease Technology in Wound Repair. In Wound Healing, Tissue Repair, and Regeneration in Diabetes; Elsevier: Amsterdam, The Netherlands, 2020; pp. 357–391. [Google Scholar] [CrossRef]
- Kartini, K.; Wati, N.; Gustav, R.; Wahyuni, R.; Anggada, Y.F.; Hidayani, R.; Raharjo, A.; Islamie, R.; Putra, S.E.D. Wound healing effects of Plantago major extract and its chemical compounds in hyperglycemic rats. Food Biosci. 2021, 41, 100937. [Google Scholar] [CrossRef]
- Gharehpapagh, A.C.; Farahpour, M.R.; Jafarirad, S. The biological synthesis of gold/perlite nanocomposite using Urtica dioica extract and its chitosan-capped derivative for healing wounds infected with methicillin-resistant Staphylococcus aureus. Int. J. Biol. Macromol. 2021, 183, 447–456. [Google Scholar] [CrossRef]
- Manjuprasanna, V.N.; Rudresha, G.V.; Urs, A.P.; Gowda, M.D.M.; Rajaiah, R.; Vishwanath, B.S. Drupin, a cysteine protease from Ficus drupacea latex accelerates excision wound healing in mice. Int. J. Biol. Macromol. 2020, 165, 691–700. [Google Scholar] [CrossRef]
- Shoham, Y.; Shapira, E.; Haik, J.; Harats, M.; Egozi, D.; Robinson, D.; Kogan, L.; Elkhatib, R.; Telek, G.; Shalom, A. Bromelain-based enzymatic debridement of chronic wounds: Results of a multicentre randomized controlled trial. Wound Repair Regen. 2021, 29, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Urs, A.P.; Manjuprasanna, V.N.; Rudresha, G.V.; Hiremath, V.; Sharanappa, P.; Rajaiah, R.; Vishwanath, B.S. Thrombin-like serine protease, antiquorin from Euphorbia antiquorum latex induces platelet aggregation via PAR1-Akt/p38 signaling axis. Biochim. Biophys. Acta BBA-Mol. Cell Res. 2021, 1868, 118925. [Google Scholar] [CrossRef]
- Stewart, J.A.; McGrane, O.L.; Wedmore, I.S. Wound Care in the Wilderness: Is There Evidence for Honey? Wilderness Environ. Med. 2014, 25, 103–110. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.S.; Sinno, S.; Khachemoune, A. Honey and Wound Healing: An Overview. Am. J. Clin. Dermatol. 2011, 12, 181–190. [Google Scholar] [CrossRef]
- Akbik, D.; Ghadiri, M.; Chrzanowski, W.; Rohanizadeh, R. Curcumin as a wound healing agent. Life Sci. 2014, 116, 1–7. [Google Scholar] [CrossRef]
- Tejada, S.; Manayi, A.; Daglia, M.; Nabavi, F.S.; Sureda, A.; Hajheydari, Z.; Gortzi, O.; Pazoki-Toroudi, H.; Nabavi, M.S. Wound Healing Effects of Curcumin: A Short Review. Curr. Pharm. Biotechnol. 2016, 17, 1002–1007. [Google Scholar] [CrossRef] [PubMed]
- Surjushe, A.; Vasani, R.; Saple, D. Aloe vera: A short review. Indian J. Dermatol. 2008, 53, 163. [Google Scholar] [CrossRef] [PubMed]
- Boudreau, M.D.; Beland, F.A. An evaluation of the biological and toxicological properties of Aloe barbadensis (miller), Aloe vera. J. Environ. Sci. Health Part C 2006, 24, 103–154. [Google Scholar] [CrossRef] [Green Version]
- Cole, L.; Heard, C. Skin permeation enhancement potential of Aloe Vera and a proposed mechanism of action based upon size exclusion and pull effect. Int. J. Pharm. 2007, 333, 10–16. [Google Scholar] [CrossRef]
- Maan, P.; Yadav, K.S.; Yadav, N.P. Wound healing activity of Azadirachta indica A. juss stem bark in mice. Pharmacogn. Mag. 2017, 13, S316. [Google Scholar]
- Das, L.; Gunindro, N.; Ghosh, R.; Roy, M.; Debbarama, A. Mechanism of action of Azadirachta Indica Linn.(Neem) aqueous leaf extract as hypoglycaemic agent. Indian Med. Gaz. 2014, 1, 29–32. [Google Scholar]
- Rex, J.; Muthukumar, N.; Selvakumar, P. Phytochemicals as a potential source for anti-microbial, anti-oxidant and wound healing—A review. MOJ Biorg. Org. Chem. 2018, 2, 61–70. [Google Scholar]
- Mogoşanu, G.D.; Grumezescu, A.M. Natural and synthetic polymers for wounds and burns dressing. Improv. Wound Dress. Nov. Approaches 2014, 463, 127–136. [Google Scholar] [CrossRef]
- Karadeniz, F.; Sung, H.K.; Kim, H.S. Natural Origin Polymers: Applications as Wound Care Materials. J. Life Sci. 2019, 29, 382–393. [Google Scholar] [CrossRef]
- Pan, Z.; Ye, H.; Wu, D. Recent advances on polymeric hydrogels as wound dressings. APL Bioeng. 2021, 5, 011504. [Google Scholar] [CrossRef]
- Graça, M.F.P.; Miguel, S.P.; Cabral, C.S.D.; Correia, I.J. Hyaluronic acid—Based wound dressings: A review. Carbohydr. Polym. 2020, 241, 116364. [Google Scholar] [CrossRef] [PubMed]
- Arif, M.M.; Khan, S.M.; Gull, N.; Tabish, T.A.; Zia, S.; Khan, R.U.; Awais, S.M.; Butt, M.A. Polymer-based biomaterials for chronic wound management: Promises and challenges. Int. J. Pharm. 2021, 598, 120270. [Google Scholar] [CrossRef]





| Dressing Composition | Dressing Material Evaluated/ Group of Subjects | Key Findings | Reference |
|---|---|---|---|
| Gelatin methacrylate (GM), methacrylate of silk fibroin (MSF), stem cells (SC) and platelet concentrate plasma (PCP) | Gauze, GM/MSF, GM/MSF/PCP, GM/MSF/PCP/SC | Wound healing, reepithelialisation, and collagen deposition are all accelerated. | [60] |
| N-carboxyethyl chitosan, hyaluronic acid–aldehyde, insulin and adipic acid dihydrazide | Control, hydrogel, hydrogel + insulin | Expedited re-epithelialization and neovascularization; shortened inflammatory phase; increased granulation tissue formation; facilitated collagen deposition | [61] |
| Polymerized ionic liquid (PL), konjac glucomannan (K) and electrical stimulation (ES) | Control, PL-K-0, PL-K-5, PL-K-10, PL-K-20, ES, PL-K-ES-20 | It has great mechanical qualities and biocompatibility, and it disinfect quickly and effectively | [62] |
| Poly [2-(methacryloyloxy) ethyl] dimethyl-(3-sulfopropyl) ammonium hydroxide (SBMA), quaternized chitosan methacrylate (QCS) and Gelatin methacrylate (GelMA) | Control, SQG hydrogel | Improves granulation tissue development and collagen deposition by demonstrating good cell activity, hemocompatibility, and histocompatibility | [63] |
| Cannabidiol (CD), Zinc (Zn2+) ions and the alginate polymer (Alg) | Control group, Tegaderm™ group (3M), Alg@Zn group, CD/Alg@Zn group | Controlling of inflammatory infiltration, collagen deposition and granulation tissue production, and blood vessel formation | [64] |
| Gelatin meth acryloyl (GMa), Cerium oxide nanoparticles (CeNs) and an antimicrobial peptide (AMP) | GMa-Dopa, GMa-Dopa-AMP, GMa-Dopa-CeNs, GMa-Dopa-AMP-CeNs | Spray ability, adhesiveness, antibacterial activity, as well as the ability to scavenge ROS and regenerating skin are all promising | [57] |
| Gelatin (G), methacrylic anhydride (M), sodium tetraborate and oxidized dextran (BD) | GelMA/OD/Borax hydrogel | Efficiently stopped the bleeding, decreasing bioburden and hastened the healing of the wound | [65] |
| blank group, positive group (PBS) and G-M-BD | |||
| G-M-BD-L prepolymer solution, G-M-BD prepolymer solution | |||
| Rose Bengal (RB), graphene oxide (GO), polyvinyl Alcohol (PVA) hybrid hydrogel and chitosan microspheres | PVA, GO/PVA, β-GO/PVA, and β-GO/RB/PVA HDs | Biocompatibility and water-absorbing capability are desirable, as is an antimicrobial impact | [66] |
| N-(9-fluorenylmethoxy carbonyl)-L-phenylalanine (Fmoc-F) and berberine chloride (BBR) | Control, Fmoc-F/BBR, Fmoc-F/BBR + Light | Antibacterial and anti-biofilm action has been improved | [67] |
| Chitosan hydrogel membrane (CS), Cerium oxide nanoparticles (CeO2) from Abelmoschus esculentus extract | CS, CS-1% CeO2, CS-5% CeO2 | Showed excellent microbicidal, antioxidant activity and proven to accelerate healing time and closure | [68] |
| Dressing Composition | Dressing Material Evaluated/ Group of Subjects | Key Findings | Reference |
|---|---|---|---|
| Chitosan, alginate, hyaluronic acid, genipin | Medical gauze, CAHS1, CAHS2 and CAHS3 | Facilitates wound closure and haemostatic | [91] |
| Kang Fuxin (K), Alginate (AG) and carboxymethyl chitosan (CMC) | Control, AC, AG, ACK-5, ACK-10, ACK-15 | Good moisture transmission, plasticity, antimicrobial features, low cytotoxicity, and haemostatic | [87] |
| Chain based sponge dressing (CSD) | CSD and gauze | Achieved haemostasis quickly | [92] |
| Janus polyurethane, Superhydrophobic silica nanoparticles, super hydrophilic polyurethane (PU) | Saline, PU Sponge, Janus PU sponge | Reduced the risk of infection, excessive wetting and accelerated the efficiency of healing | [93] |
| Platelet rich plasma (PRP), collagen sponge scaffold (CSS) with modified polydopamine (PDA) | PDA-CSS-PRP, CSS-PRP, CSS and Normal Saline | Fast angiogenesis, rapid collagen arrangement leading to less scar development | [94] |
| Corn stalk (CS), silver nanoparticles (AgNPs) and chitin sponge (CH) | Control, polyvinyl formal sponge, CH-CS-AgNPs, CH-CS, CH | Biocompatibility and nontoxicity, fast wound closure rate | [95] |
| Chitosan and H. syriacus petroleum ether extract (SPC) | Control, Standard Mebo®, SPC-plain, SPC-low, SPC-medium, SPC-high | Perfect re-epithelization and epidermal remodelling | [96] |
| recombinant collagen (rCOL) | Implantation test with rCOL, COL | Perfect biocompatibility with no sensitivity, no toxicity, no stimulation reactions observed and excellent haemostatic effect | [97] |
| Saline, rCOL sponge extraction | |||
| Saline, Saline extraction, Solvent saline, Seasame oil Solvent extraction, non-polar solvent extraction | |||
| Andrias davidianus dermal secretion (ADDS)- nanocrystals of cellulose and nanofibers of cellulose (CS) sponge | Control, Gelatin sponge, ADDS-CS3, ADDS-CS2 | Excellent haemostatic properties | [98] |
| Dressing Composition | Dressing Material Evaluated/ Group of Subjects | Key Findings | Reference |
|---|---|---|---|
| Antimicrobial peptide KRWWKWWRRC (K), Collagen nanosheet (CN) | Blank, CN, KCN, | Good reepithelialisation, rapid wound closure and low inflammation | [103] |
| Poly (caprolactone) (PCL), Poly (vinyl alcohol) (PVA), collagen nanofibers (Col), Momordica charantia pulp extract (Ex) | Control, PCL/PVA/Col, PCL/PVA/Col/Ex 1%, PCL/PVA/Col/Ex 5%, PCL/PVA/Col/Ex 10% | Hemocompatible, cytocompatibility, and prevent bacterial penetration | [104] |
| Nanofiber mat of cellulose acetate (CA) loaded with parathyroid hormone related protein (Pthrp-2) | CA, CAP-1%, CAP-5% | Promoted epithelialization, collagen deposition and blood vessel formation | [110] |
| Moringa oleifera seed (MOS) polysaccharide (PS), nanocomposite with silver (AgNPs) | Distilled water, betadine ointment, MOS-PS-AgNPs-25, MOS-PS-AgNPs-50, MOS-PS-AgNPs-100 | Supports wound tightening and tissue generation as well | [112] |
| Salvia officinalis extract-assisted biosynthesis route to synthesize zinc oxide and Magnetite-based nanocomposites | mupirocin® ointment, magnetite ointment, zinc oxide/magnetite ointment, control | Granulation tissue, collagen density and epithelization improvements observed | [113] |
| Bilayer scaffold consists curcumin dextran nanoparticles (CDN), cerium oxide nanoparticles (CON) loaded Gelatin cryogel layer and polyvinyl alcohol-poly (vinylpyrrolidone)-iodine-potassium iodide layer (GCL) | Control, GCL, GCL-CDN-CON, Tegaderm pad commercial | Strong antioxidant, antibacterial and faster wound closure | [114] |
| Dressing Composition | Dressing Material Evaluated/ Group of Subjects | Key Findings | Reference |
|---|---|---|---|
| Mesostructured cellular silica foams (MCF) decorated with silver ions (Ag) | Control, MCF and MCF-Ag | MCF-Ag antibacterial haemostatic agent with splendid water absorption and antibacterial capacity | [119] |
| Polyurethane biomacromolecule combined foam (PUC), asiaticoside (AS), Silver nanoparticle (AgNPs) | PUC-AS-AgNPs, commercial gauze | shorter wound closure time, higher reepithelialisation and less pain score | [120] |
| Hyaluronic acid, a cell wall fragment of Cutibacterium acnes (LimpiAD) | LimpiAD foam | Prevented skin lesions or any sign of skin damage | [121] |
| Dressing Composition | Dressing Material Evaluated/ Group of Subjects | Key Findings | Reference |
|---|---|---|---|
| Gauze (G), Quat 188, silver nanoparticles (AgNPs), oxytetracyline hydrochloride (Ox) and salicyl-imine-chitosan biopolymer (SIC) | Gauze fabric, Dermazin Ointment, G/Ag NPs/Ox, G/Ag NPs/Ox/CS, fabrics/Ag NPs/Ox/SIC-0.2, G/Ag NPs/Ox/SIC-0.4, G/Ag NPs/Ox/SIC-0.6 | Promoting fibrosis and collagen reorganization | [25] |
| Cotton guaze (CG), chitosan (C), Gelatin (G) and alginate (A) | Control, CG and AGCCg-5 | High fluid absorption, excellent biocompatibility, hemocompatibility, haemostatic performance, low cost, reliability, safety, and a simple manufacturing process | [123] |
| Amino-modified cotton gauze (CG), poly (carboxybetaine-co-dopamine methacrylamide) (PCM) copolymer, silver nanoparticles (AgNPs) | Blank, Pristine CG, PCM@AgNPs-CG | Showed excellent hemocompatibility, cytocompatibility, reduced the inflammatory response and wound infection | [124] |
| Gauze, polydopamine, perfluorocarbon and silver nanoparticle (Lotus@Gauze) | Vaseline® petrolatum gauze, Atrauman®Ag gauze, Lotus@Gauze, Irradiated Vaseline® petrolatum gauze, Irradiated atrauman®Ag gauze, Irradiated Lotus@Gauze | Antiadhesive and antibacterial gauzes | [125] |
| Dressing Composition | Dressing Material Evaluated/ Group of Subjects | Key Findings | Reference |
| Bergenia ciliata rhizome ethanolic extract | Control, Povidine ointment, Bergenia ciliata, 5% (w/w) and 10% (w/w) ointments | Wound healing is faster and wound contraction is better. | [128] |
| Bridelia micrantha methanol leaf extract | Silver sulphadiazine cream, aqueous cream, 10% BME aqueous cream, 2.5% BME aqueous cream, 0.625% BME aqueous cream | Angiogenesis, collagenation, and re-epithelization all improved, as did antibacterial and antioxidant activities | [127] |
| Serine protease (Tricuspidin) from Tricosanthus tricuspidata | Tricuspidin & Trypsin | Excellent proteolytic ability, anti-inflammatory effect | [133] |
| Tricuspidin & PBS | |||
| Plantago major extract (PM), ursolic acid (UA) and oleanolic acid (OA) | Gel, Gel (Mebo), PM, UA, and OA gels | Non-toxic & improve wound healing | [138] |
| Urtica dioica extract, Chitosan (C), gold (G)/perlite nanocomposite ointment(P) | Control, mupirocin® ointment, P, PG, PGC | Decreasing the length of healing time and stimulates MRSA-infected wound regeneration | [139] |
| Cysteine protease (Drupin) from Ficus drupacea (Fd) latex | Saline, Neosporin, papain, Fd-protein rich fraction, drupin, drupin-IAA | Controlled expression of MMP’s, increased collagen production, cellular migration and proliferation | [140] |
| Bromelain based Escghar ex (ESX) | ESX group, Gel arm group | Good debridement activity of the formulation | [141] |
| Serine protease (SP), Antiquorin (Aq) Euphorbia antiquorum | Saline, Aq, SP + Aq | Improved platelet aggregation and quick haemostatis | [142] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rani Raju, N.; Silina, E.; Stupin, V.; Manturova, N.; Chidambaram, S.B.; Achar, R.R. Multifunctional and Smart Wound Dressings—A Review on Recent Research Advancements in Skin Regenerative Medicine. Pharmaceutics 2022, 14, 1574. https://doi.org/10.3390/pharmaceutics14081574
Rani Raju N, Silina E, Stupin V, Manturova N, Chidambaram SB, Achar RR. Multifunctional and Smart Wound Dressings—A Review on Recent Research Advancements in Skin Regenerative Medicine. Pharmaceutics. 2022; 14(8):1574. https://doi.org/10.3390/pharmaceutics14081574
Chicago/Turabian StyleRani Raju, Nithya, Ekaterina Silina, Victor Stupin, Natalia Manturova, Saravana Babu Chidambaram, and Raghu Ram Achar. 2022. "Multifunctional and Smart Wound Dressings—A Review on Recent Research Advancements in Skin Regenerative Medicine" Pharmaceutics 14, no. 8: 1574. https://doi.org/10.3390/pharmaceutics14081574