Nanoparticles Obtained from Zein for Encapsulation of Mesalazine
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
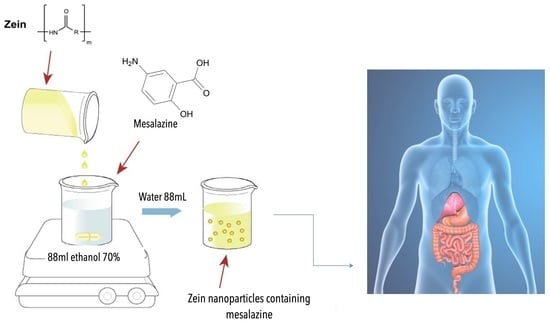
2.2. Preparation of Zein-Nanoparticle-Loaded Mesalazine (NP-ZN-MSZ)
2.3. Size, Particle Distribution and Polydispersion Index
2.4. Zeta Potential
2.5. Dosing of the Drug in the Nanoparticles
2.6. Quantification of Mesalazine using HPLC
2.7. Encapsulation Efficiency (EE%)
2.8. Scanning Electron Microscopy (SEM)
2.9. Fourier Transform Infrared Spectroscopy (FTIR)
2.10. Thermogravimetric Analysis (TGA)
2.11. In Vitro Release Kinetics Study
2.12. In Vitro Cytotoxicity Study
2.13. Statistical Analysis
3. Results and Discussion
3.1. NP Preparation and Characterization
3.2. Scanning Electron Microscopy (SEM)
3.3. Fourier Transform Infrared Spectroscopy (FTIR)
3.4. Thermogravimetric Analysis (TGA) and Differential Exploratory Calorimetry (DSC)
3.5. Study of the In Vitro Release Profile
3.6. Kinetics and Mechanism of In Vitro Drug Release
3.7. In Vitro Cytotoxicity Study
3.8. Stability Test
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad, A.; Ansari, M.; Mishra, R.K.; Kumar, A.; Vyawahare, A.; Verma, R.K.; Raza, S.S.; Khan, R. Enteric-coated gelatin nanoparticles mediated oral delivery of 5-aminosalicylic acid alleviates severity of DSS-induced ulcerative colitis. Mater. Sci. Eng. C 2020, 119, 111582. [Google Scholar] [CrossRef] [PubMed]
- Sardo, H.S.; Saremnejad, F.; Bagheri, S.; Akhgari, A.; Garekani, H.A.; Sadeghi, F. A review on 5-aminosalicylic acid colon-targeted oral drug delivery systems. Int. J. Pharm. 2019, 558, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Berends, S.E.; Strik, A.S.; Löwenberg, M.; D’Haens, G.R.; Mathôt, R.A.A. Clinical Pharmacokinetic and Pharmacodynamic Considerations in the Treatment of Ulcerative Colitis. Clin. Pharmacokinet. 2018, 58, 15–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lau, E.T.L.; Giddings, S.J.; Mohammed, S.G.; Dubois, P.; Johnson, S.K.; Stanley, R.A.; Halley, P.J.; Steadman, K.J. Encapsulation of Hydrocortisone and Mesalazine in Zein Microparticles. Pharmaceutics 2013, 5, 277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viscido, A.; Capannolo, A.; Latella, G.; Caprilli, R.; Frieri, G. Nanotechnology in the treatment of inflammatory bowel diseases. J. Crohn’s Colitis 2014, 8, 903–918. [Google Scholar] [CrossRef]
- Inchaurraga, L.; Martín-Arbella, N.; Zabaleta, V.; Quincoces, G.; Peñuelas, I.; Irache, J.M. In vivo study of the mucus-permeating properties of PEG-coated nanoparticles following oral administration. Eur. J. Pharm. Biopharm. 2015, 97, 280–289. [Google Scholar] [CrossRef]
- Ulbrich, K.; Holá, K.; Šubr, V.; Bakandritsos, A.; Tuček, J.; Zbořil, R. Targeted Drug Delivery with Polymers and Magnetic Nanoparticles: Covalent and Noncovalent Approaches, Release Control, and Clinical Studies. Chem. Rev. 2016, 116, 5338–5431. [Google Scholar] [CrossRef] [Green Version]
- Feng, S.; Sun, Y.; Wang, D.; Sun, P.; Shao, P. Effect of adjusting pH and chondroitin sulfate on the formation of curcumin-zein nanoparticles: Synthesis, characterization and morphology. Carbohydr. Polym. 2020, 250, 116970. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, J.; Qin, Y.; Jiang, B.; Zhang, T. Zein/fucoidan-based composite nanoparticles for the encapsulation of pterostilbene: Preparation, characterization, physicochemical stability, and formation mechanism. Int. J. Biol. Macromol. 2020, 158, 461–470. [Google Scholar] [CrossRef]
- Moreno, L.C.G.E.I.; Puerta, E.; Suárez-Santiago, J.E.; Santos-Magalhães, N.S.; Ramirez, M.J.; Irache, J.M. Effect of the oral administration of nanoencapsulated quercetin on a mouse model of Alzheimer’s disease. Int. J. Pharm. 2017, 517, 50–57. [Google Scholar] [CrossRef]
- Lai, L.; Guo, H. Preparation of new 5-fluorouracil-loaded zein nanoparticles for liver targeting. Int. J. Pharm. 2011, 404, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.; Duan, W.; Lee, B.-J.; Tran, T.T. Drug stabilization in the gastrointestinal tract and potential applications in the colonic delivery of oral zein-based formulations. Int. J. Pharm. 2019, 569, 118614. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; McClements, D.J.; Peng, X.; Xu, Z.; Meng, M.; Chen, L.; Jin, Z. Fabrication of zein–carboxymethyl cellulose nanoparticles for co-delivery of quercetin and resveratrol. J. Food Eng. 2022, 341, 111322. [Google Scholar] [CrossRef]
- Penalva, R.; González-Navarro, C.J.; Gamazo, C.; Esparza, I.; Irache, J.M. Zein nanoparticles for oral delivery of quercetin: Pharmacokinetic studies and preventive anti-inflammatory effects in a mouse model of endotoxemia. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 103–110. [Google Scholar] [CrossRef]
- Li, S.; Wang, X.; Li, W.; Yuan, G.; Pan, Y.; Chen, H. Preparation and characterization of a novel conformed bipolymer paclitaxel-nanoparticle using tea polysaccharides and zein. Carbohydr. Polym. 2016, 146, 52–57. [Google Scholar] [CrossRef]
- Jain, A.; Sharma, G.; Kushwah, V.; Ghoshal, G.; Jain, A.; Singh, B.; Shivhare, U.S.; Jain, S.; Katare, O.P. Beta carotene-loaded zein nanoparticles to improve the biopharmaceutical attributes and to abolish the toxicity of methotrexate: A preclinical study for breast cancer. Artif. Cells Nanomed. Biotechnol. 2018, 46, 402–412. [Google Scholar] [CrossRef] [Green Version]
- Veneranda, M.; Hu, Q.; Wang, T.; Luo, Y.; Castro, K.; Madariaga, J.M. Formation and characterization of zein-caseinate-pectin complex nanoparticles for encapsulation of eugenol. LWT 2018, 89, 596–603. [Google Scholar] [CrossRef]
- Marini, V.G.; Martelli, S.M.; Zornio, C.F.; Caon, T.; Simões, C.M.O.; Micke, G.A.; de Oliveira, M.A.L.; Machado, V.G.; Soldi, V. Biodegradable nanoparticles obtained from zein as a drug delivery system for terpinen-4-ol. Quimica Nova 2014, 37, 839–843. [Google Scholar] [CrossRef]
- Luo, Y.; Zhang, B.; Whent, M.; Yu, L.L.; Wang, Q. Preparation and characterization of zein/chitosan complex for encapsulation of α-tocopherol, and its in vitro controlled release study. Colloids Surf. B Biointerfaces 2011, 85, 145–152. [Google Scholar] [CrossRef]
- Wang, C.; Cui, B.; Sun, Y.; Wang, C.; Guo, M. Preparation, stability, antioxidative property and in vitro release of cannabidiol (CBD) in zein-whey protein composite nanoparticles. LWT 2022, 162, 113466. [Google Scholar] [CrossRef]
- Luo, Y.; Teng, Z.; Wang, Q. Development of zein nanoparticles coated with carboxymethyl chitosan for encapsulation and controlled release of vitamin D. J. Agric. Food Chem. 2012, 60, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Wang, T.; Hu, Q.; Luo, Y. Caseinate-zein-polysaccharide complex nanoparticles as potential oral delivery vehicles for curcumin: Effect of polysaccharide type and chemical cross-linking. Food Hydrocoll. 2017, 72, 254–262. [Google Scholar] [CrossRef]
- Cheng, C.J.; Ferruzzi, M.; Jones, O.G. Fate of lutein-containing zein nanoparticles following simulated gastric and intestinal digestion. Food Hydrocoll. 2018, 87, 229–236. [Google Scholar] [CrossRef]
- Chen, H.; Zhong, Q. A novel method of preparing stable zein nanoparticle dispersions for encapsulation of peppermint oil. Food Hydrocoll. 2015, 43, 593–602. [Google Scholar] [CrossRef]
- Zhang, Y.; Niu, Y.; Luo, Y.; Ge, M.; Yang, T.; Yu, L.; Wang, Q. Fabrication, characterization and antimicrobial activities of thymol-loaded zein nanoparticles stabilized by sodium caseinate–chitosan hydrochloride double layers. Food Chem. 2014, 142, 269–275. [Google Scholar] [CrossRef]
- Zou, T.; Gu, L. TPGS Emulsified Zein Nanoparticles Enhanced Oral Bioavailability of Daidzin: In Vitro Characteristics and In Vivo Performance. Mol. Pharm. 2013, 10, 2062–2070. [Google Scholar] [CrossRef]
- Zhang, Y.; Cui, L.; Li, F.; Shi, N.; Li, C.; Yu, X.; Chen, Y.; Kong, W. Design, fabrication and biomedical applications of zein-based nano/micro-carrier systems. Int. J. Pharm. 2016, 513, 191–210. [Google Scholar] [CrossRef]
- Palma, E.; Costa, N.; Molinaro, R.; Francardi, M.; Paolino, D.; Cosco, D.; Fresta, M. Improvement of the therapeutic treatment of inflammatory bowel diseases following rectal administration of mesalazine-loaded chitosan microparticles vs Asamax®. Carbohydr. Polym. 2019, 212, 430–438. [Google Scholar] [CrossRef]
- Pharmacopeia, U. Solutions: Buffer solutions. US Pharmacop. NF27 2009, 32, 861–862. [Google Scholar]
- Schaffazick, S.R.; Guterres, S.S.; Freitas, L.d.; Pohlmann, A.R. Caracterização e estabilidade físico-química de sistemas poliméricos nanoparticulados para administração de fármacos. Quimica Nova 2003, 26, 726–737. [Google Scholar] [CrossRef]
- Dias, S.F.L.; Nogueira, S.S.; Dourado, F.D.F.; Guimarães, M.A.; Pitombeira, N.A.D.O.; Gobbo, G.G.; Primo, F.L.; de Paula, R.C.M.; Feitosa, J.P.A.; Tedesco, A.C.; et al. Acetylated cashew gum-based nanoparticles for transdermal delivery of diclofenac diethyl amine. Carbohydr. Polym. 2016, 143, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Foppoli, A.; Maroni, A.; Moutaharrik, S.; Melocchi, A.; Zema, L.; Palugan, L.; Cerea, M.; Gazzaniga, A. In vitro and human pharmacoscintigraphic evaluation of an oral 5-ASA delivery system for colonic release. Int. J. Pharm. 2019, 572, 118723. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Gamboa, A.; Araujo, V.; Caro, N.; Gotteland, M.; Abugoch, L.; Tapia, C. Spray Freeze-Drying as an Alternative to the Ionic Gelation Method to Produce Chitosan and Alginate Nano-Particles Targeted to the Colon. J. Pharm. Sci. 2015, 104, 4373–4385. [Google Scholar] [CrossRef] [PubMed]
- Mohanraj, V.J.; Chen, Y. Nanoparticles—A review. Trop. J. Pharm. Res. 2006, 5, 561–573. [Google Scholar] [CrossRef] [Green Version]
- Hua, S.; Marks, E.; Schneider, J.J.; Keely, S. Advances in oral nano-delivery systems for colon targeted drug delivery in inflammatory bowel disease: Selective targeting to diseased versus healthy tissue. Nanomedicine: Nanotechnology, Biol. Med. 2015, 11, 1117–1132. [Google Scholar] [CrossRef] [Green Version]
- Tirosh, B.; Khatib, N.; Barenholz, Y.; Nissan, A.; Rubinstein, A. Transferrin as a luminal target for negatively charged liposomes in the inflamed colonic mucosa. Mol. Pharm. 2009, 6, 1083–1091. [Google Scholar] [CrossRef]
- Peterson, C.G.B.; Eklund, E.; Taha, Y.; Raab, Y.; Carlson, M. A new method for the quantification of neutrophil and eosinophil cationic proteins in feces: Establishment of normal levels and clinical application in patients with inflammatory bowel disease. Am. J. Gastroenterol. 2002, 97, 1755–1762. [Google Scholar] [CrossRef]
- Harel, E.; Rubinstein, A.; Nissan, A.; Khazanov, E.; Milbauer, M.N.; Barenholz, Y.; Tirosh, B. Enhanced Transferrin Receptor Expression by Proinflammatory Cytokines in Enterocytes as a Means for Local Delivery of Drugs to Inflamed Gut Mucosa. PLoS ONE 2011, 6, e24202. [Google Scholar] [CrossRef] [Green Version]
- Alam, M.; Tasneem, F.; Kabir, A.L.; Rouf, A.S.S. Study of Drug-Drug and Drug-Food Interactions of Mesalazine Through FTIR and DSC. Dhaka Univ. J. Pharm. Sci. 2019, 18, 257–269. [Google Scholar] [CrossRef] [Green Version]
- Barbosa, L.C.A. Espectroscopia no Infravermelho na Caracterização de Compostos Orgânicos; UFV: Abbotsford, BC, Canada, 2007. [Google Scholar]
- Muthua, S.; Paulrajb, E.I. Molecular structure, spectroscopic (FT-IR, FT-Raman, NMR) studies and first-order molecular hyperpolarizabilities of 5-amino-2 hydroxybenzoic acid (5A2HBA) by ab initio HF and density functional method. J. Chem. Pharm. Res. 2011, 5, 323–339. [Google Scholar]
- Pavia, D.L.; Lampman, G.M.; Kriz, G.S.; Vyvyan, J.R. Introdução à Espectroscopia; Cengage Learning: Boston, MA, USA, 2010. [Google Scholar]
- Tang, P.; Sun, Q.; Zhao, L.; Pu, H.; Yang, H.; Zhang, S.; Gan, R.; Gan, N.; Li, H. Mesalazine/hydroxypropyl-β-cyclodextrin/chitosan nanoparticles with sustained release and enhanced anti-inflammation activity. Carbohydr. Polym. 2018, 198, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Khatri, Z.; Oh, K.W.; Kim, I.-S.; Kim, S.H. Zein/cellulose acetate hybrid nanofibers: Electrospinning and characterization. Macromol. Res. 2014, 22, 971–977. [Google Scholar] [CrossRef]
- Dhanya, A.T.; Haridas, K.R.; Divia, N.; Sudheesh, S. Development of Zein-Pectin nanoparticle as drug carrier. Int. J. Drug Deliv. 2012, 4, 147. [Google Scholar]
- Li, M.-F.; Chen, L.; Xu, M.-Z.; Zhang, J.-L.; Wang, Q.; Zeng, Q.-Z.; Wei, X.-C.; Yuan, Y. The formation of zein-chitosan complex coacervated particles: Relationship to encapsulation and controlled release properties. Int. J. Biol. Macromol. 2018, 116, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Müller, V.; Piai, J.F.; Fajardo, A.R.; Fávaro, S.L.; Rubira, A.F.; Muniz, E.C. Preparation and Characterization of Zein and Zein-Chitosan Microspheres with Great Prospective of Application in Controlled Drug Release. J. Nanomater. 2011, 2011, 928728. [Google Scholar] [CrossRef] [Green Version]
- Markam, R.; Bajpai, A. Functionalization of ginger derived nanoparticles with chitosan to design drug delivery system for controlled release of 5-amino salicylic acid (5-ASA) in treatment of inflammatory bowel diseases: An in vitro study. React. Funct. Polym. 2020, 149, 104520. [Google Scholar] [CrossRef]
- Markam, R.; Bajpai, J.; Bajpai, A. Synthesis of ginger derived nanocarriers (GDNC) and study of in vitro release of 5-amino salicylic acid (5-ASA) as an anti inflammatory drug. J. Drug Deliv. Sci. Technol. 2019, 50, 355–364. [Google Scholar] [CrossRef]
- Wang, X.; Peng, F.; Liu, F.; Xiao, Y.; Li, F.; Lei, H.; Wang, J.; Li, M.; Xu, H. Zein-pectin composite nanoparticles as an efficient hyperoside delivery system: Fabrication, characterization, and in vitro release property. LWT 2020, 133, 109869. [Google Scholar] [CrossRef]
- Malviya, T.; Joshi, S.; Dwivedi, L.M.; Baranwal, K.; Shehala; Pandey, A.K.; Singh, V. Synthesis of Aloevera/Acrylonitrile based Nanoparticles for targeted drug delivery of 5-Aminosalicylic acid. Int. J. Biol. Macromol. 2018, 106, 930–939. [Google Scholar] [CrossRef]
- Argin, S.; Kofinas, P.; Lo, Y.M. The cell release kinetics and the swelling behavior of physically crosslinked xanthan–chitosan hydrogels in simulated gastrointestinal conditions. Food Hydrocoll. 2014, 40, 138–144. [Google Scholar] [CrossRef]
- Zhang, Y.; Huo, M.; Zhou, J.; Zou, A.; Li, W.; Yao, C.; Xie, S. DDSolver: An Add-In Program for Modeling and Comparison of Drug Dissolution Profiles. AAPS J. 2010, 12, 263–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siepmann, J.; Peppas, N.A. Modeling of drug release from delivery systems based on hydroxypropyl methylcellulose (HPMC). Adv. Drug Deliv. Rev. 2012, 64, 163–174. [Google Scholar] [CrossRef]
- Stern, H.L.F.D.A. Critical Reviews in Solid State and Materials Science. Mater. Sci. 1983, 11, 123–187. [Google Scholar]
- Bizerra, A.; Silva, V. Sistemas DE LIBERAção CONTROLADA: Mecanismos e aplicações. Revista Saúde e Meio Ambiente-RESMA 2016, 3, 1–12. [Google Scholar]









| Formulation | NP-ZN | NP-ZN-MSZ |
|---|---|---|
| Size | 218 ± 23 nm | 266.6 ± 52 nm |
| Polydispersity index | 0.20 ± 0.1 | 0.14 ± 0.1 |
| ζ Potential | −39.3 ± 2 mV | −42.4 ± 5.31 mV |
| Drug loaded | - | 43.8 µg/mg de NP |
| Encapsulation efficiency | - | 45% |
| Yield | 70% | 65% |
| Peppas–Sahlin Model | |||
|---|---|---|---|
| Medium | R2adj | K1 | K2 |
| pH 1.2 | 0.99 | 1.62 | 0.36 |
| pH 6.8 | 0.92 | −5.72 | 6.12 |
| Korsmeyer–Peppas Model | |||
| Medium | R2adj | K | n |
| pH 7.4 | 0.82 | 99.3 | −0.79 |
| Time (Days) | Size (nm) | Polydispersion Index | Zeta Potential (mV) |
|---|---|---|---|
| 0 | 266.6 ± 52 nm | 0.14 ± 0.1 | −42.4 ± 5.31 mV |
| 30 | 296.2 ± 25 nm | 0.11 ± 0.02 | −40.4 ± 6 mV |
| 60 | 300.6 ± 81 nm | 0.12 ± 0.1 | −41.6 ± 9.3 mV |
| 90 | 269 ± 31 nm | 0.14 ± 0.1 | −37.4 ± 8.24 mV |
| 120 | 291.6 ± 32 nm | 0.13 ± 0.03 | −32.4 ± 6.7 mV |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima, I.B.C.; Moreno, L.C.G.A.I.; Peres, A.V.; Santana, A.C.G.; Carvalho, A.; Chaves, M.H.; Lima, L.; Sousa, R.W.; Dittz, D.; Rolim, H.M.L.; et al. Nanoparticles Obtained from Zein for Encapsulation of Mesalazine. Pharmaceutics 2022, 14, 2830. https://doi.org/10.3390/pharmaceutics14122830
Lima IBC, Moreno LCGAI, Peres AV, Santana ACG, Carvalho A, Chaves MH, Lima L, Sousa RW, Dittz D, Rolim HML, et al. Nanoparticles Obtained from Zein for Encapsulation of Mesalazine. Pharmaceutics. 2022; 14(12):2830. https://doi.org/10.3390/pharmaceutics14122830
Chicago/Turabian StyleLima, Izabela Borges C., Lina Clara G. A. I. Moreno, Ana Victória Peres, Ana Cristina Gramoza Santana, Adonias Carvalho, Mariana H. Chaves, Lorena Lima, Rayran Walter Sousa, Dalton Dittz, Hercília M. L. Rolim, and et al. 2022. "Nanoparticles Obtained from Zein for Encapsulation of Mesalazine" Pharmaceutics 14, no. 12: 2830. https://doi.org/10.3390/pharmaceutics14122830