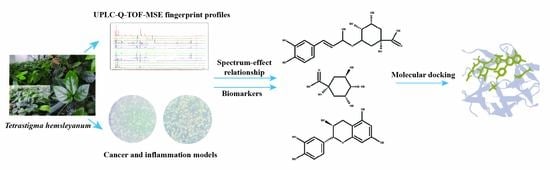
1. Introduction
Tetrastigma hemsleyanum Diels et Gilg. (
T. hemsleyanum), also known as Sanyeqing, Jinxiandiaohulu, Shefuzi, or Shilaoshu in China, is a widely used herbaceous plant in traditional Chinese medicine [
1,
2]. This rare plant grows in subtropical areas of mainland China. It was used to treat fever for the first time in the 3rd century in the book Ming Yi Bie Lu [
2]. In the 16th century, the Compendium of Materia Medica focused on the plant’s ability to dispel heat and toxins as well as promote blood circulation [
3]. Today,
T. hemsleyanum is commonly used in folk medicine to treat high fever, infantile febrile convulsion, pneumonia, snake bite, and jaundice. It has been cited in both the Flora of China and the Chinese Pharmacopeia (2015 edition) [
3,
4,
5].
T. hemsleyanum root tubers have been used as herbal medicine in Chinese local pharmacopoeia, while its leaves are popularly consumed as a functional tea or dietary supplement in local regions of China [
6]. Due to its wide range of pharmacological effects, the plant is cultivated on a large scale.
Currently, systematic chemical profile studies have been conducted for different organs of
T. hemsleyanum, including roots, stems, and leaves [
1]. The chemical constituents of
T. hemsleyanum are mainly flavonoids and polyphenolic acids, such as kaempferol, apigenin-8-C-glucoside, chlorogenic acid, and catechins [
2]. Recent pharmacological studies have shown that kaempferol and other components of
T. hemsleyanum possess a broad range of activities [
7,
8]. For example, in a variety of human cancer cell lines, kaempferol has exhibited a wide range of anti-cancer activities, such as apoptosis, cell cycle arrest, downregulation of tissue transformation-related markers, and phosphoinositide 3-kinase/protein kinase B signaling pathways [
9,
10,
11]. In addition, chlorogenic acid has a strong anti-inflammatory effect on the metabolism of arachidonic acid by inhibiting the activation of inflammatory factors such as HIF-1a, ICAM-1, VCAM-1, TNF-α, IL-6, and nuclear factor kappa B (NK-KB) p65. This protects against cerebral ischemia/reperfusion injury and liver fibrosis [
12,
13]. Due to its remarkable anti-cancer and anti-inflammatory effects,
T. hemsleyanum has been suggested as a promising option for the treatment of various pathological conditions in clinical settings.
Liver cancer is one of the six most common forms of cancer worldwide, with a very high mortality rate [
14]. HepG2 cells originate from a human liver embryonic tumor cell line, have a similar phenotype to hepatocytes, and have been widely used as an in vitro model to evaluate the potential of drug-drug interactions [
15,
16]. HuH-7 is another hepatocellular carcinoma cell line, which is highly correlated with primary human hepatocytes in gene expression profiles of drug-metabolizing enzymes and transporters [
17,
18]. Therefore, a large number of studies have been conducted on HepG2 and HuH-7 cell lines to evaluate the induction and reversal of human hepatocellular carcinoma resistance treatment by natural products [
19,
20]. Inflammation is a defensive response of the body to stimuli triggered by tissue damage, such as invading microbes and tissue injury [
21]. Due to the massive secretion of effectors downstream of the inflammatory pathway, inflammatory inducers trigger the production of a large number of various inflammatory cytokines, which in turn alter the function of many tissues and organs [
22]. Inflammatory cytokines usually play a role in activating macrophages and promoting cell-mediated immune responses against invasive intracellular pathogens, such as tumor-necrosis factor-α (TNF-α), IL-1, IL-6, and many others [
23]. Inflammation can contribute to cancer progression, and conversely, cancer can induce inflammatory processes [
24]. Screening of key components from natural products that antagonize inflammatory cells and cytokines, inhibit tumor growth, and enhance anti-cancer immunity will help find new strategies to prevent or treat cancer.
Currently, the effective components of T. hemsleyanum are not clearly known, which presents a challenge to the quality control of T. hemsleyanum entering the market circulation. Thus, we proposed a strategy to establish the spectrum-effect relationship of T. hemsleyanum using UPLC-Q-TOF-MS and activity analysis. The correlations between the common peaks and anti-inflammatory and anti-cancer activities were studied by principal components analysis (PCA) and partial least squares regression (PLSR) statistical methods to further reveal the active substance basis of T. hemsleyanum. This provides a basis for exploring the quality markers of the medicinal plant T. hemsleyanum, establishing its quality evaluation method, and investigating the mechanism of action of its medicinal components.
4. Materials and Methods
4.1. Reagents and Materials
Methanol (MeOH), formic acid, acetonitrile, ethanol (all liquid chromatography–mass spectrometry (LC–MS) grade), dulbecco’s modified eagle medium/nutrient mixture F12 (DMEM/F-12), penicillin-streptomycin (100×), phosphate buffered saline (PBS), fetal bovine serum, and trypsin were obtained from Thermo Fisher Scientific (Waltham, MA, USA). Lipopolysaccharide (LPS) was purchased from Sigma-Aldrich (Shanghai, China). Cell Counting Kit-8 (CCK-8) reagent was purchased from Vazyme Biotech (Nanjing, China). The reverse transcription kit and Real-time Quantitative polymerase chain reaction (RT-PCR) kit are purchased from TOYOBO Life Sciences Company (Shanghai, China). RNA extraction kits are available at Clpbio (Montclair, CA, USA), and RNA-Quick Purification Kit was purchased from Shanghai Yishun Biotechnology company (Shanghai, China).
T. hemsleyanum samples were harvested from Zhejiang, Anhui, Fujian, Guizhou, Guangxi, Guangdong, Jiangxi, and Sichuan Province, China. The sources of batches of samples are shown in
Supplementary Table S1. The voucher specimens were deposited in the Key Laboratory of Plant Secondary Metabolism, Zhejiang Sci-Tech University, China (No. 2021011–42).
4.2. Extraction
The dried powder T. hemsleyanum was extracted with 80% aqueous methanol using ultrasound-assisted extraction (UAE) with an ultrasonic power setting of 200 W, 40 Hz, at 60 °C for 30 min. The combined filtrates were filtered and then rotary evaporated until there was no alcoholic smell. Finally, freeze-dried to dry cream extraction, stored in the refrigerator at 4 °C.
4.3. Liquid Chromatography Tandem Mass Spectrometry (LC–MS/MS) Analysis for Metabolite Identification
About 0.01 g of root extract was homogenized by adding 10 mL of MeOH/H2O (70:30, v/v), centrifuged for 5 min, and sonicated for 20 min. Then the supernatant was centrifuged at 10,000 rpm for 10 min and passed through a membrane with a pore size of 0.22 μm. The filtrate was transferred to sample bottles for LC-MS/MS analysis.
Chromatographic separation was achieved using an Accucore BEHC18 column (1.7 μm, 2.1 mm × 100.0 mm; Waters Corporation, Milford, MA, USA). The column temperature and flow rate were set at 30 °C and 0.3 mL·min−1, respectively. The column temperature was 30 °C and the flow rate was 0.3 mL·min−1. The injection volume of the column was 2 μL and the detection wavelength was 200~600 nm, respectively. The mobile phase consisted of 0.1% formic acid (A) and acetonitrile (B) with a gradient elution program of 95% A (0–1 min), 95–90% A (1–3 min), 90–80% A (3–6 min), 80–30% A (6–16 min) and 30–0% A (16–20 min).
An electrospray ionization source (ESI) was used to connect the above UPLC system to a Synapt G2 mass spectrometer detector (Waters Corporation, Milford, MA, USA). Argon was used as the desolvation and collision gas. The full-scan data ranged from 70 to 1200 Da; the source temperature was 120 °C; the capillary voltage was 2.6 kV; and the sample cone was 40 V. The low collision energy was set to 2 eV, and the high collision energy was steadily increased from 20 eV to 45 eV. The desolvation temperature was set at 450 °C with a desolvation gas flow of 800 L·h−1.
4.4. Assessing the Effect of T. hemsleyanum Extract on Cancer Cell Proliferation
4.4.1. Cell Culture
The human hepatocellular carcinomas (HepG2 cell line) were donated by Liu Kuancheng’s research group at Sun Yat-Sen University (Guangzhou, China), and the human hepatocellular carcinomas (HuH-7 cell line) were donated by Li Gongchu’s research group at Zhejiang Sci-Tech University (Hangzhou, China). The cells were cultured in Dulbecco’s Modified Eagle Medium/Nutrient Mixture F12 (DMEM/F-12, ThermoFisher Scientific, Waltham, MA, USA) and supplemented with 10% fetal bovine serum (ThermoFisher Scientific, Waltham, MA, USA), 5 mL penicillin–streptomycin (100×, ThermoFisher Scientific, Waltham, MA, USA) under standard conditions (37 °C, 5% CO2).
4.4.2. Cell Viability Detected by CCK-8 Assay
The 100 μL of cell suspension (1 × 105/mL) was inoculated into 96-well culture plates, and 100 μL of PBS was added around the 96-well culture plates and incubated until the cells were plastered. The medium in the wells was removed, and 100 μL of the blank medium was added to the blank control group. The test groups were treated with 100 μL of samples at final concentrations of 0, 50, 100, 150, 200, 250, and 500 μg/mL for 24 h, and then 10 μL of MTT solution was added to continue the incubation for 2 h (6 replicate wells were set up for each dose group, and each group was repeated three times). The temperature of the microplate reader was set at 37 °C, and the absorbance was measured every 30 s for 5 min at 450 nm.
4.5. Assessment of the Effect of T. hemsleyanum Extract on Inflammatory Model Injury
4.5.1. Cytotoxicity Assays
To evaluate the effect of
T. hemsleyanum and LPS on the viability of RAW264.7 cells, cytotoxicity assays were performed using the Cell Counting Kit-8. Briefly, cells in the logarithmic growth phase were subjected to the passage procedure, and finally, 100 μL of cell suspension (with a cell density of 1 × 10
5 /mL) was added to each well of the 96-well plate. 100 μL of PBS was added around the 96-well plate, and the blank control was a cell-free cell culture medium. The final concentrations of the master mix were 0, 0.1, 1, 10, and 100 μg/mL, and 6 replicate wells were added for each concentration, and each group was repeated three times. For the blank control, only 100 μL of cell culture medium was added. After incubation for 12 and 24 h, 10 μL of CCK-8 was added to each well and incubated for 2 h. The absorbance (OD) was measured at 450 nm on an enzyme marker. In addition, the concentration gradient of cytotoxicity of
T. hemsleyanum extract on RAW264.7 was the same as that in
Section 4.4.2.
4.5.2. LPS-Induced RAW264.7 Cell Model
RAW264.7 cells in the logarithmic growth phase (1 × 10
5/mL) were inoculated with 2.5 mL of 96-well culture plates and incubated in a CO
2 incubator for 6 h. The culture medium was replaced with a LPS-containing medium, so that the LPS concentration was “
Section 4.5.1”, the optimal concentration and the induction time was “
Section 4.5.1” optimal time. Each group was set up with two replicate wells and replicated three times. For the blank control group, 2.5 mL of culture medium was added, and for the complete control group, 2.5 mL of culture medium containing 1 × 10
6 cells was added. The RNA was extracted from the supernatant of the cells after 24 h. The internal reference gene was selected as GAPDH, and the contents of IL-6, TNF-α, and IL-1β were measured. According to the SYBR method, 2^
−△△CT values were calculated to evaluate the feasibility of the LPS-induced inflammation model in RAW264.7 cells.
4.5.3. Effect of T. hemsleyanum Extract on the Secretion Function of Pro-Inflammatory Cytokines in an Inflammatory Model
LPS-induced RAW264.7 cell model in
Section 4.5.2. After 12 h of LPS incubation, the culture was replaced with prepared
T. hemsleyanum extract at 2.5 mL for each group, 2.5 mL of culture medium for the blank control group, and 2.5 mL of culture medium containing 1 × 10
6 cells for the complete control group. Similarly, the internal reference gene GAPDH was selected to determine the content of IL-6, TNF-α, and IL-1β. According to the SYBR method, 2^
−△△CT values were calculated to evaluate the effect of
T. hemsleyanum extract on the secretion of pro-inflammatory cytokines in an inflammatory model.
4.5.4. Molecular Docking
The crystal structure of IL-1β was obtained from the RCSB Protein Data Bank (PDB). 4G6M(PDB ID) is the crystal structure of human IL-1beta in complex with the therapeutic antibody binding fragment of gevokizumab. PyMol was used to extract IL-1β from the whole structure. The crystal structure of IL-1β was selected based on the best resolution available. The protein preparation module of Schrodinger’s Maestro Molecular Modeling Suit was utilized for the preparation of the protein crystal structure. The SiteMap module of the Maestro molecular modeling package was used to find the potential pockets of IL-1β. Sobia Halim’s article helped to find the most possible pocket [
49]. Glide was used for all the docking simulations and calculations. Maestro interface was used for visualization of the ligand-target interactions, including hydrogen bonding, ion-pair interactions, hydrophobic interactions, and the binding modes of the identified compounds.
4.6. Data Analysis
Mass spectrometry data were collected using MassLynx V4.2 software (Waters Corporation, Milford, MA, USA) for automatic peak identification, peak matching, peak alignment, peak extraction, peak integration, and peak normalization. The metabolites and their possible cleavage modes were identified using secondary mass spectrometry data from Unifi (Waters Corporation, Milford, MA, USA), online databases (SciFinder, ChemSpider, and PubMed), and data from previous studies. The median inhibitory concentration (IC50) of different samples was obtained by processing the efficacy data with GraphPad software. Unsupervised principal component analysis (PCA) was performed using ChemDateSolution software to obtain overall metabolic differences between sample regions and determine the magnitude of variation between samples within groups. The spectrum-effect relationships study combined with partial least squares (PLS) was performed to identify the chemical substance basis associated with the pharmacodynamic activity.
5. Conclusions
In this study, the UPLC-Q-TOF-MS fingerprints of extracts from T. hemsleyanum were established, and their medicinal effects were evaluated by three kinds of cell lines. Therefore, T. hemsleyanum appears to be a valuable natural source for developing anti-cancer and anti-inflammatory treatments. In addition, the relationships between the bioactive properties and UPLC-Q-TOF-MS fingerprints were analyzed by PCA and PLSR methods, and the main representative active compounds were finally discovered, such as chlorogenic acid, quinic acid, kaempferol 3-rutinoside, and apigenin 8-C-glucoside-arabinoside. The screened quality markers can provide a reference for establishing accurate, universal, and measurable scientific quality control and evaluation methods for T. hemsleyanum. The strategy might provide a valuable reference mode to elucidate the material basis of the complex system of TCM.