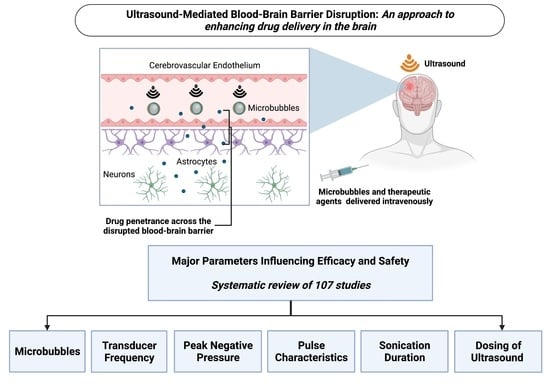
Ultrasound-Mediated Blood–Brain Barrier Disruption for Drug Delivery: A Systematic Review of Protocols, Efficacy, and Safety Outcomes from Preclinical and Clinical Studies
Abstract
:1. Introduction
1.1. The Blood–Brain Barrier and Drug Delivery
1.2. Ultrasound-Mediated Drug Delivery and the BBB
1.3. Challenges with Ultrasound-Mediated BBB Disruption
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Information Sources
2.3. Search Strategy
- ‘Ultrasound’ OR ‘focused ultrasound’ OR ‘MRI-guided ultrasound’ OR ‘MR-guided focused ultrasound’;
- ‘Blood brain barrier’ OR ‘BBB’;
- ‘Disruption’ OR ‘permeabilisation’ OR ‘permeabilization’ OR ‘opening’;
- ‘Drug delivery’.
2.4. Study Selection
2.5. Data Items and Collection Process
3. Results
3.1. Included Studies
3.2. Ultrasound Devices
3.2.1. Single-Element Ultrasound Devices
3.2.2. Multi-Element Ultrasound Devices
3.2.3. Diagnostic Ultrasound Devices
3.2.4. Implantable Ultrasound Devices
3.3. Methods for Assessing Successful BBB Disruption and Opening
3.3.1. MRI
3.3.2. Tracer Molecules
3.3.3. Therapeutic Agent Quantification
3.3.4. Comparing BBB Disruption between Studies
| In Vivo Subject | Study and Year Published | Assessments of BBB Disruption and Opening | |||||||
|---|---|---|---|---|---|---|---|---|---|
| MRI | Tracer Molecules | Quantified Therapeutic Uptake | |||||||
| EB | TB | Fl | FD | HRP | Antibodies | ||||
| Mouse | Baghirov et al., 2018 [38] | X | X (Polymeric nanoparticles) | ||||||
| Baseri et al., 2010 [73] | X | X | |||||||
| Bing et al., 2009 [39] | X | ||||||||
| Chen et al., 2013 [74] | X | ||||||||
| Chen et al., 2014 [67] | X | ||||||||
| Choi et al., 2010 [75] | X | ||||||||
| Choi et al., 2011 [76] | X | ||||||||
| Choi et al., 2011 [60] | X | X | |||||||
| Choi et al., 2008 [77] | X | ||||||||
| Choi et al., 2010 [68] | X | ||||||||
| Englander et al., 2021 [78] | X | X | X (Etoposide) | ||||||
| Jordao et al., 2013 [61] | X | X (Anti-endogenous IgG and IgM) | |||||||
| Kinoshita et al., 2006 [31] | X | X | X (Herceptin) | ||||||
| Kinoshita et al., 2006 [62] | X | X | X (Anti-D4 IgG) | ||||||
| Lapin et al., 2020 [79] | X | ||||||||
| Liu et al., 2014 [80] | X | X | X (Temozolomide) | ||||||
| McDannold et al., 2017 [81] | X | ||||||||
| McMahon et al., 2020 [59] | X | X | X (Anti-albumin IgG) | ||||||
| Morse et al., 2022 [82] | X (Fluorescently labelled, unloaded liposomes) | ||||||||
| Morse et al., 2019 [83] | X | X (Anti-albumin IgG) | |||||||
| Olumolade et al., 2016 [84] | X | ||||||||
| Omata et al., 2019 [85] | X | X | |||||||
| Raymond et al., 2007 [86] | X | X | |||||||
| Raymond et al., 2008 [87] | X | X | X | X (Anti-amyloid + anti-endogenous IgG) | |||||
| Samiotaki et al., 2012 [66] | X | ||||||||
| Shen et al., 2016 [69] | X | X (Fluorescently labelled, unloaded liposomes) | |||||||
| Sierra et al., 2017 [88] | X | X | |||||||
| Vlachos et al., 2011 [72] | X | ||||||||
| Wu et al., 2014 [89] | X (Liposomal doxorubicin) | ||||||||
| Zhang, D. et al., 2020 [43] | X | X (Paclitaxel—free and protein-bound) | |||||||
| Zhao, B. et al., 2018 [40] | X | ||||||||
| Rat | Ali et al., 2018 [90] | X | X | X (Doxorubicin) | |||||
| Aryal et al., 2017 [15] | X | X | |||||||
| Aryal et al., 2015 [91] | X | X | X (Doxorubicin) | ||||||
| Aryal et al., 2015 [70] | X | X | X (Doxorubicin) | ||||||
| Aslund et al., 2017 [92] | X | X (Pegylated macromolecule) | |||||||
| Cho et al., 2016 [14] | X | X | |||||||
| Chopra et al., 2010 [93] | X | ||||||||
| Fan et al., 2016 [64] | X | X (SPIO-labelled, doxorubicin-loaded microbubbles) | |||||||
| Fan et al., 2014 [45] | X | X (Carmustine loaded microbubbles) | |||||||
| Fan et al., 2015 [94] | X | X (Carmustine loaded microbubbles) | |||||||
| Goutal et al., 2018 [95] | X | X | |||||||
| Han et al., 2021 [96] | X | ||||||||
| Huh et al., 2020 [97] | X | ||||||||
| Jung et al., 2019 [98] | X | X | X (Doxorubicin) | ||||||
| Kobus et al., 2016 [99] | X | ||||||||
| Kovacs et al., 2017 [27] | X | X (Anti-albumin IgG) | |||||||
| Kovacs et al., 2018 [100] | X | ||||||||
| Liu et al., 2009 [65] | X | X | |||||||
| Liu et al., 2010 [101] | X | X | X (Carmustine) | ||||||
| Liu et al., 2010 [102] | X | ||||||||
| Liu et al., 2008 [32] | X | X | |||||||
| Liu et al., 2010 [103] | X | ||||||||
| Marty et al., 2012 [58] | X | ||||||||
| McDannold et al., 2019 [48] | X | X (Carboplatin) | |||||||
| McDannold et al., 2020 [49] | X | X (Irinotecan and SN-38) | |||||||
| McDannold et al., 2011 [104] | X | X | |||||||
| Mcmahon et al., 2017 [26] | X | ||||||||
| Mcmahon et al., 2020 [105] | X | X | |||||||
| Mcmahon et al., 2020 [106] | X | ||||||||
| O’Reilly et al., 2017 [107] | X | ||||||||
| O’Reilly et al., 2011 [108] | X | ||||||||
| Park et al., 2017 [109] | X | X | X (Doxorubicin) | ||||||
| Park et al., 2012 [71] | X | X | X (Doxorubicin) | ||||||
| Shin et al., 2018 [19] | X | ||||||||
| Song et al., 2017 [110] | X | ||||||||
| Treat et al., 2007 [63] | X | X | X (Doxorubicin) | ||||||
| Tsai et al., 2018 [33] | X | ||||||||
| Wei et al., 2013 [111] | X | X | X (Temozolomide) | ||||||
| Wu et al., 2017 [112] | X | X | |||||||
| Yang et al., 2013 [113] | X | X | |||||||
| Yang et al., 2014 [114] | X | X | |||||||
| Yang et al., 2012 [34] | X | X | |||||||
| Yang et al., 2011 [115] | X | X | |||||||
| Yang et al., 2012 [116] | X | X | |||||||
| Zhang, Y. et al., 2016 [117] | X | ||||||||
| Rabbit | Beccaria et al., 2013 [20] | X | X | ||||||
| Chopra et al., 2010 [93] | X | ||||||||
| Hynyen et al., 2005 [28] | X | X | |||||||
| Hynyen et al., 2006 [30] | X | X | |||||||
| McDannold et al., 2006 [118] | X | ||||||||
| McDannold et al., 2007 [25] | X | ||||||||
| McDannold et al., 2008 [119] | X | ||||||||
| McDannold et al., 2008 [120] | X | ||||||||
| Mei et al., 2009 [121] | X | X | X (Methotrexate) | ||||||
| Wang et al., 2009 [122] | X | X | |||||||
| Dog | O’Reilly et al., 2017 [123] | X | |||||||
| Pig | Liu et al., 2011 [23] | X | X | X (SPIO nanoparticles) | |||||
| Sheep | Pelekanos et al., 2018 [29] | X | X (Anti-endogenous IgG) | ||||||
| Yoon et al., 2019 [124] | X | ||||||||
| Non-Human Primate (NHP) | Arvantis et al., 2012 [50] | X | |||||||
| Downs et al., 2015 [21,22] | X | ||||||||
| Goldwirt et al., 2016 [44] | X | X (Carboplatin) | |||||||
| Horodyckid et al., 2017 [56] | X | ||||||||
| Marquet et al., 2014 [125] | X | ||||||||
| Marquet et al., 2011 [24] | X | ||||||||
| McDannold et al., 2012 [126] | X | X | |||||||
| Pouliopoulos et al., 2019 [57] | X | ||||||||
| Wu et al., 2016 [127] | X | ||||||||
| Human | Abrahao et al., 2019 [8] | X | |||||||
| Anastasiadis et al., 2021 [51] | X | X | |||||||
| Chen et al., 2021 [54] | X | ||||||||
| Gasca-Salas et al., 2021 [52] | X | ||||||||
| Idbaidh et al., 2019 [9] | X | ||||||||
| Lipsman et al., 2018 [10] | X | ||||||||
| Mainprize et al., 2019 [11] | X | X (Liposomal doxorubicin and temozolomide) | |||||||
| Park et al., 2020 [53] | X | ||||||||
3.4. Methods of Assessing Safety Outcomes
3.4.1. Macroscopic Assessments
3.4.2. Histological Assessments
3.4.3. Biochemical Assessments
3.4.4. Electrophysiological Assessments
3.4.5. Physical and Behavioural Assessments
| In Vivo Subject | Study Reference | Safety Assessments | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Macroscopic | Histological | Biochemical | Electrophysiological | Physical/Behvaioural | |||||||||||||
| MRI | PET | ΔT | Gross | H/E | TUNEL | VF | LB | CV | PB | GFAP | Iba1 | Other | |||||
| Mouse | [38] | X | X | ||||||||||||||
| [73] | X | X | X | ||||||||||||||
| [39] | X | ||||||||||||||||
| [74] | X | ||||||||||||||||
| [67] | X | ||||||||||||||||
| [75] | X | ||||||||||||||||
| [76] | X | ||||||||||||||||
| [60] | X | X | |||||||||||||||
| [77] | X | X | |||||||||||||||
| [68] | X | ||||||||||||||||
| [78] | X | X | X | ||||||||||||||
| [61] | X | X | X | X (PCR + WB) | |||||||||||||
| [31] | X | X | X | X | |||||||||||||
| [62] | X | X | |||||||||||||||
| [79] | X | ||||||||||||||||
| [80] | X | ||||||||||||||||
| [81] | X | ||||||||||||||||
| [59] | X | X | |||||||||||||||
| [82] | X | ||||||||||||||||
| [83] | X | ||||||||||||||||
| [84] | X | X | X | X | |||||||||||||
| [85] | X | X | |||||||||||||||
| [86] | X | X | X | ||||||||||||||
| [87] | X | ||||||||||||||||
| [66] | X | ||||||||||||||||
| [69] | X | ||||||||||||||||
| [88] | X | X | X | ||||||||||||||
| [72] | X | X | |||||||||||||||
| [89] | X | X | X | ||||||||||||||
| [43] | X | ||||||||||||||||
| [40] | X | X | |||||||||||||||
| Rat | [90] | X | X | X | |||||||||||||
| [15] | X | X | |||||||||||||||
| [91] | X | X | |||||||||||||||
| [70] | X | X | |||||||||||||||
| [92] | X | X | |||||||||||||||
| [14] | X | X | X | ||||||||||||||
| [93] | X | X | |||||||||||||||
| [64] | X | ||||||||||||||||
| [45] | X | X | X | ||||||||||||||
| [94] | X | ||||||||||||||||
| [95] | X | X | |||||||||||||||
| [96] | X | X | X | X | X (AQP-4) | ||||||||||||
| [97] | X | ||||||||||||||||
| [98] | X | X | X | ||||||||||||||
| [99] | X | X | X | ||||||||||||||
| [27] | X | X | X | X | X | X (ELISA, PCR, WB) | |||||||||||
| [100] | X | X | X | X | X | X (WB) | |||||||||||
| [65] | X | X | X | X | |||||||||||||
| [101] | X | X | |||||||||||||||
| [102] | X | ||||||||||||||||
| [32] | X | X | X | ||||||||||||||
| [103] | X | X | X | ||||||||||||||
| [58] | X | ||||||||||||||||
| [48] | X | X | X | ||||||||||||||
| [49] | X | X | |||||||||||||||
| [104] | X | X | |||||||||||||||
| [26] | X | X | X (PCR) | ||||||||||||||
| [105] | X | X | X (PCR) | ||||||||||||||
| [106] | X | X | X | X (ELISA) | |||||||||||||
| [107] | X | X | |||||||||||||||
| [108] | X | X | |||||||||||||||
| [109] | X | X | |||||||||||||||
| [71] | X | X | |||||||||||||||
| [19] | X | X | |||||||||||||||
| [110] | X | X | |||||||||||||||
| [63] | X | X | |||||||||||||||
| [33] | X | X | X | X (Plasma fibrinogen) | X | ||||||||||||
| [111] | X | X | |||||||||||||||
| [112] | X | X | |||||||||||||||
| [113] | X | ||||||||||||||||
| [114] | X | ||||||||||||||||
| [34] | X | X | |||||||||||||||
| [115] | X | ||||||||||||||||
| [116] | X | X | |||||||||||||||
| [117] | X | X | X | ||||||||||||||
| Rabbit | [20] | X | X | ||||||||||||||
| [93] | X | X | |||||||||||||||
| [28] | X | X | X | ||||||||||||||
| [30] | X | X | X | ||||||||||||||
| [118] | X | ||||||||||||||||
| [25] | X | X | |||||||||||||||
| [119] | X | ||||||||||||||||
| [120] | X | ||||||||||||||||
| [121] | X | ||||||||||||||||
| [122] | X | ||||||||||||||||
| Dog | [123] | X | X | X | X | X | |||||||||||
| Pig | [23] | X | X | X | |||||||||||||
| Sheep | [29] | X | X | X | X | X | |||||||||||
| [124] | X | X | X | X | |||||||||||||
| NHP | [50] | X | X | X | |||||||||||||
| [21,22] | X | X (EMG) | X | ||||||||||||||
| [44] | X | ||||||||||||||||
| [56] | X | X | X | X | X | X (EEG, SSEP) | X | ||||||||||
| [125] | X | ||||||||||||||||
| [24] | X | X | |||||||||||||||
| [126] | X | X | X | X | X | X | |||||||||||
| [57] | X | ||||||||||||||||
| [127] | X | ||||||||||||||||
| Human | [8] | X | X | X (EEG) | X | ||||||||||||
| [51] | X | X | X | X | |||||||||||||
| [54] | X | X | X | ||||||||||||||
| [52] | X | X | X | X | |||||||||||||
| [9] | X | X | |||||||||||||||
| [10] | X | X | X | X | |||||||||||||
| [11] | X | X | |||||||||||||||
| [53] | X | X | |||||||||||||||
3.5. Parameters Influencing Ultrasound-Mediated BBB Disruption
3.5.1. Microbubbles
Microbubble Characteristics
Microbubble Dosing
Timing and Method of Microbubble Administration
| Agent | Manufacturer | Shell Composition | Gas Core Composition | Mean Bubble Diameter (µm) | Bubble Concentration (Bubbles/mL) | Use in Identified Studies |
|---|---|---|---|---|---|---|
| Definity®/Luminity® | Lantheus Medical Imaging | Lipid | C3F8 | 1.1–3.3 | 1.2 × 1010 | Used in n = 42 preclinical studies (typical doses: 10–20 µL/kg) and n = 6 clinical studies (typical dose: 4 µL/kg) |
| Optison® | GE Healthcare | Protein | C3F8 | 3.0–4.5 | 5–8 × 108 | Used in n = 14 preclinical studies (typical doses: 50–100 µL/kg but significantly varied in mice studies) |
| SonoVue®/Lumason® | Bracco Diagnostics | Lipid | SF6 | 1.5–2.5 | 1.5–5.6 × 108 | Used in n = 29 preclinical studies (typical doses 25–150 µL/kg) and n = 2 clinical studies (typical dose: 100 µL/kg) |
| Usphere Prime® | Trust Bio-sonics | Lipid | C3F8 | 1.0 | 2.8 × 1010 | Used in n = 1 preclinical study |
| Sonazoid® | GE Healthcare | Lipid | C4F10 | 2.0–3.0 | 9 × 108 | Used in n = 1 preclinical study |
3.5.2. Transducer Frequency
3.5.3. Peak Negative Pressure
3.5.4. Pulse Characteristics
Pulse Length
Phasic vs. Tonic Pulses
Pulse Repetition Frequency
3.5.5. Sonication Duration
3.5.6. Dosing (Number and Frequency) of Ultrasound Applications
Ultrasound Sonications
Ultrasound Sessions
4. Discussion
5. Conclusions
| Parameter | Safe and Effective Parameters Commonly Used | Parameters Compared | Reported Effects on BBBD (Efficacy Outcomes) | Reported Safety Outcomes |
|---|---|---|---|---|
| Transducer Frequency | Preclinical: 0.20–1.50 MHz Clinical: 0.22, 0.50, and 1.05 MHz | 0.26, 0.69, 1.63, 2.04 MHz [119] | Increasing frequency: greater PNP required to achieve BBBD [19,45,94,119]; smaller foci/area of BBBD [19,45,94] | Increasing frequency: increased density of microhaemorrhagic activity [119]; decreased haemorrhagic [19,45,94] and oedematous activity [45] |
| 1 and 10 MHz [45,94] | ||||
| 0.5 and 1.6 MHz [19] | ||||
| PNP | Preclinical: 0.2–0.5 MPa with <1 MHz transducers Clinical: 0.48–1.15 MPa and 2.5–60 W power | 0.30, 0.46, 0.61, 0.75, 0.98 MPa [73] | Increasing PNP: increasing BBBD after surpassing threshold PNP [9,15,45,65,73]; eventual saturation point in BBBD [32]; prolonged BBB opening [65,66]; prolonged P-glycoprotein downregulation [15] | Increasing PNP: increased haemorrhagic [15,19,65,66,73,93] and microhaemorrhagic change [73,93]; neuropil loss; neuronal loss [73,93] and necrosis [93]; evidence of apoptosis [45]; cerebral oedema [9,45]; hypoactivity/ataxia/tremor [33] |
| 0.55, 0.81 MPa [15] | ||||
| 0.27, 0.39, 0.59, 0.78 MPa [93] | ||||
| 0.3, 0.5, 1.0, 1.5, 2.0, 2.5, 4.5 MPa [45,94] | ||||
| 1.1, 1.9, 2.45, and 3.5 MPa [65] | ||||
| 0.45, 0.62, 0.98, 1.32 MPa [101] | ||||
| 0.55, 0.78, 1.1, 1.9, 2.45, 3.47, 4.9 MPa [32] | ||||
| 0.2, 0.3, 0.6, 1.5 MPa [19] | ||||
| 0.30, 0.51, 0.89 MPa [33] | ||||
| 0.4, 0.5 0.8, 1.1, 1.4, 2.3, 3.1 MPa [28] | ||||
| 0.2, 0.4, 0.5, 0.8, 1.1, 1.8 MPa [25] | ||||
| 0.78, 0.90, 1.03, 1.15 MPa [9] | ||||
| PL | Preclinical: 10 ms Clinical: 2–3, 10, and 23.6 ms | 0.1, 0.2, 1.0, 2.0, 10, 20, 30 ms [76] | Increasing PL: increasing BBBD with PL 0.1–10 ms; statistically non-significant increase in BBBD after PL > 10 ms [7,76]; decreased PNP threshold (PL = 0.1–10 ms) [120]; heterogeneous distribution of BBBD/greater perivascular accumulation of tracer [76] | Increasing PL: no microhaemorrhagic change with PL ≤ 10 ms [19,120]; significant haemorrhagic change with PL = 100 ms [19,23]; evidence of apoptosis with PL = 100 ms [23] |
| 1, 10, 100 ms [19] | ||||
| 10, 100 ms [7] | ||||
| 0.1, 1, 10 ms [120] | ||||
| 30, 100 ms [23] | ||||
| 10, 50 and 100 ms [102] | ||||
| PRF | Preclinical: 1–10 Hz Clinical: 1–10 Hz and 30–31 Hz | 0.1, 1, 1, 10, 25 Hz[76] | Increasing PRF: no BBBD with PRF = 0.1 Hz [76]; inconsistent improvements in BBBD with tonic pulsed sequences, some being statistically significant [19] and others not [76,120]; improvements in BBBD with rapid, phasic pulses [60,108] | Increasing PRF: no increase in adverse safety outcomes, via MRI [108] and histology [19,60,76,108,120] |
| 0.5, 1, 2, 5 Hz [120] | ||||
| 1, 2, 5 Hz [19] | ||||
| 1, 1667, 3333, 16,667, 166,667 Hz [108] | ||||
| 6250, 25,000, 100,000 Hz [60] | ||||
| SD | Preclinical: 30–120 sClinical: 30–120 s; 150–270 s in one study | 30, 660 s [76] | Increasing SD: improved BBBD with pulsed [19,93,102] and continuously [40,121] applied US; plateauing effect thereafter [93,115]; one study reported no improvement in BBBD [76] | Increasing SD: minimal change in adverse safety outcomes with small increases, and significantly worsening safety outcomes with excessive increases [19,40,93,102,121]; no increase in histopathological outcomes in one study [76] |
| 240, 360, 480, 600 s [102] | ||||
| 30, 60, 120, 300 s [19] | ||||
| 30, 180, 300, 600, 1200 s [93] | ||||
| 60, 120, 180, 240 s[40] | ||||
| 6, 8 and 10 s [121] | ||||
| Dosing (Number and Frequency) of Sonications | Preclinical: 1–13 sonications/session (ISI = 5 min) Clinical: 1–8 sonications/session (ISI not stated) ISIs are listed within brackets | 1, 2 (10 min), 2 (120 min) sonications [71] | Increasing sonication #: increase in BBBD [71,115]; improved doxorubicin uptake with shorter ISI [71] | Increasing sonication #: no [71] or mild [115] histopathological change (increased neuropil vacuolation) |
| 1, 2 (20 min), 2 (40 min) sonications [115] | ||||
| Dosing (Number and Frequency) of Sessions | Preclinical: 1–27 Clinical: 1–10 sessions Intersession intervals are listed within brackets | 2–10, 2–6 sessions (biweekly and monthly) [84] | Increasing session #: higher PNP sonications required to achieve similar BBBD, but likely due to animal model growth [84] as not observed in developed adult clinical trials [9,11] | Increasing session #: no adverse safety outcomes [53,123]; transient MRI changes [9,21,22]; cortical atrophy, ventricular dilation, and lesion formation on MRI [100]; increased phosphorylated tau deposition [100]; increased neurogenesis [100] no change in motor and behavioural outcomes in rodents [84]; increased tissue damage and macrophage infiltration with doxorubicin co-delivery [70,91]; increasing number of apoptotic cells (significantly larger microbubble dose) [33]; mild increase in white matter vacuolation and mild neuronal injury (significantly larger microbubble dose) [43] |
| 1, 8 (3 days) sessions [43] | ||||
| 1, 4 (weekly) sessions [123] | ||||
| 1, 6 (weekly) sessions [100] | ||||
| 1, 3 (weekly) sessions [93] | ||||
| 1, 3 (weekly) sessions [70,91] | ||||
| 1, 2 (2 days), 3 (2 days) sessions [33] | ||||
| 3 (monthly), 6 (monthly) sessions [53] | ||||
| 4–27 (varying intersession intervals) sessions [21,22] | ||||
| 1–10 (monthly) sessions [9] |
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ballabh, P.; Braun, A.; Nedergaard, M. The blood-brain barrier: An overview: Structure, regulation, and clinical implications. Neurobiol. Dis. 2004, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Vykhodtseva, N.; McDannold, N.; Hynynen, K. Progress and problems in the application of focused ultrasound for blood-brain barrier disruption. Ultrasonics 2008, 48, 279–296. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Drug and gene targeting to the brain with molecular Trojan horses. Nat. Rev. Drug Discov. 2002, 1, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Mackay, S.M.; Myint, M.A.; Easingwood, R.A.; Hegh, D.Y.; Wickens, J.R.; Hyland, B.I.; Jameson, G.N.L.; Reynolds, J.N.J.; Tan, E.W. Dynamic control of neurochemical release with ultrasonically-sensitive nanoshell-tethered liposomes. Commun. Chem. 2019, 2, 122. [Google Scholar] [CrossRef]
- Nakano, T.; Rizwan, S.B.; Myint, D.M.; Gray, J.; Mackay, S.M.; Harris, P.; Perk, C.G.; Hyland, B.I.; Empson, R.; Tan, E.W.; et al. An On-Demand Drug Delivery System for Control of Epileptiform Seizures. Pharmaceutics 2022, 14, 468. [Google Scholar] [CrossRef]
- Bakay, L.; Ballantine, T., Jr.; Hueter, T.F.; Sosa, D. Ultrasonically produced changes in the blood-brain barrier. AMA Arch. Neurol. Psychiatry 1956, 76, 457–467. [Google Scholar] [CrossRef]
- Hynynen, K.; McDannold, N.; Vykhodtseva, N.; Jolesz, F.A. Jolesz. Noninvasive MR imaging-guided focal opening of the blood-brain barrier in rabbits. Radiology 2001, 220, 640–646. [Google Scholar] [CrossRef]
- Abrahao, A.; Meng, Y.; Llinas, M.; Huang, Y.; Hamani, C.; Mainprize, T.; Aubert, I.; Heyn, C.; Black, S.E.; Hynynen, K.; et al. First-in-human trial of blood-brain barrier opening in amyotrophic lateral sclerosis using MR-guided focused ultrasound. Nat. Commun. 2019, 10, 4373. [Google Scholar] [CrossRef]
- Idbaih, A.; Canney, M.; Belin, L.; Desseaux, C.; Vignot, A.; Bouchoux, G.; Asquier, N.; Law-Ye, B.; Leclercq, D.; Bissery, A.; et al. Safety and Feasibility of Repeated and Transient Blood-Brain Barrier Disruption by Pulsed Ultrasound in Patients with Recurrent Glioblastoma. Clin. Cancer Res. 2019, 25, 3793–3801. [Google Scholar] [CrossRef]
- Lipsman, N.; Meng, Y.; Bethune, A.J.; Huang, Y.; Lam, B.; Masellis, M.; Herrmann, N.; Heyn, C.; Aubert, I.; Boutet, A.; et al. Blood-brain barrier opening in Alzheimer’s disease using MR-guided focused ultrasound. Nat. Commun. 2018, 9, 2336. [Google Scholar] [CrossRef]
- Mainprize, T.; Lipsman, N.; Huang, Y.; Meng, Y.; Bethune, A.; Ironside, S.; Heyn, C.; Alkins, R.; Trudeau, M.; Sahgal, A.; et al. Blood-Brain Barrier Opening in Primary Brain Tumors with Non-invasive MR-Guided Focused Ultrasound: A Clinical Safety and Feasibility Study. Sci. Rep. 2019, 9, 321. [Google Scholar] [CrossRef] [PubMed]
- Sheikov, N.; McDannold, N.; Sharma, S.; Hynynen, K. Effect of focused ultrasound applied with an ultrasound contrast agent on the tight junctional integrity of the brain microvascular endothelium. Ultrasound Med. Biol. 2008, 34, 1093–1104. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Lee, E.H.; Han, M.; An, S.H.; Park, J. Diminished Expression of P-glycoprotein Using Focused Ultrasound Is Associated With JNK-Dependent Signaling Pathway in Cerebral Blood Vessels. Front. Neurosci. 2019, 13, 1350. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.S.; Lee, H.; Han, M.; Choi, J.R.; Lee, T.; Ahn, S.; Chang, Y.; Park, J. Localised down-regulation of p-glycoprotein by Focused Ultrasound and Microbubbles induced Blood-Brain Barrier Disruption in Rat Brain. J. Ther. Ultrasound 2016, 5 (Suppl. S1), 16–17. [Google Scholar]
- Aryal, M.; Fischer, K.; Gentile, C.; Gitto, S.; Zhang, Y.Z.; McDannold, N. Effects on P-Glycoprotein Expression after Blood-Brain Barrier Disruption Using Focused Ultrasound and Microbubbles. PLoS ONE 2017, 12, e0166061. [Google Scholar] [CrossRef]
- Sheikov, N.; McDannold, N.; Jolesz, F.; Zhang, Y.Z.; Tam, K.; Hynynen, K. Brain arterioles show more active vesicular transport of blood-borne tracer molecules than capillaries and venules after focused ultrasound-evoked opening of the blood-brain barrier. Ultrasound Med. Biol. 2006, 32, 1399–1409. [Google Scholar] [CrossRef]
- McMahon, D.; Poon, C.; Hynynen, K. Evaluating the safety profile of focused ultrasound and microbubble-mediated treatments to increase blood-brain barrier permeability. Expert Opin. Drug Deliv. 2019, 16, 129–142. [Google Scholar] [CrossRef]
- Aryal, M.; Arvanitis, C.D.; Alexander, P.M.; McDannold, N. Ultrasound-mediated blood-brain barrier disruption for targeted drug delivery in the central nervous system. Adv. Drug Deliv. Rev. 2014, 72, 94–109. [Google Scholar] [CrossRef]
- Shin, J.; Kong, C.; Cho, J.S.; Lee, J.; Koh, C.S.; Yoon, M.S.; Na, Y.C.; Chang, W.S.; Chang, J.W. Focused ultrasound-mediated noninvasive blood-brain barrier modulation: Preclinical examination of efficacy and safety in various sonication parameters. Neurosurg. Focus 2018, 44, E15. [Google Scholar] [CrossRef]
- Beccaria, K.; Canney, M.; Goldwirt, L.; Fernandez, C.; Adam, C.; Piquet, J.; Autret, G.; Clement, O.; Lafon, C.; Chapelon, J.-Y.; et al. Opening of the blood-brain barrier with an unfocused ultrasound device in rabbits. J. Neurosurg. 2013, 119, 887–898. [Google Scholar] [CrossRef]
- Downs, M.E.; Buch, A.; Karakatsani, M.E.; Konofagou, E.E.; Ferrera, V.P. Blood-Brain Barrier Opening in Behaving Non-Human Primates via Focused Ultrasound with Systemically Administered Microbubbles. Sci. Rep. 2015, 5, 15076. [Google Scholar] [CrossRef] [PubMed]
- Downs, M.E.; Buch, A.; Sierra, C.; Karakatsani, M.E.; Teichert, T.; Chen, S.; Konofagou, E.E.; Ferrera, V.P. Long-Term Safety of Repeated Blood-Brain Barrier Opening via Focused Ultrasound with Microbubbles in Non-Human Primates Performing a Cognitive Task. PLoS ONE 2015, 10, e0125911. [Google Scholar]
- Liu, H.-L.; Chen, P.-Y.; Yang, H.-W.; Wu, J.-S.; Tseng, I.-C.; Ma, Y.-J.; Huang, C.-Y.; Tsai, H.-C.; Chen, S.-M.; Lu, Y.-J.; et al. In Vivo MR quantification of superparamagnetic iron oxide nanoparticle leakage during low-frequency-ultrasound-induced blood-brain barrier opening in swine. J. Magn. Reson. Imaging 2011, 34, 1313–1324. [Google Scholar] [CrossRef] [PubMed]
- Marquet, F.; Tung, Y.S.; Teichert, T.; Ferrera, V.P.; Konofagou, E.E. Noninvasive, transient and selective blood-brain barrier opening in non-human primates in vivo. PLoS ONE 2011, 6, e22598. [Google Scholar] [CrossRef] [PubMed]
- McDannold, N.; Vykhodtseva, N.; Hynynen, K. Use of Ultrasound Pulses Combined with Definity for Targeted Blood-Brain Barrier Disruption: A Feasibility Study. Ultrasound Med. Biol. 2007, 33, 584–590. [Google Scholar] [CrossRef] [PubMed]
- McMahon, D.; Hynynen, K. Acute Inflammatory Response Following Increased Blood-Brain Barrier Permeability Induced by Focused Ultrasound is Dependent on Microbubble Dose. Theranostics 2017, 7, 3989–4000. [Google Scholar] [CrossRef]
- Kovacs, Z.I.; Kim, S.; Jikaria, N.; Qureshi, F.; Milo, B.; Lewis, B.K.; Bresler, M.; Burks, S.R.; Frank, J.A. Disrupting the blood-brain barrier by focused ultrasound induces sterile inflammation. Proc. Natl. Acad. Sci. USA 2017, 114, E75–E84. [Google Scholar] [CrossRef]
- Hynynen, K.; McDannold, N.; Sheikov, N.A.; Jolesz, F.A.; Vykhodtseva, N. Local and reversible blood-brain barrier disruption by noninvasive focused ultrasound at frequencies suitable for trans-skull sonications. Neuroimage 2005, 24, 12–20. [Google Scholar] [CrossRef]
- Pelekanos, M.; Leinenga, G.; Odabaee, M.; Odabaee, M.; Saifzadeh, S.; Steck, R.; Gotz, J. Establishing sheep as an experimental species to validate ultrasound-mediated blood-brain barrier opening for potential therapeutic interventions. Theranostics 2018, 8, 2583–2602. [Google Scholar] [CrossRef]
- Hynynen, K.; McDannold, N.; Vykhodtseva, N.; Raymond, S.; Weissleder, R.; Jolesz, F.A.; Sheikov, N. Focal disruption of the blood-brain barrier due to 260-kHz ultrasound bursts: A method for molecular imaging and targeted drug delivery. J. Neurosurg. 2006, 105, 445–454. [Google Scholar] [CrossRef]
- Kinoshita, M.; McDannold, N.; Jolesz, F.A.; Hynynen, K. Noninvasive localized delivery of Herceptin to the mouse brain by MRI-guided focused ultrasound-induced blood-brain barrier disruption. Proc. Natl. Acad. Sci. USA 2006, 103, 11719–11723. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.L.; Wai, Y.Y.; Chen, W.S.; Chen, J.C.; Hsu, P.H.; Wu, X.Y.; Huang, W.C.; Yen, T.C.; Wang, J.J. Hemorrhage detection during focused-ultrasound induced blood-brain-barrier opening by using susceptibility-weighted magnetic resonance imaging. Ultrasound Med. Biol. 2008, 34, 598–606. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.C.; Tsai, C.H.; Chen, W.S.; Inserra, C.; Wei, K.C.; Liu, H.L. Safety evaluation of frequent application of microbubble-enhanced focused ultrasound blood-brain-barrier opening. Sci. Rep. 2018, 8, 17720. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.Y.; Lee, P.Y. Efficiency of drug delivery enhanced by acoustic pressure during blood-brain barrier disruption induced by focused ultrasound. Int. J. Nanomed. 2012, 7, 2573–2582. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Bramer, W.M.; Giustini, D.; de Jonge, G.B.; Holland, L.; Bekhuis, T. De-duplication of database search results for systematic reviews in EndNote. J. Med. Libr. Assoc. 2016, 104, 240–243. [Google Scholar] [CrossRef]
- Baghirov, H.; Snipstad, S.; Sulheim, E.; Berg, S.; Hansen, R.; Thorsen, F.; Morch, Y.; Davies, C.L.; Aslund, A.K.O. Ultrasound-mediated delivery and distribution of polymeric nanoparticles in the normal brain parenchyma of a metastatic brain tumour model. PLoS ONE 2018, 13, e0191102. [Google Scholar] [CrossRef]
- Bing, K.F.; Howles, G.P.; Qi, Y.; Palmeri, M.L.; Nightingale, K.R. Blood-Brain Barrier (BBB) Disruption Using a Diagnostic Ultrasound Scanner and Definity in Mice. Ultrasound Med. Biol. 2009, 35, 1298–1308. [Google Scholar] [CrossRef]
- Zhao, B.; Chen, Y.; Liu, J.; Zhang, L.; Wang, J.; Yang, Y.; Lv, Q.; Xie, M. Blood-brain barrier disruption induced by diagnostic ultrasound combined with microbubbles in mice. Oncotarget 2018, 9, 4897–4914. [Google Scholar] [CrossRef]
- Kyriakou, A.; Neufeld, E.; Werner, B.; Paulides, M.M.; Szekely, G.; Kuster, N. A review of numerical and experimental compensation techniques for skull-induced phase aberrations in transcranial focused ultrasound. Int. J. Hyperth. 2014, 30, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Beccaria, K.; Canney, M.; Bouchoux, G.; Puget, S.; Grill, J.; Carpentier, A. Blood-brain barrier disruption with low-intensity pulsed ultrasound for the treatment of pediatric brain tumors: A review and perspectives. Neurosurg. Focus 2020, 48, E10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Y.; Dmello, C.; Chen, L.; Arrieta, V.A.; Gonzalez-Buendia, E.; Kane, J.R.; Magnusson, L.P.; Baran, A.; James, C.D.; Horbinski, C.; et al. Ultrasound-mediated Delivery of Paclitaxel for Glioma: A Comparative Study of Distribution, Toxicity, and Efficacy of Albumin-bound Versus Cremophor Formulations. Clin. Cancer Res. 2020, 26, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Goldwirt, L.; Canney, M.; Horodyckid, C.; Poupon, J.; Mourah, S.; Vignot, A.; Chapelon, J.Y.; Carpentier, A. Enhanced brain distribution of carboplatin in a primate model after blood-brain barrier disruption using an implantable ultrasound device. Cancer Chemother. Pharmacol. 2016, 77, 211–216. [Google Scholar] [CrossRef]
- Fan, C.H.; Liu, H.L.; Ting, C.Y.; Lee, Y.H.; Huang, C.Y.; Ma, Y.J.; Wei, K.C.; Yen, T.C.; Yeh, C.K. Submicron-bubble-enhanced focused ultrasound for blood-brain barrier disruption and improved CNS drug delivery. PLoS ONE 2014, 9, e96327. [Google Scholar] [CrossRef]
- Hynynen, K.; Jones, R.M. Image-guided ultrasound phased arrays are a disruptive technology for non-invasive therapy. Phys. Med. Biol. 2016, 61, R206–R248. [Google Scholar] [CrossRef]
- Timbie, K.F.; Mead, B.P.; Price, R.J. Drug and gene delivery across the blood-brain barrier with focused ultrasound. J. Control. Release 2015, 219, 61–75. [Google Scholar] [CrossRef]
- McDannold, N.; Zhang, Y.; Supko, J.G.; Power, C.; Sun, T.; Peng, C.; Vykhodtseva, N.; Golby, A.J.; Reardon, D.A. Acoustic feedback enables safe and reliable carboplatin delivery across the blood-brain barrier with a clinical focused ultrasound system and improves survival in a rat glioma model. Theranostics 2019, 9, 6284–6299. [Google Scholar] [CrossRef]
- McDannold, N.; Zhang, Y.; Supko, J.G.; Power, C.; Sun, T.; Vykhodtseva, N.; Golby, A.J.; Reardon, D.A. Blood-brain barrier disruption and delivery of irinotecan in a rat model using a clinical transcranial MRI-guided focused ultrasound system. Sci. Rep. 2020, 10, 8766. [Google Scholar] [CrossRef]
- Arvanitis, C.D.; Livingstone, M.S.; Vykhodtseva, N.; McDannold, N. Controlled ultrasound-induced blood-brain barrier disruption using passive acoustic emissions monitoring. PLoS ONE 2012, 7, e45783. [Google Scholar] [CrossRef]
- Anastasiadis, P.; Gandhi, D.; Guo, Y.; Ahmed, A.K.; Bentzen, S.M.; Arvanitis, C.; Woodworth, G.F. Localized blood-brain barrier opening in infiltrating gliomas with MRI-guided acoustic emissions-controlled focused ultrasound. Proc. Natl. Acad. Sci. USA 2021, 118, e2103280118. [Google Scholar] [CrossRef] [PubMed]
- Gasca-Salas, C.; Fernández-Rodríguez, B.; Pineda-Pardo, J.A.; Rodríguez-Rojas, R.; Obeso, I.; Hernández-Fernández, F.; del Álamo, M.; Mata, D.; Guida, P.; Ordás-Bandera, C.; et al. Blood-brain barrier opening with focused ultrasound in Parkinson’s disease dementia. Nat. Commun. 2021, 12, 779. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Kim, M.J.; Jung, H.H.; Chang, W.S.; Choi, H.S.; Rachmilevitch, I.; Zadicario, E.; Chang, J.W. Safety and feasibility of multiple blood-brain barrier disruptions for the treatment of glioblastoma in patients undergoing standard adjuvant chemotherapy. J. Neurosurg. 2020, 134, 475–483. [Google Scholar] [CrossRef]
- Chen, K.T.; Chai, W.Y.; Lin, Y.J.; Lin, C.J.; Chen, P.Y.; Tsai, H.C.; Huang, C.Y.; Kuo, J.S.; Liu, H.L.; Wei, K.C. Neuronavigation-guided focused ultrasound for transcranial blood-brain barrier opening and immunostimulation in brain tumors. Sci. Adv. 2021, 7, eabd0772. [Google Scholar] [CrossRef] [PubMed]
- Beccaria, K.; Canney, M.; Bouchoux, G.; Desseaux, C.; Grill, J.; Heimberger, A.B.; Carpentier, A. Ultrasound-induced blood-brain barrier disruption for the treatment of gliomas and other primary CNS tumors. Cancer Lett. 2020, 479, 13–22. [Google Scholar] [CrossRef]
- Horodyckid, C.; Canney, M.; Vignot, A.; Boisgard, R.; Drier, A.; Huberfeld, G.; François, C.; Prigent, A.; Santin, M.D.; Adam, C.; et al. Safe long-term repeated disruption of the blood-brain barrier using an implantable ultrasound device: A multiparametric study in a primate model. J. Neurosurg. 2017, 126, 1351–1361. [Google Scholar] [CrossRef]
- Pouliopoulos, A.N.; Wu, S.Y.; Burgess, M.T.; Karakatsani, M.E.; Kamimura, H.A.S.; Konofagou, E.E. A Clinical System for Non-invasive Blood-Brain Barrier Opening Using a Neuronavigation-Guided Single-Element Focused Ultrasound Transducer. Ultrasound Med. Biol. 2020, 46, 73–89. [Google Scholar] [CrossRef] [PubMed]
- Marty, B.; Larrat, B.; Van Landeghem, M.; Robic, C.; Robert, P.; Port, M.; Le Bihan, D.; Pernot, M.; Tanter, M.; Lethimonnier, F.; et al. Dynamic study of blood-brain barrier closure after its disruption using ultrasound: A quantitative analysis. J. Cereb. Blood Flow Metab. 2012, 32, 1948–1958. [Google Scholar] [CrossRef]
- McMahon, D.; Deng, L.; Hynynen, K. Comparing rapid short-pulse to tone burst sonication sequences for focused ultrasound and microbubble-mediated blood-brain barrier permeability enhancement. J. Control. Release 2020, 329, 696–705. [Google Scholar] [CrossRef]
- Choi, J.J.; Selert, K.; Vlachos, F.; Wong, A.; Konofagou, E.E. Noninvasive and localized neuronal delivery using short ultrasonic pulses and microbubbles. Proc. Natl. Acad. Sci. USA 2011, 108, 16539–16544. [Google Scholar] [CrossRef]
- Jordao, J.F.; Thévenot, E.; Markham-Coultes, K.; Scarcelli, T.; Weng, Y.-Q.; Xhima, K.; O’Reilly, M.; Huang, Y.; McLaurin, J.; Hynynen, K.; et al. Amyloid-beta plaque reduction, endogenous antibody delivery and glial activation by brain-targeted, transcranial focused ultrasound. Exp. Neurol. 2013, 248, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, M.; McDannold, N.; Jolesz, F.A.; Hynynen, K. Targeted delivery of antibodies through the blood-brain barrier by MRI-guided focused ultrasound. Biochem. Biophys. Res. Commun. 2006, 340, 1085–1090. [Google Scholar] [CrossRef]
- Treat, L.H.; McDannold, N.; Vykhodtseva, N.; Zhang, Y.; Tam, K.; Hynynen, K. Targeted delivery of doxorubicin to the rat brain at therapeutic levels using MRI-guided focused ultrasound. Int. J. Cancer 2007, 121, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.H.; Cheng, Y.H.; Ting, C.Y.; Ho, Y.J.; Hsu, P.H.; Liu, H.L.; Yeh, C.K. Ultrasound/Magnetic Targeting with SPIO-DOX-Microbubble Complex for Image-Guided Drug Delivery in Brain Tumors. Theranostics 2016, 6, 1542–1556. [Google Scholar] [CrossRef]
- Liu, H.L.; Hsu, P.H.; Chu, P.C.; Wai, Y.Y.; Chen, J.C.; Shen, C.R.; Yen, T.C.; Wang, J.J. Magnetic resonance imaging enhanced by superparamagnetic iron oxide particles: Usefulness for distinguishing between focused ultrasound-induced blood-brain barrier disruption and brain hemorrhage. J. Magn. Reson. Imaging 2009, 29, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Samiotaki, G.; Vlachos, F.; Tung, Y.S.; Konofagou, E.E. A quantitative pressure and microbubble-size dependence study of focused ultrasound-induced blood-brain barrier opening reversibility in vivo using MRI. Magn. Reson. Med. 2012, 67, 769–777. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Konofagou, E.E. The size of blood-brain barrier opening induced by focused ultrasound is dictated by the acoustic pressure. J. Cereb. Blood Flow Metab. 2014, 34, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.J.; Wang, S.; Tung, Y.S.; Morrison, B., III; Konofagou, E.E. Molecules of various pharmacologically-relevant sizes can cross the ultrasound-induced blood-brain barrier opening in vivo. Ultrasound Med. Biol. 2010, 36, 58–67. [Google Scholar] [CrossRef]
- Shen, Y.; Guo, J.; Chen, G.; Chin, C.T.; Chen, X.; Chen, J.; Wang, F.; Chen, S.; Dan, G. Delivery of liposomes with different sizes to mice brain after sonication by focused ultrasound in the presence of microbubbles. Ultrasound Med. Biol. 2016, 42, 1499–1511. [Google Scholar] [CrossRef]
- Aryal, M.; Vykhodtseva, N.; Zhang, Y.Z.; McDannold, N. Multiple sessions of liposomal doxorubicin delivery via focused ultrasound mediated blood-brain barrier disruption: A safety study. J. Control. Release 2015, 204, 60–69. [Google Scholar] [CrossRef]
- Park, J.; Zhang, Y.; Vykhodtseva, N.; Jolesz, F.A.; McDannold, N.J. The kinetics of blood brain barrier permeability and targeted doxorubicin delivery into brain induced by focused ultrasound. J. Control. Release 2012, 162, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Vlachos, F.; Tung, Y.S.; Konofagou, E. Permeability dependence study of the focused ultrasound-induced blood-brain barrier opening at distinct pressures and microbubble diameters using DCE-MRI. Magn. Reson. Med. 2011, 66, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Baseri, B.; Choi, J.J.; Tung, Y.S.; Konofagou, E.E. Multi-modality safety assessment of blood-brain barrier opening using focused ultrasound and definity microbubbles: A short-term study. Ultrasound Med. Biol. 2010, 36, 1445–1459. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Sheeran, P.S.; Wu, S.Y.; Olumolade, O.O.; Dayton, P.A.; Konofagou, E.E. Targeted drug delivery with focused ultrasound-induced blood-brain barrier opening using acoustically-activated nanodroplets. J. Control. Release 2013, 172, 795–804. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.J.; Feshitan, J.A.; Baseri, B.; Wang, S.; Tung, Y.S.; Borden, M.A.; Konofagou, E.E. Microbubble-size dependence of focused ultrasound-induced blood-brain barrier opening in mice in vivo. IEEE Trans. Biomed. Eng. 2010, 57, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.J.; Selert, K.; Gao, Z.; Samiotaki, G.; Baseri, B.; Konofagou, E.E. Noninvasive and localized blood-brain barrier disruption using focused ultrasound can be achieved at short pulse lengths and low pulse repetition frequencies. J. Cereb. Blood Flow Metab. 2011, 31, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.J.; Wang, S.; Brown, T.R.; Small, S.A.; Duff, K.E.; Konofagou, E.E. Noninvasive and transient blood-brain barrier opening in the hippocampus of Alzheimer’s double transgenic mice using focused ultrasound. Ultrason. Imaging 2008, 30, 189–200. [Google Scholar] [CrossRef]
- Englander, Z.K.; Wei, H.-J.; Pouliopoulos, A.N.; Bendau, E.; Upadhyayula, P.; Jan, C.-I.; Spinazzi, E.F.; Yoh, N.; Tazhibi, M.; McQuillan, N.M.; et al. Focused ultrasound mediated blood-brain barrier opening is safe and feasible in a murine pontine glioma model. Sci. Rep. 2021, 11, 6521. [Google Scholar] [CrossRef]
- Lapin, N.A.; Gill, K.; Shah, B.R.; Chopra, R. Consistent opening of the blood brain barrier using focused ultrasound with constant intravenous infusion of microbubble agent. Sci. Rep. 2020, 10, 16546. [Google Scholar] [CrossRef]
- Liu, H.L.; Huang, C.Y.; Chen, J.Y.; Wang, H.Y.; Chen, P.Y.; Wei, K.C. Pharmacodynamic and therapeutic investigation of focused ultrasound-induced blood-brain barrier opening for enhanced temozolomide delivery in glioma treatment. PLoS ONE 2014, 9, e114311. [Google Scholar]
- McDannold, N.; Zhang, Y.; Vykhodtseva, N. The Effects of Oxygen on Ultrasound-Induced Blood-Brain Barrier Disruption in Mice. Ultrasound Med. Biol. 2017, 43, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Morse, S.V.; Mishra, A.; Chan, T.G.; R, T.M.d.R.; Choi, J.J. Liposome delivery to the brain with rapid short-pulses of focused ultrasound and microbubbles. J. Control. Release 2022, 341, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Morse, S.V.; Pouliopoulos, A.N.; Chan, T.G.; Copping, M.J.; Lin, J.; Long, N.J.; Choi, J.J. Rapid Short-pulse Ultrasound Delivers Drugs Uniformly across the Murine Blood-Brain Barrier with Negligible Disruption. Radiology 2019, 291, 459–466. [Google Scholar] [CrossRef]
- Olumolade, O.O.; Wang, S.; Samiotaki, G.; Konofagou, E.E. Longitudinal Motor and Behavioral Assessment of Blood-Brain Barrier Opening with Transcranial Focused Ultrasound. Ultrasound Med. Biol. 2016, 42, 2270–2282. [Google Scholar] [CrossRef]
- Omata, D.; Maruyama, T.; Unga, J.; Hagiwara, F.; Munakata, L.; Kageyama, S.; Shima, T.; Suzuki, Y.; Maruyama, K.; Suzuki, R. Effects of encapsulated gas on stability of lipid-based microbubbles and ultrasound-triggered drug delivery. J. Control. Release 2019, 311, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Raymond, S.B.; Skoch, J.; Hynynen, K.; Bacskai, B.J. Multiphoton imaging of ultrasound/Optison mediated cerebrovascular effects in vivo. J. Cereb. Blood Flow Metab. 2007, 27, 393–403. [Google Scholar] [CrossRef]
- Raymond, S.B.; Treat, L.H.; Dewey, J.D.; McDannold, N.J.; Hynynen, K.; Bacskai, B.J. Ultrasound enhanced delivery of molecular imaging and therapeutic agents in Alzheimer’s disease mouse models. PLoS ONE 2008, 3, e2175. [Google Scholar] [CrossRef]
- Sierra, C.; Acosta, C.; Chen, C.; Wu, S.Y.; Karakatsani, M.E.; Bernal, M.; Konofagou, E.E. Lipid microbubbles as a vehicle for targeted drug delivery using focused ultrasound-induced blood-brain barrier opening. J. Cereb. Blood Flow Metab. 2017, 37, 1236–1250. [Google Scholar] [CrossRef]
- Wu, S.K.; Chiang, C.F.; Hsu, Y.H.; Lin, T.H.; Liou, H.C.; Fu, W.M.; Lin, W.L. Short-time focused ultrasound hyperthermia enhances liposomal doxorubicin delivery and antitumor efficacy for brain metastasis of breast cancer. Int. J. Nanomed. 2014, 9, 4485–4494. [Google Scholar]
- Alli, S.; Figueiredo, C.A.; Golbourn, B.; Sabha, N.; Wu, M.Y.; Bondoc, A.; Luck, A.; Coluccia, D.; Maslink, C.; Smith, C.; et al. Brainstem blood brain barrier disruption using focused ultrasound: A demonstration of feasibility and enhanced doxorubicin delivery. J. Control. Release 2018, 281, 29–41. [Google Scholar] [CrossRef]
- Aryal, M.; Park, J.; Vykhodtseva, N.; Zhang, Y.Z.; McDannold, N. Enhancement in blood-tumor barrier permeability and delivery of liposomal doxorubicin using focused ultrasound and microbubbles: Evaluation during tumor progression in a rat glioma model. Phys. Med. Biol. 2015, 60, 2511–2527. [Google Scholar] [CrossRef] [PubMed]
- Aslund, A.K.; Snipstad, S.; Healey, A.; Kvale, S.; Torp, S.H.; Sontum, P.C.; Davies, C.L.; van Wamel, A. Efficient Enhancement of Blood-Brain Barrier Permeability Using Acoustic Cluster Therapy (ACT). Theranostics 2017, 7, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Chopra, R.; Vykhodtseva, N.; Hynynen, K. Influence of exposure time and pressure amplitude on blood-brain-barrier opening using transcranial ultrasound exposures. ACS Chem. Neurosci. 2010, 1, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.H.; Ting, C.Y.; Chang, Y.C.; Wei, K.C.; Liu, H.L.; Yeh, C.K. Drug-loaded bubbles with matched focused ultrasound excitation for concurrent blood-brain barrier opening and brain-tumor drug delivery. Acta Biomater. 2015, 15, 89–101. [Google Scholar] [CrossRef]
- Goutal, S.; Gerstenmayer, M.; Auvity, S.; Caille, F.; Meriaux, S.; Buvat, I.; Larrat, B.; Tournier, N. Physical blood-brain barrier disruption induced by focused ultrasound does not overcome the transporter-mediated efflux of erlotinib. J. Control. Release 2018, 292, 210–220. [Google Scholar] [CrossRef]
- Han, M.; Seo, H.; Choi, H.; Lee, E.H.; Park, J. Localized Modification of Water Molecule Transport after Focused Ultrasound-Induced Blood-Brain Barrier Disruption in Rat Brain. Front. Neurosci. 2021, 15, 685977. [Google Scholar] [CrossRef]
- Huh, H.; Park, T.Y.; Seo, H.; Han, M.; Jung, B.; Choi, H.J.; Lee, E.H.; Pahk, K.J.; Kim, H.; Park, J. A local difference in blood-brain barrier permeability in the caudate putamen and thalamus of a rat brain induced by focused ultrasound. Sci. Rep. 2020, 10, 19286. [Google Scholar] [CrossRef]
- Jung, B.; Huh, H.; Lee, E.H.; Han, M.; Park, J. An advanced focused ultrasound protocol improves the blood-brain barrier permeability and doxorubicin delivery into the rat brain. J. Control. Release 2019, 315, 55–64. [Google Scholar] [CrossRef]
- Kobus, T.; Vykhodtseva, N.; Pilatou, M.; Zhang, Y.; McDannold, N. Safety Validation of Repeated Blood-Brain Barrier Disruption Using Focused Ultrasound. Ultrasound Med. Biol. 2016, 42, 481–492. [Google Scholar] [CrossRef]
- Kovacs, Z.I.; Tu, T.W.; Sundby, M.; Qureshi, F.; Lewis, B.K.; Jikaria, N.; Burks, S.R.; Frank, J.A. MRI and histological evaluation of pulsed focused ultrasound and microbubbles treatment effects in the brain. Theranostics 2018, 8, 4837–4855. [Google Scholar] [CrossRef]
- Liu, H.L.; Hua, M.Y.; Chen, P.Y.; Chu, P.C.; Pan, C.H.; Yang, H.W.; Huang, C.Y.; Wang, J.J.; Yen, T.C.; Wei, K.C. Blood-brain barrier disruption with focused ultrasound enhances delivery of chemotherapeutic drugs for glioblastoma treatment. Radiology 2010, 255, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.L.; Pan, C.H.; Ting, C.Y.; Hsiao, M.J. Opening of the blood-brain barrier by low-frequency (28-kHz) ultrasound: A novel pinhole-assisted mechanical scanning device. Ultrasound Med. Biol. 2010, 36, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.L.; Wai, Y.Y.; Hsu, P.H.; Lyu, L.A.; Wu, J.S.; Shen, C.R.; Chen, J.C.; Yen, T.C.; Wang, J.J. In Vivo assessment of macrophage CNS infiltration during disruption of the blood-brain barrier with focused ultrasound: A magnetic resonance imaging study. J. Cereb. Blood Flow Metab. 2010, 30, 177–186. [Google Scholar] [CrossRef] [PubMed]
- McDannold, N.; Zhang, Y.; Vykhodtseva, N. Blood-brain barrier disruption and vascular damage induced by ultrasound bursts combined with microbubbles can be influenced by choice of anesthesia protocol. Ultrasound Med. Biol. 2011, 37, 1259–1270. [Google Scholar] [CrossRef]
- McMahon, D.; Lassus, A.; Gaud, E.; Jeannot, V.; Hynynen, K. Microbubble formulation influences inflammatory response to focused ultrasound exposure in the brain. Sci. Rep. 2020, 10, 21534. [Google Scholar] [CrossRef]
- McMahon, D.; Oakden, W.; Hynynen, K. Investigating the effects of dexamethasone on blood-brain barrier permeability and inflammatory response following focused ultrasound and microbubble exposure. Theranostics 2020, 10, 1604–1618. [Google Scholar] [CrossRef]
- O’Reilly, M.A.; Hough, O.; Hynynen, K. Blood-Brain Barrier Closure Time After Controlled Ultrasound-Induced Opening Is Independent of Opening Volume. J. Ultrasound Med. 2017, 36, 475–483. [Google Scholar] [CrossRef]
- O’Reilly, M.A.; Waspe, A.C.; Ganguly, M.; Hynynen, K. Focused-ultrasound disruption of the blood-brain barrier using closely-timed short pulses: Influence of sonication parameters and injection rate. Ultrasound Med. Biol. 2011, 37, 587–594. [Google Scholar] [CrossRef]
- Park, J.; Aryal, M.; Vykhodtseva, N.; Zhang, Y.Z.; McDannold, N. Evaluation of permeability, doxorubicin delivery, and drug retention in a rat brain tumor model after ultrasound-induced blood-tumor barrier disruption. J. Control. Release 2017, 250, 77–85. [Google Scholar] [CrossRef]
- Song, K.H.; Fan, A.C.; Hinkle, J.J.; Newman, J.; Borden, M.A.; Harvey, B.K. Microbubble gas volume: A unifying dose parameter in blood-brain barrier opening by focused ultrasound. Theranostics 2017, 7, 144–152. [Google Scholar] [CrossRef]
- Wei, K.C.; Chu, P.-C.; Wang, H.-Y.J.; Huang, C.-Y.; Chen, P.-Y.; Tsai, H.-C.; Lu, Y.-J.; Lee, P.-Y.; Tseng, I.-C.; Feng, L.-Y.; et al. Focused ultrasound-induced blood-brain barrier opening to enhance temozolomide delivery for glioblastoma treatment: A preclinical study. PLoS ONE 2013, 8, e58995. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.K.; Chu, P.C.; Chai, W.Y.; Kang, S.T.; Tsai, C.H.; Fan, C.H.; Yeh, C.K.; Liu, H.L. Characterization of Different Microbubbles in Assisting Focused Ultrasound-Induced Blood-Brain Barrier Opening. Sci. Rep. 2017, 7, 46689. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.Y.; Chen, C.C.; Kao, Y.H.; Chen, C.L.; Ko, C.E.; Horng, S.C.; Chen, R.C. Evaluation of Dose Distribution of Molecular Delivery after Blood-Brain Barrier Disruption by Focused Ultrasound with Treatment Planning. Ultrasound Med. Biol. 2013, 39, 620–627. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.Y.; Ko, C.E.; Huang, S.Y.; Chung, I.F.; Chen, G.S. Pharmacokinetic changes induced by focused ultrasound in glioma-bearing rats as measured by dynamic contrast-enhanced MRI. PLoS ONE 2014, 9, e92910. [Google Scholar] [CrossRef]
- Yang, F.Y.; Lin, Y.S.; Kang, K.H.; Chao, T.K. Reversible blood-brain barrier disruption by repeated transcranial focused ultrasound allows enhanced extravasation. J. Control. Release 2011, 150, 111–116. [Google Scholar] [CrossRef]
- Yang, F.Y.; Wang, H.E.; Lin, G.L.; Lin, H.H.; Wong, T.T. Evaluation of the increase in permeability of the blood-brain barrier during tumor progression after pulsed focused ultrasound. Int. J. Nanomed. 2012, 7, 723–730. [Google Scholar] [CrossRef]
- Zhang, Y.; Tan, H.; Bertram, E.H.; Aubry, J.-F.; Lopes, M.-B.; Roy, J.; Dumont, E.; Xie, M.; Zuo, Z.; Klibanov, A.L.; et al. Non-Invasive, Focal Disconnection of Brain Circuitry Using Magnetic Resonance-Guided Low-Intensity Focused Ultrasound to Deliver a Neurotoxin. Ultrasound Med. Biol. 2016, 42, 2261–2269. [Google Scholar] [CrossRef]
- McDannold, N.; Vykhodtseva, N.; Hynynen, K. Targeted disruption of the blood-brain barrier with focused ultrasound: Association with cavitation activity. Phys. Med. Biol. 2006, 51, 793–807. [Google Scholar] [CrossRef]
- McDannold, N.; Vykhodtseva, N.; Hynynen, K. Blood-brain barrier disruption induced by focused ultrasound and circulating preformed microbubbles appears to be characterized by the mechanical index. Ultrasound Med. Biol. 2008, 34, 834–840. [Google Scholar] [CrossRef]
- McDannold, N.; Vykhodtseva, N.; Hynynen, K. Effects of acoustic parameters and ultrasound contrast agent dose on focused-ultrasound induced blood-brain barrier disruption. Ultrasound Med. Biol. 2008, 34, 930–937. [Google Scholar] [CrossRef]
- Mei, J.; Cheng, Y.; Song, Y.; Yang, Y.; Wang, F.; Liu, Y.; Wang, Z. Experimental study on targeted methotrexate delivery to the rabbit brain via magnetic resonance imaging-guided focused ultrasound. J. Ultrasound Med. 2009, 28, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Cheng, Y.; Mei, J.; Song, Y.; Yang, Y.Q.; Liu, Y.; Wang, Z. Focused ultrasound microbubble destruction-mediated changes in blood-brain barrier permeability assessed by contrast-enhanced magnetic resonance imaging. J. Ultrasound Med. 2009, 28, 1501–1509. [Google Scholar] [CrossRef]
- O’Reilly, M.A.; Jones, R.M.; Barrett, E.; Schwab, A.; Head, E.; Hynynen, K. Investigation of the Safety of Focused Ultrasound-Induced Blood-Brain Barrier Opening in a Natural Canine Model of Aging. Theranostics 2017, 7, 3573–3584. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.; Lee, W.; Chen, E.; Lee, J.E.; Croce, P.; Cammalleri, A.; Foley, L.; Tsao, A.L.; Yoo, S.S. Localized Blood-Brain Barrier Opening in Ovine Model Using Image-Guided Transcranial Focused Ultrasound. Ultrasound Med. Biol. 2019, 45, 2391–2404. [Google Scholar] [CrossRef] [PubMed]
- Marquet, F.; Teichert, T.; Wu, S.Y.; Tung, Y.S.; Downs, M.; Wang, S.; Chen, C.; Ferrera, V.; Konofagou, E.E. Real-time, transcranial monitoring of safe blood-brain barrier opening in non-human primates. PLoS ONE 2014, 9, e84310. [Google Scholar] [CrossRef] [PubMed]
- McDannold, N.; Arvanitis, C.D.; Vykhodtseva, N.; Livingstone, M.S. Temporary disruption of the blood-brain barrier by use of ultrasound and microbubbles: Safety and efficacy evaluation in rhesus macaques. Cancer Res. 2012, 72, 3652–3663. [Google Scholar] [CrossRef]
- Wu, S.Y.; Sanchez, C.S.; Samiotaki, G.; Buch, A.; Ferrera, V.P.; Konofagou, E.E. Characterizing Focused-Ultrasound Mediated Drug Delivery to the Heterogeneous Primate Brain In Vivo with Acoustic Monitoring. Sci. Rep. 2016, 6, 37094. [Google Scholar] [CrossRef]
- Pasquinelli, C.; Hanson, L.G.; Siebner, H.R.; Lee, H.J.; Thielscher, A. Safety of transcranial focused ultrasound stimulation: A systematic review of the state of knowledge from both human and animal studies. Brain Stimul. 2019, 12, 1367–1380. [Google Scholar] [CrossRef]
- Helfield, B.L.; Huo, X.; Williams, R.; Goertz, D.E. The effect of preactivation vial temperature on the acoustic properties of Definity. Ultrasound Med. Biol. 2012, 38, 1298–1305. [Google Scholar] [CrossRef]
- Goertz, D.E.; de Jong, N.; van der Steen, A.F. Attenuation and size distribution measurements of Definity and manipulated Definity populations. Ultrasound Med. Biol. 2007, 33, 1376–1388. [Google Scholar] [CrossRef]
- Meng, Y.; Reilly, R.M.; Pezo, R.C.; Trudeau, M.; Sahgal, A.; Singnurkar, A.; Perry, J.; Myrehaug, S.; Pople, C.B.; Davidson, B.; et al. MR-guided focused ultrasound enhances delivery of trastuzumab to Her2-positive brain metastases. Sci. Transl. Med. 2021, 13, eabj4011. [Google Scholar] [CrossRef] [PubMed]
- Bing, C.; Hong, Y.; Hernandez, C.; Rich, M.; Cheng, B.; Munaweera, I.; Szczepanski, D.; Xi, Y.; Bolding, M.; Exner, A.; et al. Characterization of different bubble formulations for blood-brain barrier opening using a focused ultrasound system with acoustic feedback control. Sci. Rep. 2018, 8, 7986. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Baik, K.; Jeon, S.; Chang, W.S.; Ye, B.S.; Chang, J.W. Extensive frontal focused ultrasound mediated blood-brain barrier opening for the treatment of Alzheimer’s disease: A proof-of-concept study. Transl. Neurodegener. 2021, 10, 44. [Google Scholar] [CrossRef]
- Yin, X.; Hynynen, K. A numerical study of transcranial focused ultrasound beam propagation at low frequency. Phys. Med. Biol. 2005, 50, 1821–1836. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, M.A.; Huang, Y.; Hynynen, K. The impact of standing wave effects on transcranial focused ultrasound disruption of the blood-brain barrier in a rat model. Phys. Med. Biol. 2010, 55, 5251–5267. [Google Scholar] [CrossRef]
- Le Floc’h, J.; Lu, H.D.; Lim, T.L.; Demore, C.; Prud’homme, R.K.; Hynynen, K.; Foster, F.S. Transcranial Photoacoustic Detection of Blood-Brain Barrier Disruption Following Focused Ultrasound-Mediated Nanoparticle Delivery. Mol. Imaging Biol. 2020, 22, 324–334. [Google Scholar] [CrossRef]
- Wang, P.H.; Liu, H.L.; Hsu, P.H.; Lin, C.Y.; Wang, C.R.; Chen, P.Y.; Wei, K.C.; Yen, T.C.; Li, M.L. Gold-nanorod contrast-enhanced photoacoustic micro-imaging of focused-ultrasound induced blood-brain-barrier opening in a rat model. J. Biomed. Opt. 2012, 17, 061222. [Google Scholar] [CrossRef]
- Meng, Y.; Pople, C.B.; Budiansky, D.; Li, D.; Suppiah, S.; Lim-Fat, M.J.; Perry, J.; Sahgal, A.; Lipsman, N. Current state of therapeutic focused ultrasound applications in neuro-oncology. J. Neurooncol. 2022, 156, 49–59. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gandhi, K.; Barzegar-Fallah, A.; Banstola, A.; Rizwan, S.B.; Reynolds, J.N.J. Ultrasound-Mediated Blood–Brain Barrier Disruption for Drug Delivery: A Systematic Review of Protocols, Efficacy, and Safety Outcomes from Preclinical and Clinical Studies. Pharmaceutics 2022, 14, 833. https://doi.org/10.3390/pharmaceutics14040833
Gandhi K, Barzegar-Fallah A, Banstola A, Rizwan SB, Reynolds JNJ. Ultrasound-Mediated Blood–Brain Barrier Disruption for Drug Delivery: A Systematic Review of Protocols, Efficacy, and Safety Outcomes from Preclinical and Clinical Studies. Pharmaceutics. 2022; 14(4):833. https://doi.org/10.3390/pharmaceutics14040833
Chicago/Turabian StyleGandhi, Kushan, Anita Barzegar-Fallah, Ashik Banstola, Shakila B. Rizwan, and John N. J. Reynolds. 2022. "Ultrasound-Mediated Blood–Brain Barrier Disruption for Drug Delivery: A Systematic Review of Protocols, Efficacy, and Safety Outcomes from Preclinical and Clinical Studies" Pharmaceutics 14, no. 4: 833. https://doi.org/10.3390/pharmaceutics14040833